Ionic Bonding Basics of Chemical Bonds Ionic Bonds
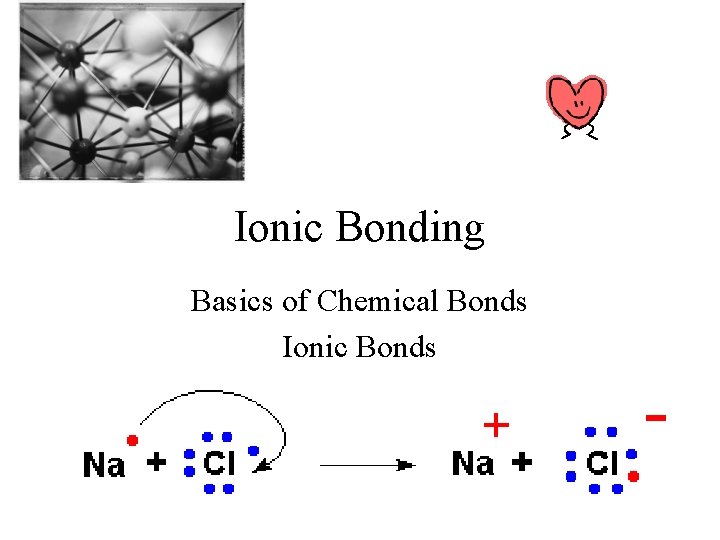
Ionic Bonding Basics of Chemical Bonds Ionic Bonds

Chemical Bonds Force that holds atoms or groups of atoms together Ionic Bond- transfer of valence electrons

Octet Rule • An outer shell of eight valence electrons is most desirable/stable • Electrons gained/lost or shared in the process • Lowest energy configuration • Example: sodium (Na)

Learning Check • Group 18 (Noble Gases) – rarely form compounds. • Why?
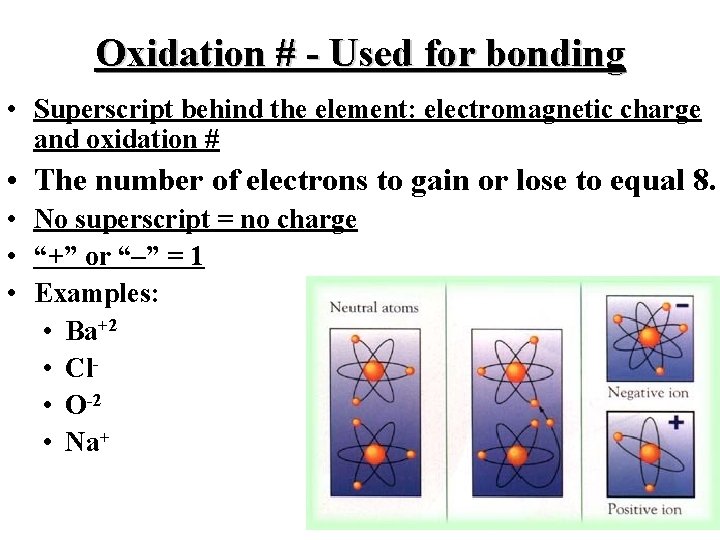
Oxidation # - Used for bonding • Superscript behind the element: electromagnetic charge and oxidation # • The number of electrons to gain or lose to equal 8. • No superscript = no charge • “+” or “–” = 1 • Examples: • Ba+2 • Cl • O-2 • Na+
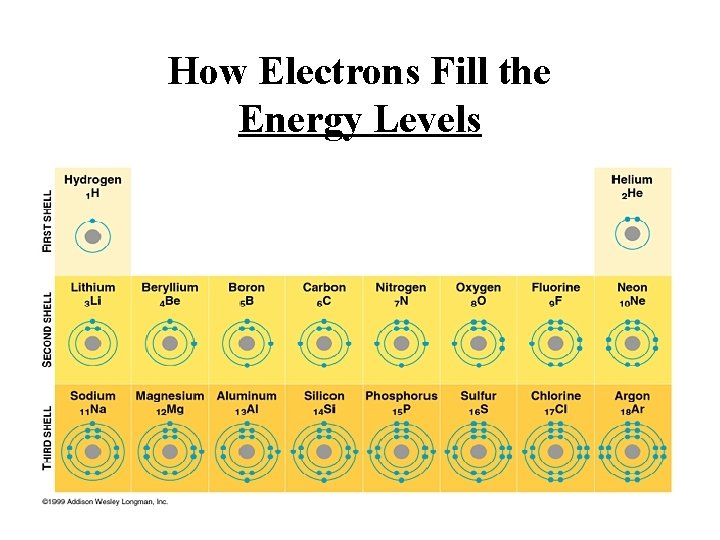
How Electrons Fill the Energy Levels

Formation of Ions from Metals Ionic compounds result when metals react with nonmetals Metals lose electrons to match the number of valence electrons of their nearest noble gas Positive ions form when the number of electrons are less than the number of protons Group 1 metals ion 1+ Group 2 metals ion 2+ Group 13 metals ion 3+

The Formation of a Sodium Ion Sodium atom Na 11 p+ 11 e 0 – e Sodium ion Na + 11 p+ 10 e 1+

The Formation of a Magnesium Ion Magnesium atom Mg 12 p+ 12 e 0 – 2 e Magnesium ion Mg 2+ 12 p+ 10 e 2+

Some Common Ions With a Positive Charge (cations) Group 1 Group 2 Group 13 (3 A) H+ Mg 2+ Al 3+ Li+ Ca 2+ Na+ Sr 2+ K+ Ba 2+

Ionic Bonding
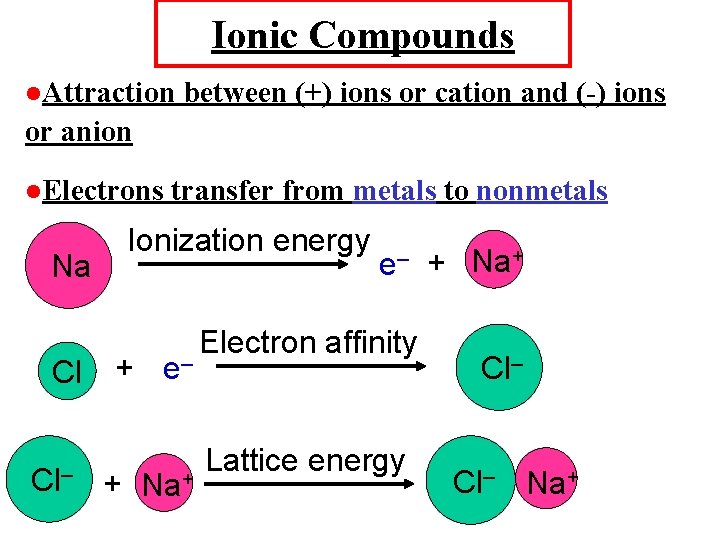
Ionic Compounds Attraction between (+) ions or cation and (-) ions or anion Electrons Na Cl Cl– transfer from metals to nonmetals Ionization energy + e– + Na+ Electron affinity Lattice energy Cl– Na+

Ions from Nonmetal Elements In ionic compounds, nonmetals in groups 15, 16, and 17 gain electrons from metals Nonmetals add electrons to achieve the octet arrangement (8 valence electrons) Nonmetal ionic charge: 3 -, 2 -, or 1 -
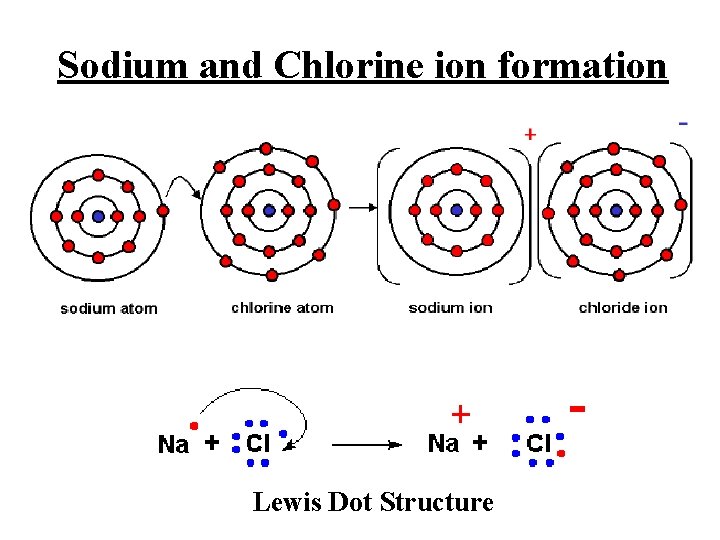
Sodium and Chlorine ion formation Lewis Dot Structure

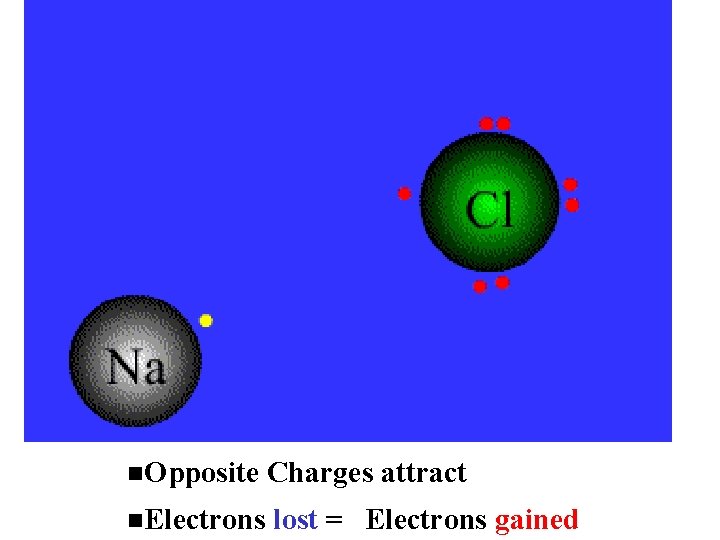
Opposite Charges attract Electrons lost = Electrons gained

Formulas of Ionic Compounds o Write symbol for (+) ion first followed by (–) ion. o Assign subscripts to assure compound is electrically neutral

Ionic Solids Dissociate in Water

Properties of Ionic Compounds • • • Solids at room temperature Nonconductive as solids Dissolve in water by dissociation Conductive when dissolved High melting points Hard, Brittle Crystals

Polyatomic Ions • • Charged group of covalently bonded atoms. Bonds ionically to oppositely charged ions. Listed on STAAR Chart Examples: nitrate (NO 3)-1, sulfate (SO 42 -), ammonium (NH 4+), phosphate (PO 43 -)

Steps for Naming Ionic Compounds Step 1. Name each element Step 2. End the last element in –ide Example: Mg. Cl 2 Step 1. Magnesium Chlorine Step 2. Magnesium Chloride

Naming Ionic Compounds Containing Transition Elements • Name of metal (+) ion first; then nonmetal (-) ion • Many transition elements have variable charges. • Historical suffix usage • “-ic” for higher of two; • “-ous” for lower • Modern approach • Name of metal followed by Roman numeral indicating charge
- Slides: 22