IONIC BONDING An electrical attraction between two oppositely


IONIC BONDING An electrical attraction between two oppositely charged atoms or groups of atoms. Happens when an atom of a nonmetal takes one or more electrons from an atom of a metal so both atoms end up with eight valence electrons
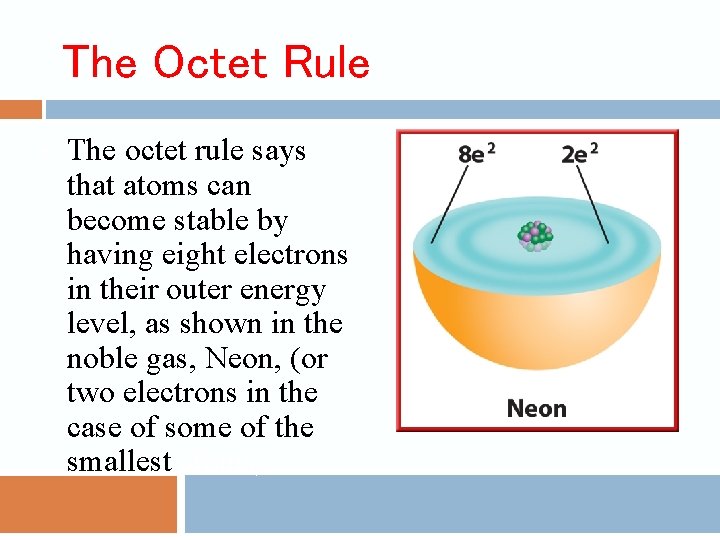
The Octet Rule • The octet rule says that atoms can become stable by having eight electrons in their outer energy level, as shown in the noble gas, Neon, (or two electrons in the case of some of the smallest atoms).

Electron Dots For Cations Metals will have few valence electrons These will come off Ca

Electron Dots For Cations Metals will have few valence electrons These will come off Forming positive ions 2+ Ca

Electron Dots For Anions Nonmetals will have many valence electrons. They will gain electrons to fill outer shell. P 3 P

IONIC COMPOUNDS metal with nonmetal + ion - ion Ca+ion anion

Predicting Oxidation Number or Charge from Periodic Table
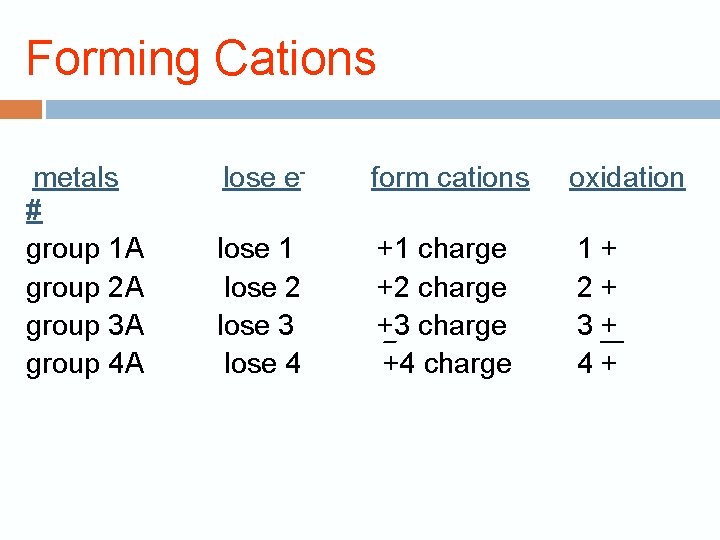
Forming Cations metals # group 1 A group 2 A group 3 A group 4 A lose e- form cations oxidation lose 1 lose 2 lose 3 lose 4 +1 charge +2 charge +3 charge +4 charge 1+ 2+ 3+ 4+

Forming Anions Non-metals group 7 A group 6 A group 5 A gain egain 1 gain 2 gain 3 form anions -1 charge -2 charge -3 charge oxidation # 123 -
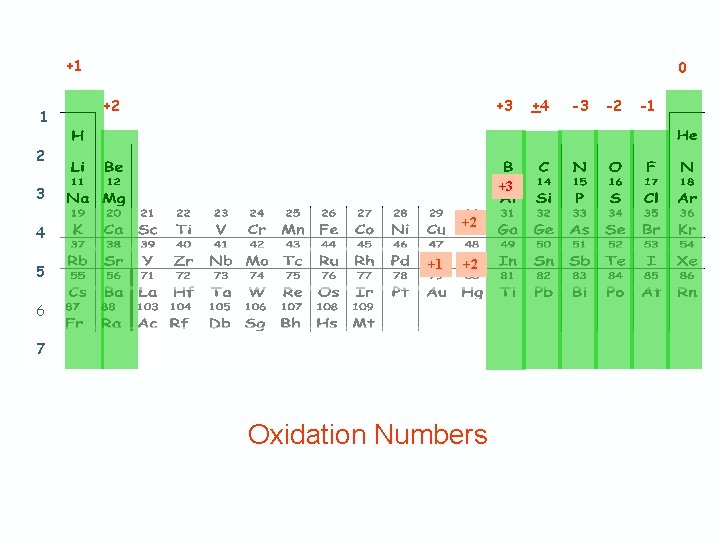
+1 1 0 +2 +3 3 +2 4 5 +1 +2 6 7 Oxidation Numbers +4 -3 -2 -1

IONIC BONDING Neutral atoms come near each other. Electron(s) are transferred from the Metal atom to the Non-metal atom. They stick together because of electrostatic forces, like magnets.

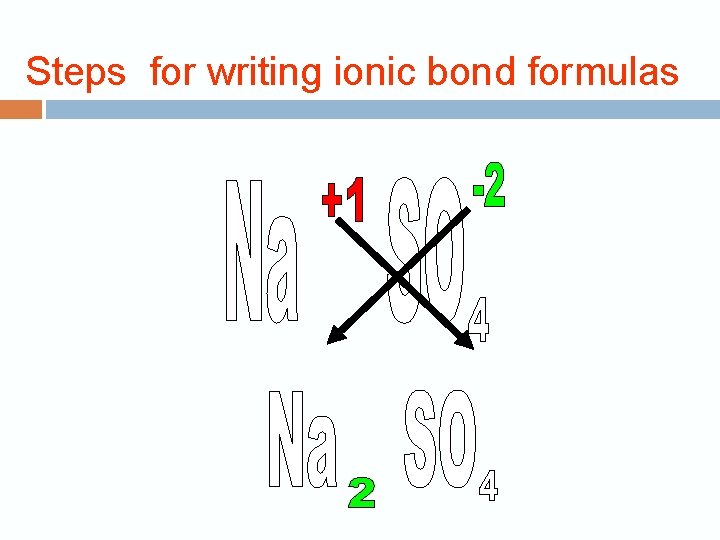
Steps for writing ionic bond formulas 1. Determine the oxidation numbers for the two elements or ions. Ca = +2, Cl = -1

Steps for writing ionic bond formulas 2. Write the chemical symbols in the correct order, with the metal ion first and write the oxidation numbers as superscripts. Ca 2+ Cl -1

Steps for writing ionic bond formulas 3. Crisscross the numbers only—not the charge signs—writing the oxidation number of one element as a subscript for the other. Don’t write number 1 either. 2+ 1 - Ca Cl Ca 1 Cl 2 Ca Cl 2

Steps for writing ionic bond formulas 4. Determine whether the formula is in its simplest form. Reduce the subscripts to the simplest from by dividing by a common denominator. Ca+2 O-2 Ca 2 O 2 Ca. O = Ca 2 O 2

Steps for writing ionic bond formulas 5. Check the formula by calculating the total positive and negative charges and confirming that the total charge on the compound is zero. Ca. Cl 2 Oxidation Number Subscript Calcium (+2) x (1) = +2 Chlorine (-) x (2) = -2

Practice these! magnesium and oxygen aluminum and bromine sodium and sulfur potassium and nitrogen Mg. O Al. Br 3 Na 2 S K 3 N

More Practice! potassium bromide calcium oxide Silver fluoride KBr Ca. O Ag. F

Polyatomic ions Groups of atoms that stick together as a unit, and have a charge PO 43 - phosphate CO 32 - carbonate C 2 H 3 O 41 - acetate Names often end in –ate or –ite

Steps for writing ionic bond formulas 6. POLYATOMIC IONS ACT JUST LIKE ANY OTHER NEGATIVE ION WHEN BONDING Treat polyatomic ion as single unit— don’t change it in any way!
Steps for writing ionic bond formulas

Steps for writing ionic bond formulas polyatomic ions are enclosed in parenthesis if taken more than once in the formula. A subscript outside the parenthesis multiplies everything inside.
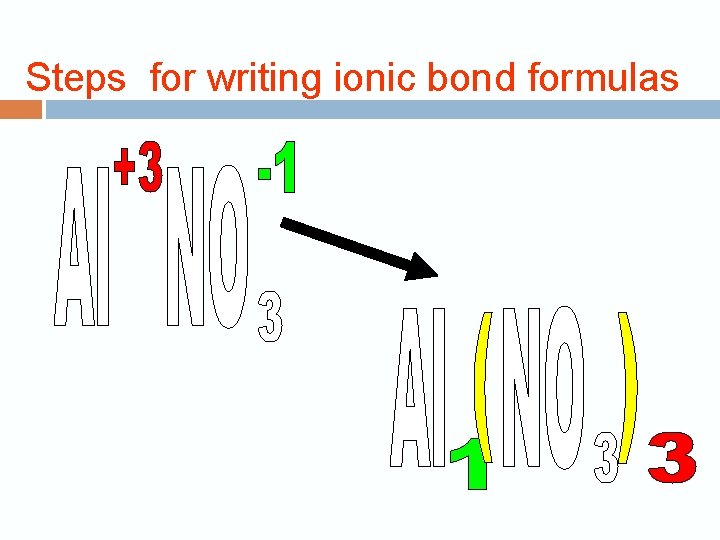
Steps for writing ionic bond formulas
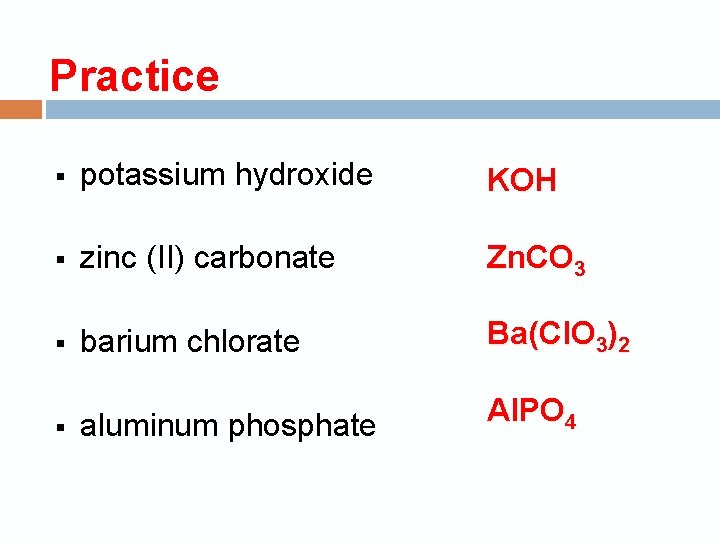
Practice § potassium hydroxide KOH § zinc (II) carbonate Zn. CO 3 § barium chlorate Ba(Cl. O 3)2 aluminum phosphate Al. PO 4 §
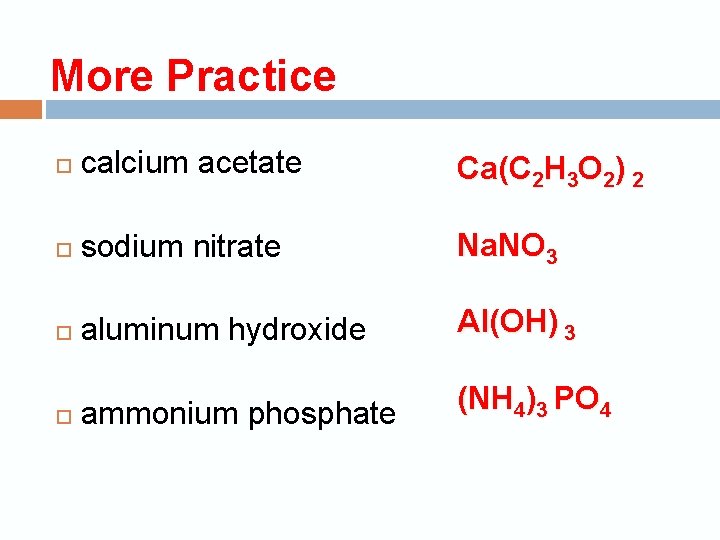
More Practice calcium acetate Ca(C 2 H 3 O 2) 2 sodium nitrate Na. NO 3 aluminum hydroxide Al(OH) 3 ammonium phosphate (NH 4)3 PO 4

Naming Ionic Compounds 1. name cation - then anion 2. if anion (nonmetal) is a single element …. . then name ends in “-ide”

Naming Ionic Compounds 3. If metal have more than one possible charge (include roman numeral in parenthesis to give charge)* *All metals except: Ag+1, Zn+2, Cd+2, Al+3
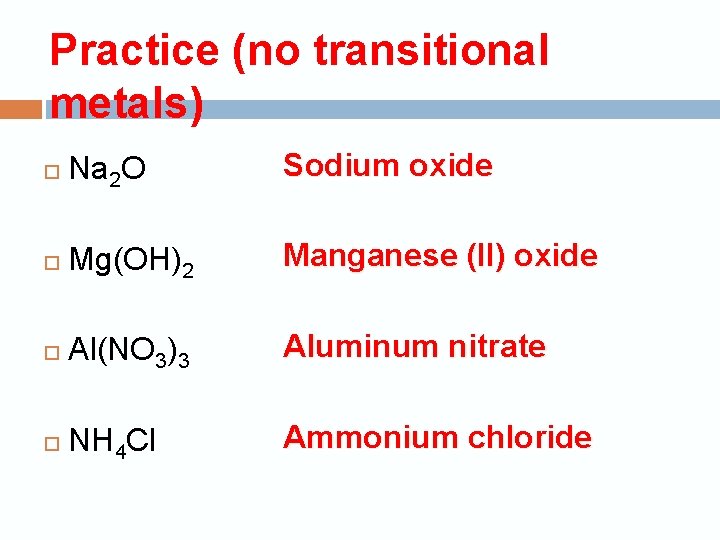
Practice (no transitional metals) Na 2 O Sodium oxide Mg(OH)2 Manganese (II) oxide Al(NO 3)3 Aluminum nitrate NH 4 Cl Ammonium chloride
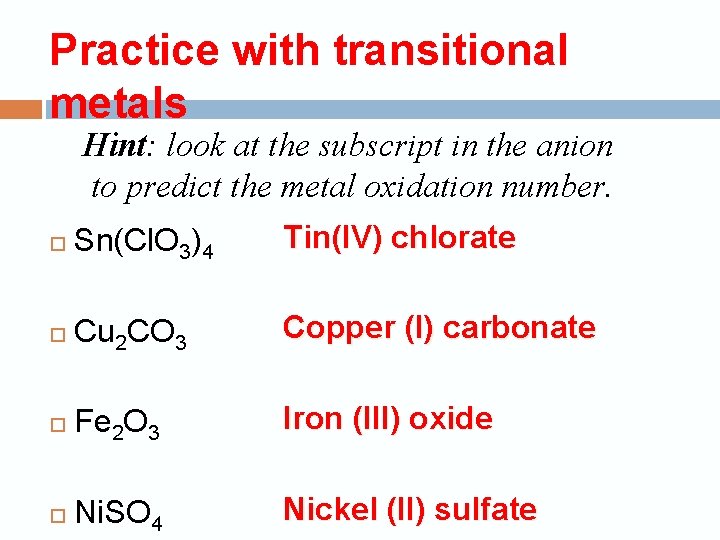
Practice with transitional metals Hint: look at the subscript in the anion to predict the metal oxidation number. Sn(Cl. O 3)4 Tin(IV) chlorate Cu 2 CO 3 Copper (I) carbonate Fe 2 O 3 Iron (III) oxide Ni. SO 4 Nickel (II) sulfate

Properties of Ionic Compounds Crystalline structure. A regular repeating arrangement of ions in the solid. Ions are strongly bonded. Structure is rigid. High melting points- because of strong forces between ions.
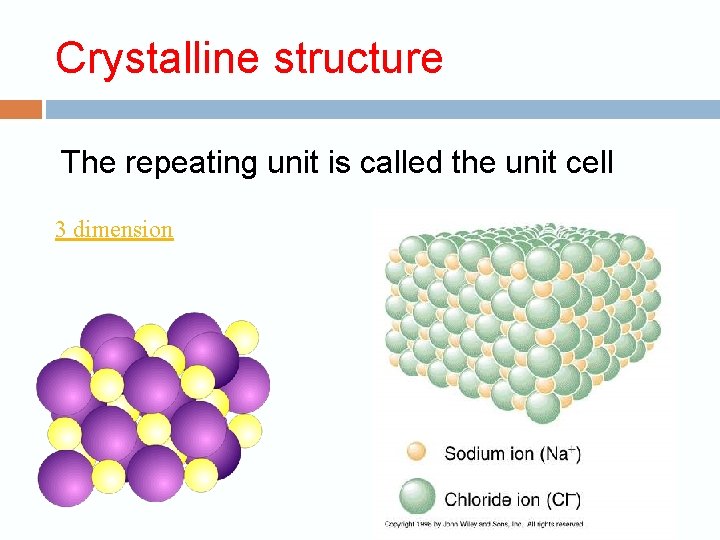
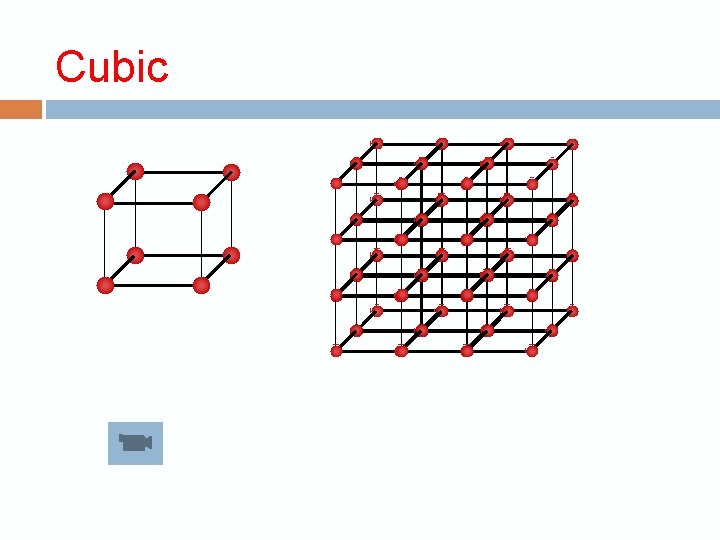
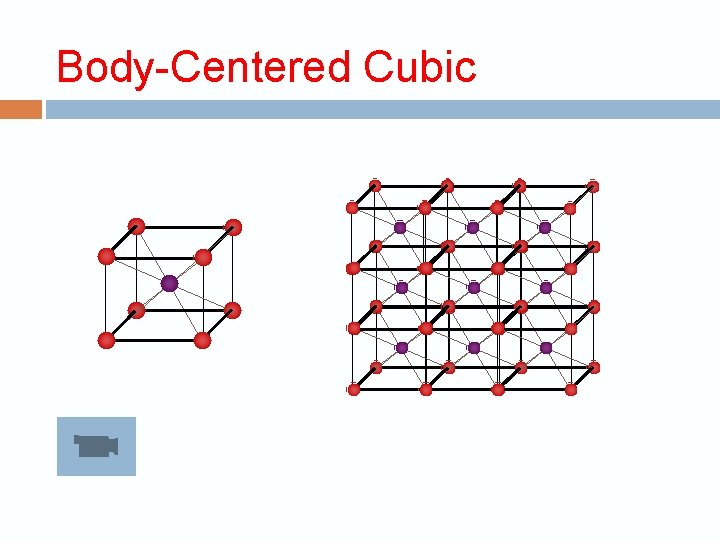
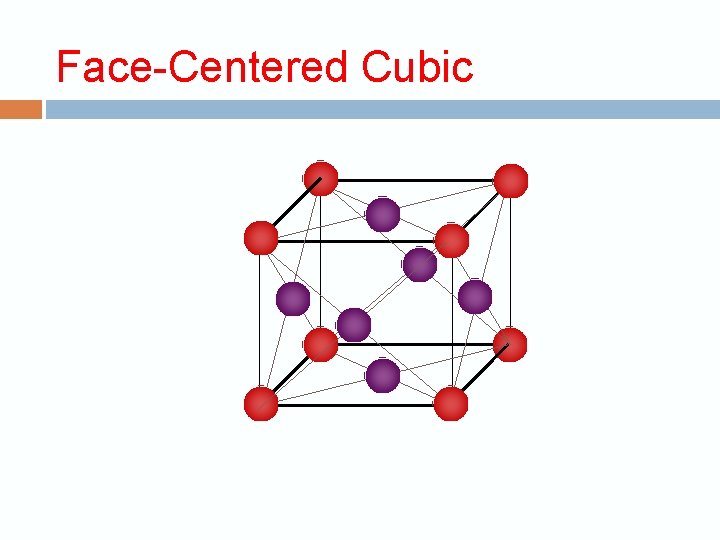
Crystalline structure The repeating unit is called the unit cell 3 dimension
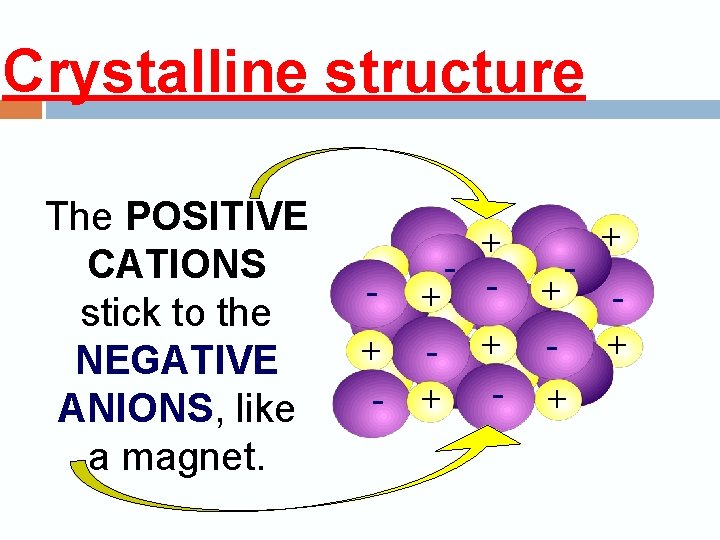
Crystalline structure The POSITIVE CATIONS stick to the NEGATIVE ANIONS, like a magnet. + - + - + + - - + +
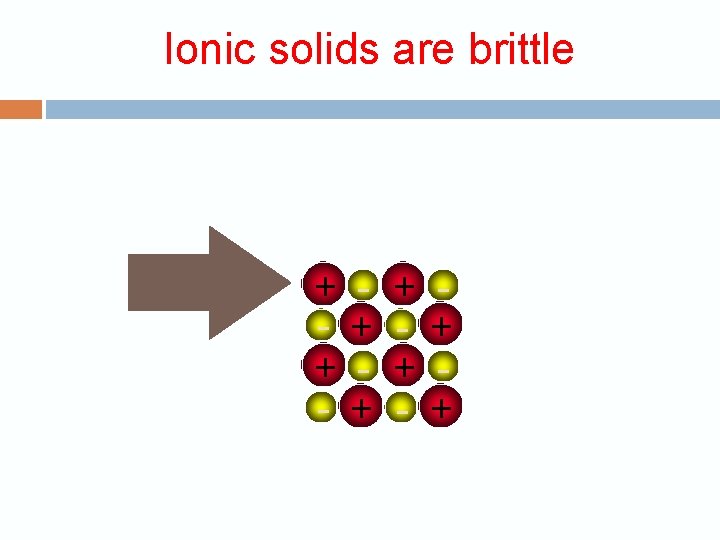
Ionic solids are brittle + + - + +
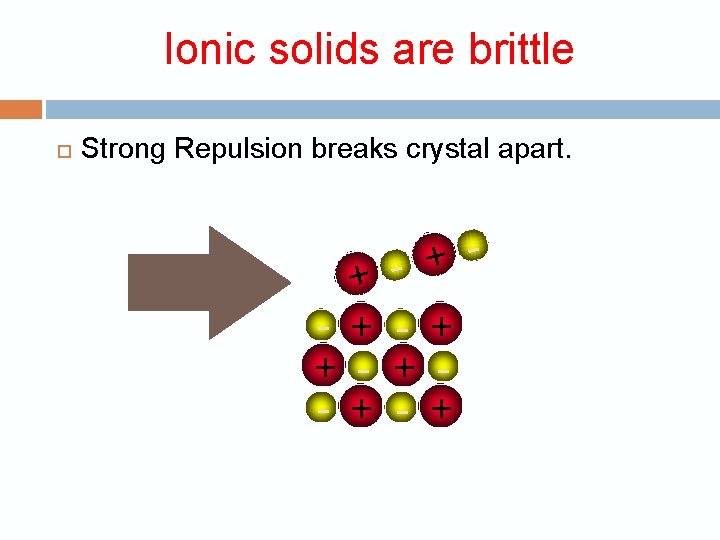
Ionic solids are brittle Strong Repulsion breaks crystal apart. + + - + - + - +
Cubic
Body-Centered Cubic
Face-Centered Cubic

Do they Conduct? Conducting electricity is allowing charges to move. In a solid, the ions are locked in place. Ionic solids are insulators. When melted, the ions can move around. Melted ionic compounds conduct. First get them to 800ºC. Dissolved in water they conduct.

- Slides: 40