Ionic Bond Metal Nonmetal Metals lose valence electrons


Ionic Bond: Metal + Nonmetal • Metals: lose valence electrons – form cation (+ ion) • Nonmetals: gain electrons – form anion (- ion)

IONIC BONDING • Involves transfer of electrons – electrons go from metal to nonmetal • # electrons Lost by Cation (metal) = # electrons Gained by Anion (nonmetal)

Metals are losers! • Na has configuration 2 -8 -1 • Na loses 1 electron → Na+1 – Na+1 configuration is 2 -8 (Ne) • What can take the valence electron from Na? ?
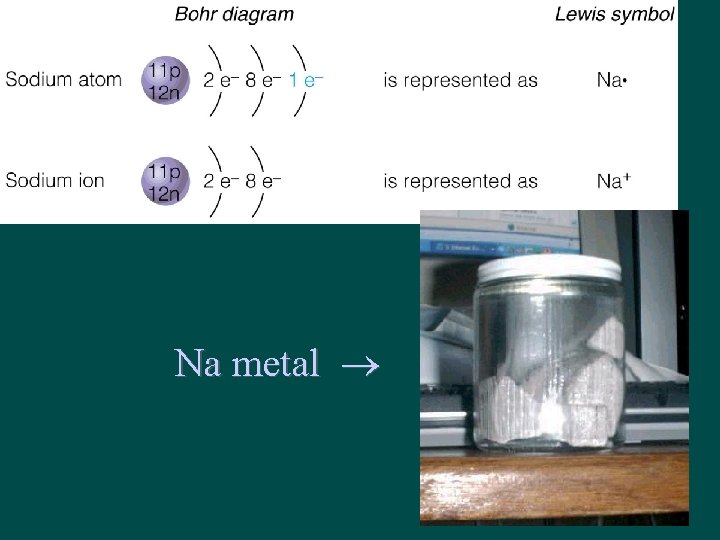
Na metal

Nonmetals are winners! • Cl has configuration 2 -8 -7 • Cl gains 1 electron → Cl-1 – Cl-1 configuration 2 -8 -8 (Ar) • What can donate an electron to Cl ? ?
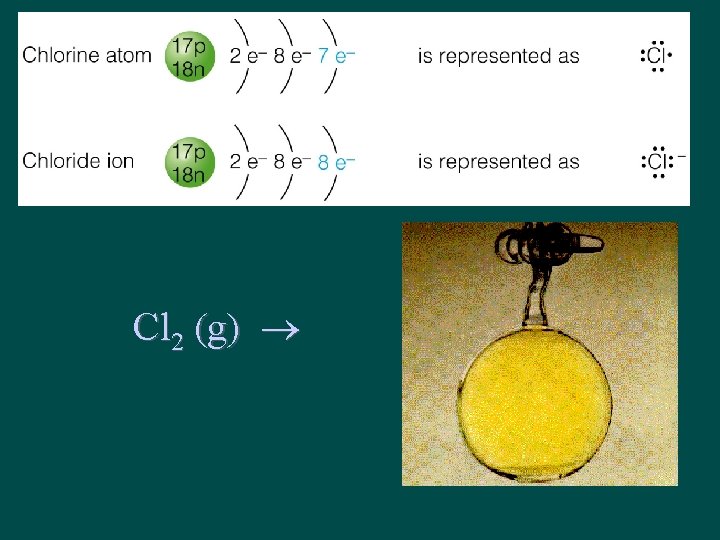
Cl 2 (g)
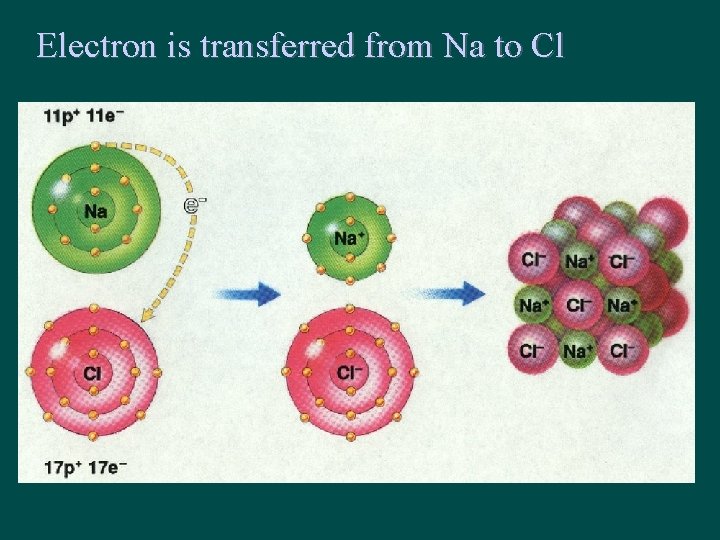
Electron is transferred from Na to Cl

Structure of Ionic Compounds • Ions are attracted to each other by strong electrostatic interactions (between +/- ions) • Ions form crystal lattice – regular 3 -D pattern or array – Ions are held in fixed positions (solid state)

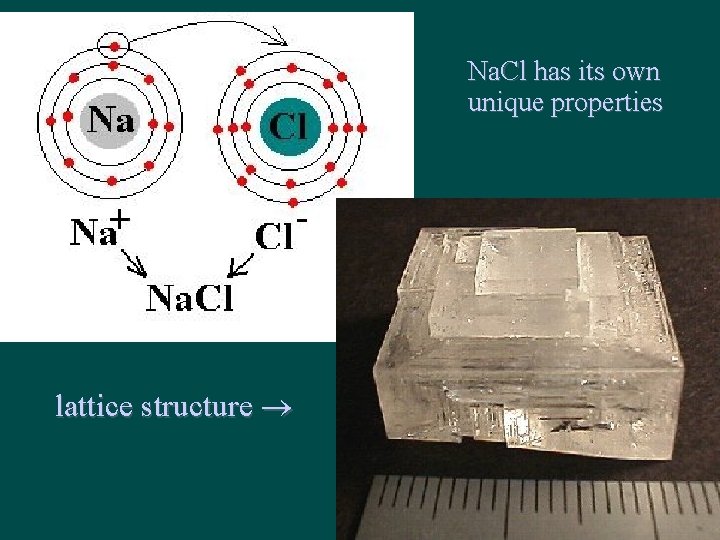
Na. Cl has its own unique properties lattice structure
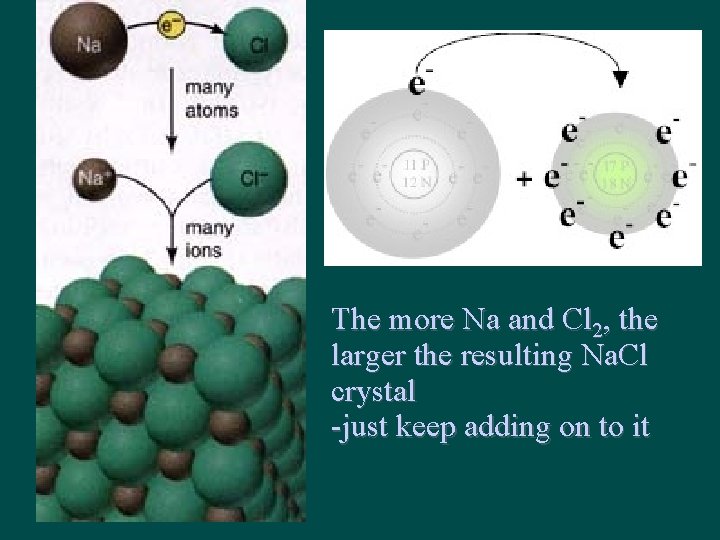
The more Na and Cl 2, the larger the resulting Na. Cl crystal -just keep adding on to it
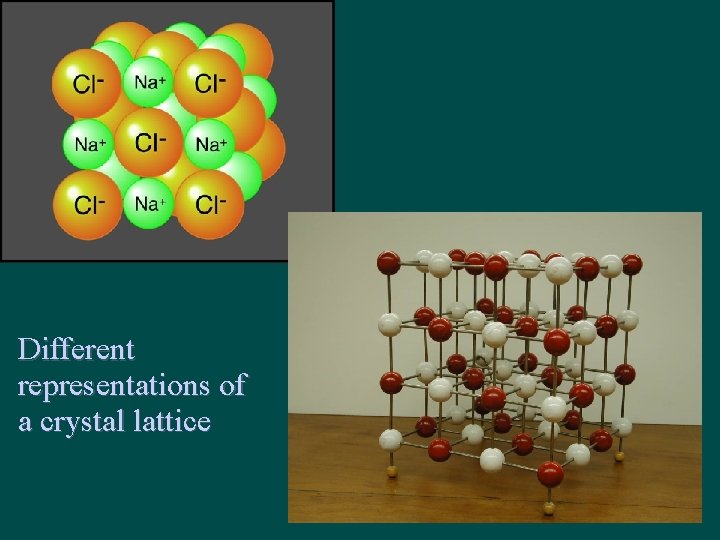
Different representations of a crystal lattice

Lewis Diagrams for Ionic Compounds Na. Cl's Lewis structure: • • • • -1 +1 [Na] [ • Cl • ] • • Remember: Lewis diagrams show valence electrons!

Predicting Ionic Compounds • What is ionic compound formed from calcium and iodine? • Calcium - metal - 2 valence electrons - loses both electrons [Ca]+2 • Iodine - nonmetal - 7 valence electrons • • -1 - gains 1 electron [ • • I • • ] • • How many iodine ions do you need to make a neutral compound? 2

Lewis Diagram of Calcium iodide Ca 2 -8 -8 -2 → Ca+2 2 -8 -8 I 2 -8 -18 -18 -7 → I-1 2 -8 -18 -18 -8 ·· -1 +2 [Ca] [: I: ] ·· -1 +2 or [: I: ] [Ca] [: I: ] · ·· · Note: total charge adds up to zero - compounds are neutral
- Slides: 16