IONIC BOND Dr Vikas A Thakur Department Of

IONIC BOND Dr. Vikas A. Thakur, Department Of Chemistry Rayat Shikshan Sanstha’s M. P. A. S. C. College, Panvel For S. Y. B. Sc. 1

INTRODUCTION � Noble gases have most stable outer electronic configuration of ns 2 and np 6. � The atoms of the other elements either lose , gain or share electrons to attain stable gas configuration. � In a molecule, atoms are held together by binding forces called as chemical bonds. � Hence chemical bonds are the forces which holds the atoms together in a molecule. � This phenomenon of combination of atoms to form molecules is called chemical bonding. � Valence electrons are the electrons in the highest occupied energy level of the atom. � Valence electrons are the only electrons generally involved in bond formation � The two principal types of chemical bonds are: � 1. Ionic or electrovalent bond. � 2. Covalent bond. 2

Ionic Bond � The strongly electro-positive and strong electro-negative elements combine and form compounds involving ionic bonds. � Strong electropositive elements lose electrons which are accepted by strong electronegative elements. � Due to this transfer of electrons from the electropositive elements to the electro-negative elements, the former attains positive charge and becomes a cation. � While latter attains negative charge to form anion. � Positive ions and negative ions are attracted by coulombic forces of attraction � As ions are attracted to one another, the bond between them is known as ionic bond. � The electrostatic forces of attraction between oppositely charged ions are called as ionic bonds. 3

SCHEMATIC REPRESENTATION OF IONIC BONDS �When sodium and Chlorine react together, the 4 outer electron of sodium is transferred to chlorine atom to produce sodium ion, Na+ and Chloride ion, Cl-. �The oppositely charged ions are then held together by electrostatic force of attraction i, e. By an ionic bond. �The formation of ionic bond can be shown diagrammatically by showing only outer electrons. �Similarly the outer two electrons of magnesium ions are transferred to chlorine atoms one each forming magnesium ions and chloride ion. �Formation of Mg. Cl 2 can be represented as.
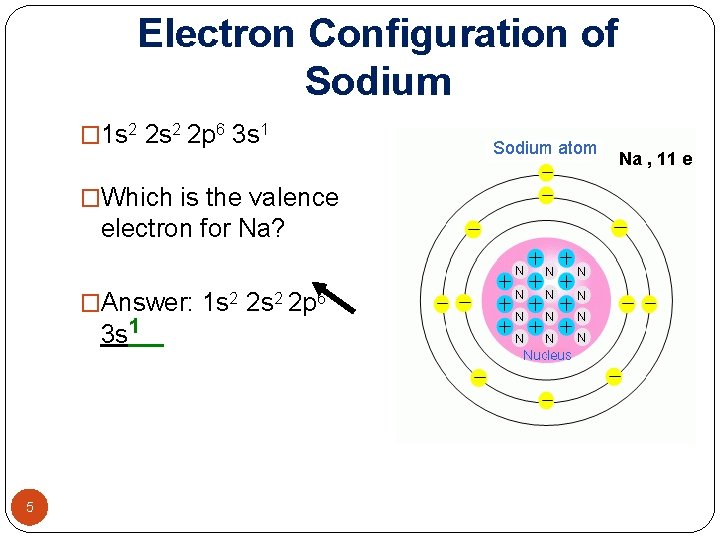
Electron Configuration of Sodium � 1 s 2 2 p 6 3 s 1 Na , 11 e �Which is the valence electron for Na? �Answer: 1 s 2 2 p 6 3 s 1 5
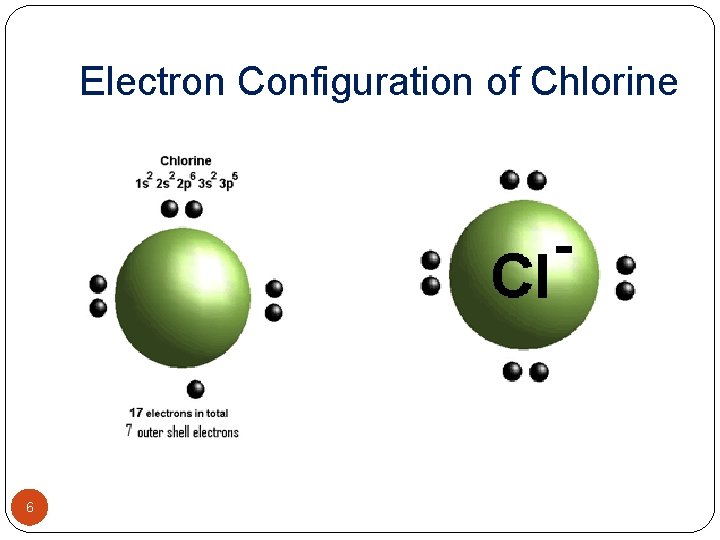
Electron Configuration of Chlorine 6
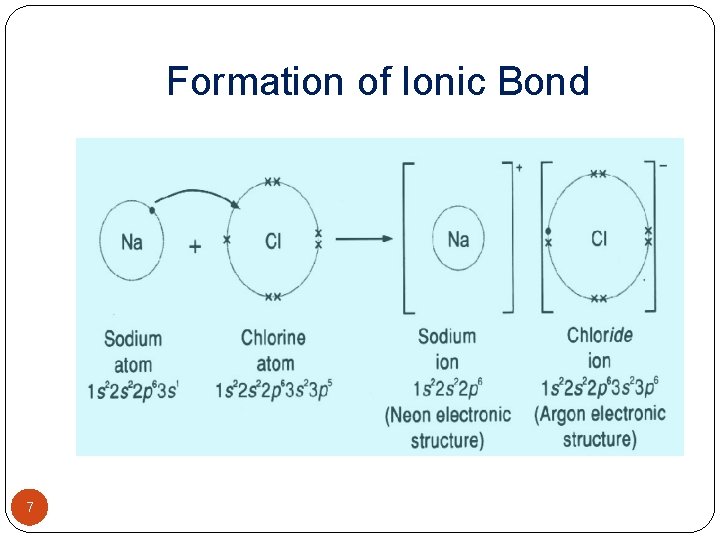
Formation of Ionic Bond 7

SCHEMATIC REPRESENTATION OF IONIC BONDS �Thus, in simple words, Electrovalent bond involves transfer of certain number of electrons from electro-positive element to electronegative element so that both acquire stable inert gas configuration. �The number of electrons lost or gained by element during the formation of an electrovalent bond is termed as valency number. �E, g. Na has +1, Ca has +2 and Aluminium has +3 while Cl has -1, Sulphur has -2 while N has -3 valency numbers. 8

Formation of Ionic Bond ØNeutral atoms come near each other. Electron(s) are transferred from the Metal atom to the Nonmetal atom. They stick together because of electrostatic forces, like magnets ØThe formation of Ionic compounds depends upon the ease of formation of cation and anion and forces of attraction between oppositely charged ions. Ø Factors playing a very important role in the formation of ionic compound. Ø 1. Ionization Energy: Lower the value of ionization energy of an 9 atom, greater will be ease of

Formation of Ionic Bond � 2. Electron Affinity : Higher the electron affinity of an 10 atom greater will be ease of formation of the anion. � 3. Electrostatic force of attraction : The forces of attraction between two oppositely charged ions can be determined by assuming the two ions as hard spheres. � If two spheres are imagined with charges +e 1 and –e 2 at a distant ‘r’ apart, then attractive force is given by Coulomb’s Law as � F = e 1 x e 2 /r 2 § The energy changes in the formation of an ionic compounds can be explained in a simplified way by neglecting the treatment of repulsive forces as the electron shells of two ions begin to overlap. § As ions are brought closer, first the attractive forces dominate over the repulsive forces. § It results into ions taking up an equilibrium position with a finite and measurable internuclear distance.
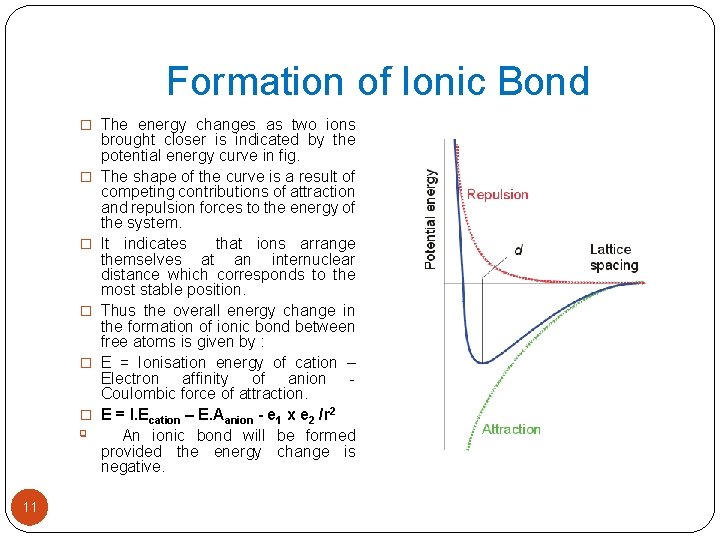
Formation of Ionic Bond � The energy changes as two ions � � � q 11 brought closer is indicated by the potential energy curve in fig. The shape of the curve is a result of competing contributions of attraction and repulsion forces to the energy of the system. It indicates that ions arrange themselves at an internuclear distance which corresponds to the most stable position. Thus the overall energy change in the formation of ionic bond between free atoms is given by : E = Ionisation energy of cation – Electron affinity of anion Coulombic force of attraction. E = I. Ecation – E. Aanion - e 1 x e 2 /r 2 An ionic bond will be formed provided the energy change is negative.
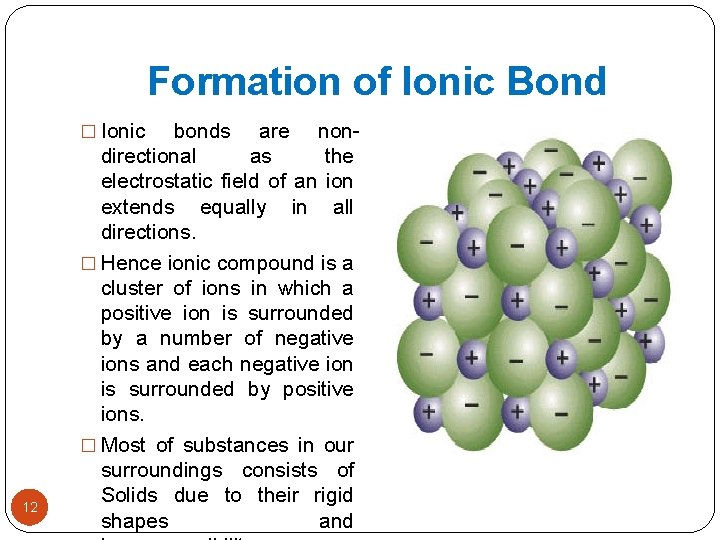
Formation of Ionic Bond � Ionic 12 bonds are nondirectional as the electrostatic field of an ion extends equally in all directions. � Hence ionic compound is a cluster of ions in which a positive ion is surrounded by a number of negative ions and each negative ion is surrounded by positive ions. � Most of substances in our surroundings consists of Solids due to their rigid shapes and

IONIC BONDING o An amorphous substance has no well-defined arrangements of its constituent units of atoms, molecules or ions. e, g. Glasss. o A Crystalline substance is made up of one or more crystals each having well-defined ordered arrangement of constituent units. e, g. Sodium chloride and sugar. o Depending upon nature of overall cohesive force existing between constituent particles, crystalline solids have classified into five categories. q 1. Ionic solids. q 2. Covalent crystals. q 3. Molecular crystals(vander waals crystals). q 4. Metallic crystals. q 5. Hydrogen bonded crystals. 13

Ionic Crystals 14

Crystals are everywhere! 15
More crystals 16

Early ideas � Crystals are solid - but solids are not necessarily crystalline � Crystals have symmetry (Kepler) and long range ? order � Spheres and small shapes can be packed to produces regular shapes (Hooke, Hauy) 17

Ionic Crystals � An ionic crystal is formed when cations and anions are brought together forming a three-dimensional array. � The ions arrange themselves in a definite geometrical manner known as crystal lattice. � The cohesive energy of ionic crystals is commonly known as lattice energy or lattice enthalpy. � It is derived from electrostatic interaction(both attraction and repulsion) among ions in crystal lattice. � The packing of oppositely charged ions into stable structure depends on: Ø 1. Stoichiometry of the compound. Ø 2. Electrical neutrality of the structure. Ø 3. Balance of electrostatic attraction and repulsion(coulombic) among ions. Ø 4. Pauli repulsion(non-coulombic) between the electrons. 18

Important Terms: Crystal lattice or Space lattice � Lattice means network. � Crystal lattice is an orderly array of repeating unit. � The three dimensional arrangement of particles 19 constituting crystal and represented by points is called crystal lattice or space lattice. � A space lattice is an array of points. � It shows how particles(atoms, ions or molecules) are arranged at different sites in three dimensional space so that the nature of spatial arrangement is clear. � Points signify the position of centre of particles and not the size. � Lines are mercely drawn to make nature of spatial arrangement clearer to reader. � Points constitute crystal lattice and not lines. � In other words Space lattice is a regular distribution of points in space in such a manner that every point has identical surroundings.

Lattice Points �The points at which the particles are arranged are 20 called lattice points. Or �The positions occupied by particles in the crystal lattice are called lattice points. �Lattice Constants : �The lattice is made up of repetitions of unit cells. �The unit cell is completely desribed by three vectors a, b and c which give length of the edges of unit cell and the angles between them is (alpha), ß(beta) and £(gama). �These lattice vectors and interfacial angles collectively are known as unit cell parameters or lattice constants.

3. The unit cell � Unit � � 21 cell is the smallest geometrical portion of the crystal which can be used to build up the whole crystal. The smallest repeating unit of a crystal structure, in 3 D, which shows the full symmetry of the structure is called Unit cell. It is smallest repeating unit of space latice which repeated over and over again results in crystal. In most of crystals unit cells are parallelopiped or cubes having three sets of parallel faces. The unit cell is a box with: • 3 sides - a, b, c • 3 angles - , ,

Lattice Constants � A crystal has same type of symmetry as unit cell have. � Crystals are bound by plane surfaces called faces. � Faces meet to form straight edges which intersect to form an angle. � Plane faces, straight edges and angles are characteristics of crystals. � The definite orderly arrangement of faces and edges of crystal are known as crystal symmetry. � From geometrical considerations, A Bravais showed that there only be fourteen different ways in which similar points can be arranged in three dimensional space. � Thus based on symmetry, crystals are grouped into 14 types of space lattice. 22
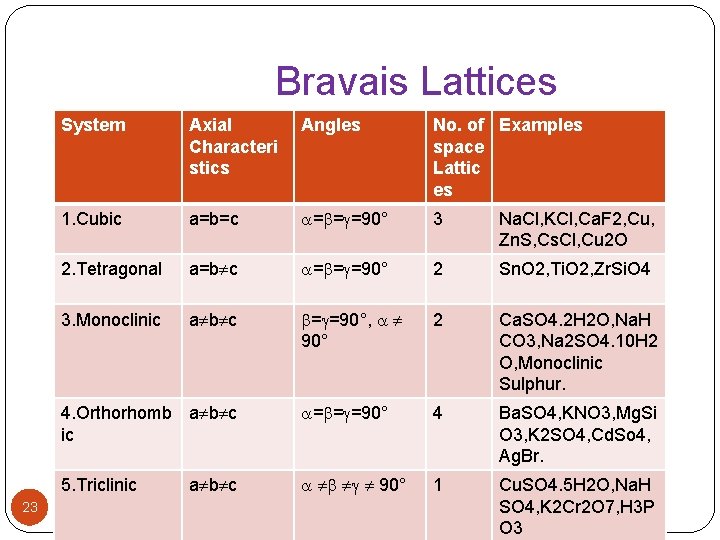
Bravais Lattices 23 System Axial Characteri stics Angles No. of Examples space Lattic es 1. Cubic a=b=c = = =90° 3 Na. Cl, KCl, Ca. F 2, Cu, Zn. S, Cs. Cl, Cu 2 O 2. Tetragonal a=b c = = =90° 2 Sn. O 2, Ti. O 2, Zr. Si. O 4 3. Monoclinic a b c = =90°, 90° 2 Ca. SO 4. 2 H 2 O, Na. H CO 3, Na 2 SO 4. 10 H 2 O, Monoclinic Sulphur. 4. Orthorhomb a b c ic = = =90° 4 Ba. SO 4, KNO 3, Mg. Si O 3, K 2 SO 4, Cd. So 4, Ag. Br. 5. Triclinic 90° 1 Cu. SO 4. 5 H 2 O, Na. H SO 4, K 2 Cr 2 O 7, H 3 P O 3 a b c
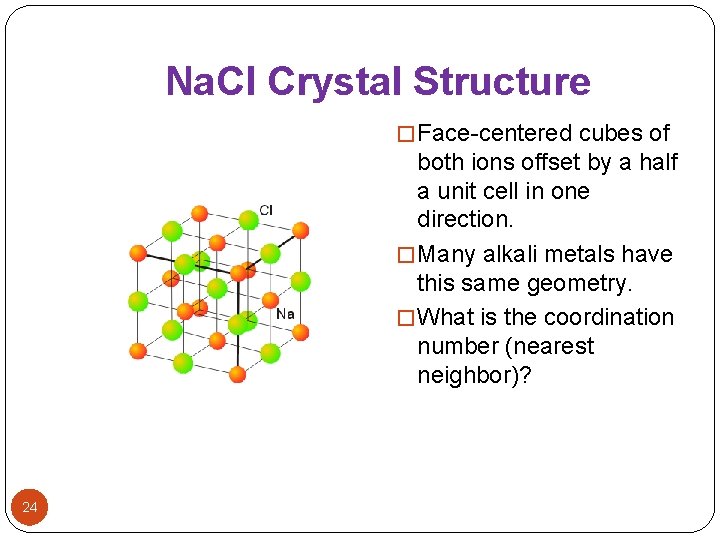
Na. Cl Crystal Structure � Face-centered cubes of both ions offset by a half a unit cell in one direction. � Many alkali metals have this same geometry. � What is the coordination number (nearest neighbor)? 24

Closest packing of rigid spheres �The external appearance of crystal may reflect the 25 internal structure which depends on the arrangement of particles. �The internal structures of solids can be understood by assuming atoms as hard spheres of equal dimension and arranging them in closest packing. �Closest packing is the way of arranging equidimensional spheres in space so that available space is filled upefeectively or more efficiently. �E, g. A bunch of grapes, a honeycomb, a raft soap bubbles, a pile of oranges.

�In three dimensions, there are three different ways in which hard spheres can be arranged. � 1. Hexagonal close packing(hcp). � 2. Cubic close packing(ccp) � 3. Body centered cubic close packing(bcc). �The spheres are arranged in such a way that a sphere is in contact with maximum number of nearest neighbours. �The number of nearest neighbours for an atom or ion is called as coordination number. 26
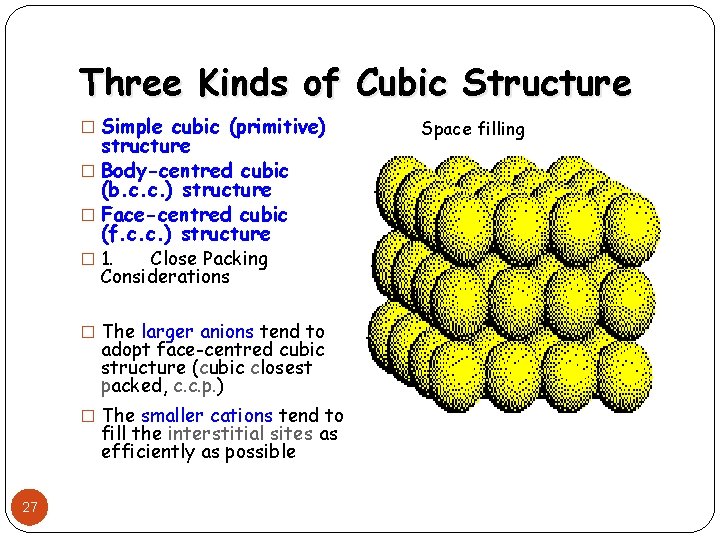
Three Kinds of Cubic Structure � Simple cubic (primitive) structure � Body-centred cubic (b. c. c. ) structure � Face-centred cubic (f. c. c. ) structure � 1. Close Packing Considerations � The larger anions tend to adopt face-centred cubic structure (cubic closest packed, c. c. p. ) � The smaller cations tend to fill the interstitial sites as efficiently as possible 27 Space filling
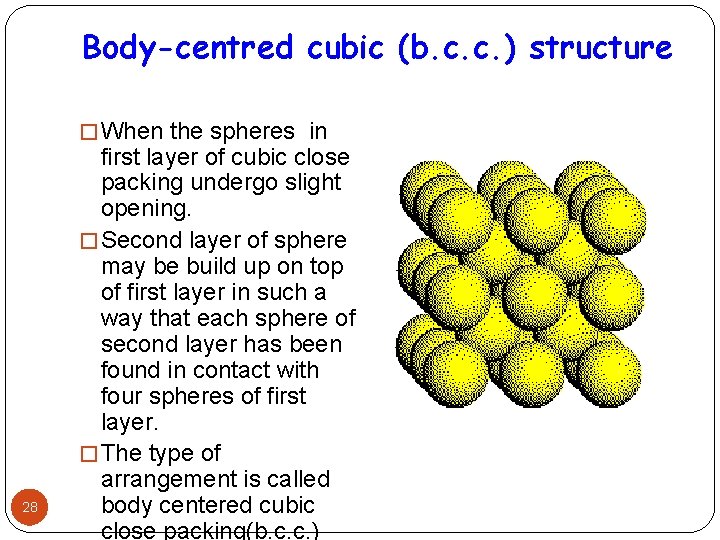
Body-centred cubic (b. c. c. ) structure � When the spheres in 28 first layer of cubic close packing undergo slight opening. � Second layer of sphere may be build up on top of first layer in such a way that each sphere of second layer has been found in contact with four spheres of first layer. � The type of arrangement is called body centered cubic close packing(b. c. c. )
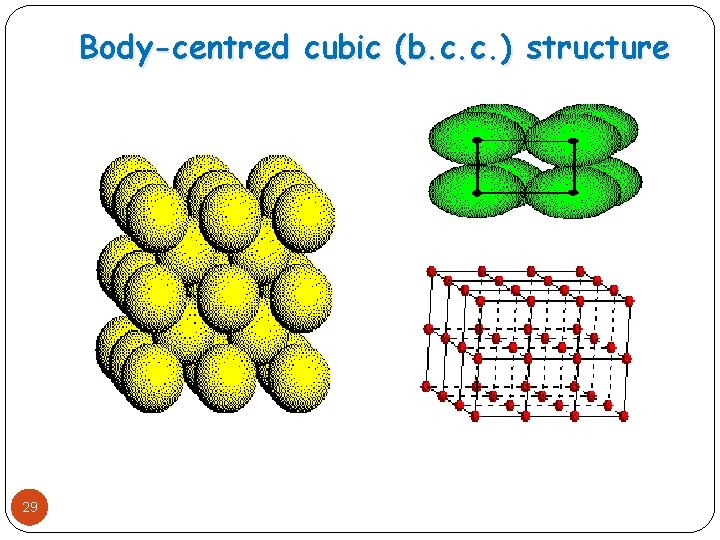
Body-centred cubic (b. c. c. ) structure 29
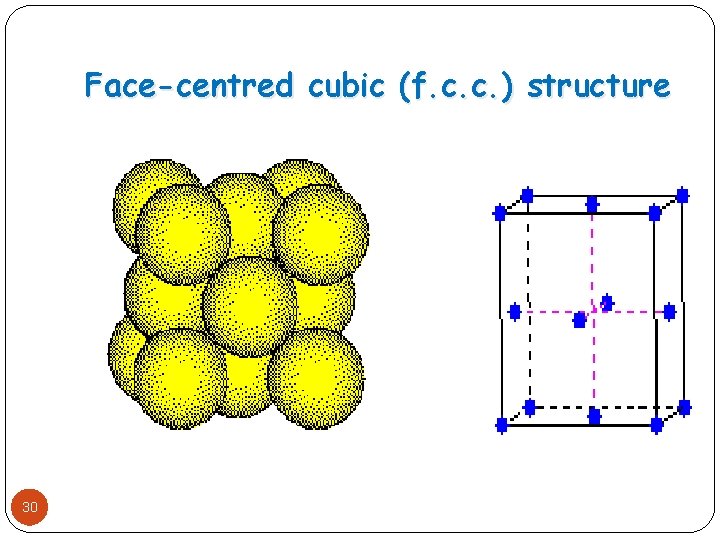
Face-centred cubic (f. c. c. ) structure 30
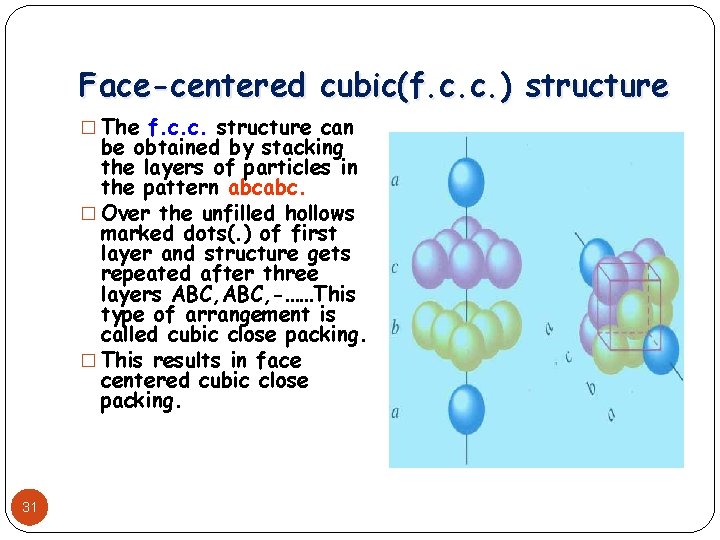
Face-centered cubic(f. c. c. ) structure � The f. c. c. structure can be obtained by stacking the layers of particles in the pattern abcabc. � Over the unfilled hollows marked dots(. ) of first layer and structure gets repeated after three layers ABC, -……This type of arrangement is called cubic close packing. � This results in face centered cubic close packing. 31
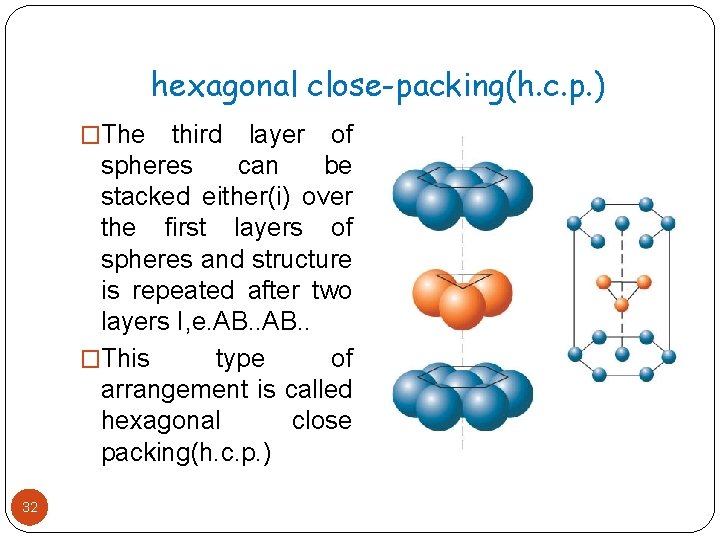
hexagonal close-packing(h. c. p. ) �The third layer of spheres can be stacked either(i) over the first layers of spheres and structure is repeated after two layers I, e. AB. . �This type of arrangement is called hexagonal close packing(h. c. p. ) 32

Voids in Ionic Crystals � Depending on the number of 33 anions and the manner in which they surround cation there are four types of voids. � 1. Trigonal or triangular void(C. N. of cation 3). � Tetrahedral void(C. N. of cation 4). � Octahedral void(C. N. of cation 6). � Cubic void(C. N. of cation 8). � 1. Trigonal or triangular void: � When three speres which represents anions are placed at the corners of equilateral triangle, vacant space left between these spheres(anions) is called trigonal void. � Tetrahedral Voids : � In this three spheres of first layer are represented by dark circles and spheres of second layer represented by broken lines. � The centres of these four spheres lie at the corners of a regular tetrahedron and the small space enclosed by the spheres represents a tetrahedral void or hole or site. � Thus tetrahedral voids results when a triangular void in a closest packed layer has a sphere directly over is as shown in fig.

Tetrahedral Voids � It is the space between the 3 spheres of one layer and a fourth sphere on the upper layer. � In an f. c. c. unit cell, the tetrahedral site is the space bounded by a corner atom and the three facecentred atoms nearest to it. 34

Tetrahedral Voids 35

Octahedral voids � An octahedral hole results when a triangular void of 36 one closest packed layer pointing up is covered with a triangular void pointing down of an adjacent layer. � An ion in octahedral hole is surrounded by six close packed oppositely charged ions situated at corners of regular octahedron. � The centres of spheres of first layer and second layer found at corners of two parallel equilateral triangles with their apices pointing in opposite directions which are also corners of octahedron. q Octahedral hole is larger than tetrahedral hole. q There are double number of tetrahedral holes than that of octahedral holes in closest packed arrangement.

Octahedral voids �It is the space between the 3 spheres in one layer and 3 other spheres in the adjacent layer. 37
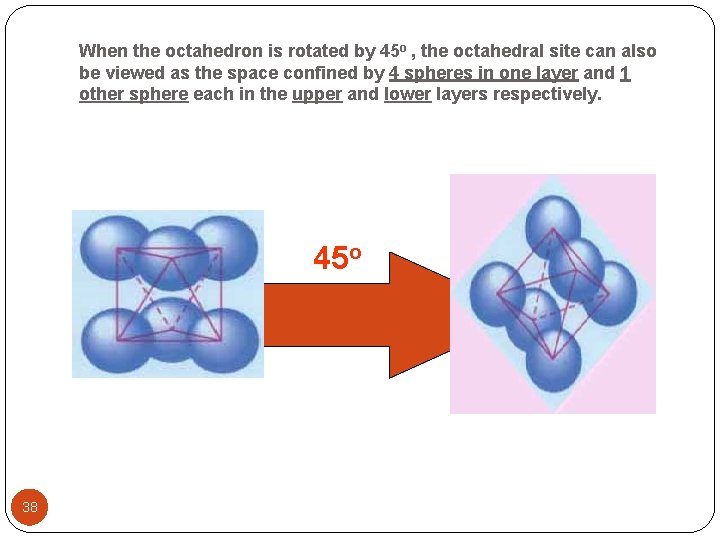
When the octahedron is rotated by 45 o , the octahedral site can also be viewed as the space confined by 4 spheres in one layer and 1 other sphere each in the upper and lower layers respectively. 45 o 38

TYPES OF IONIC CRYSTALS �The ionic crystals are classified into five types based on number of cations and anions. Ø 1. AB Type: Sodium chloride(Na. Cl), Caesium Chloride(Cs. Cl), Zinc Sulphide, Zn. S). Ø 2. AB 2 Type : Calcium fluorite Ca. F 2, Titanium dioxide Ti. O 2(Rutile) Ø 3. A 2 B Type : Antifluorite structure(Na 2 O, Li 2 O) Ø 4. AB 3 Type : Cryolite(Na 3 Al. F 6) Ø 5. A 2 B 3 Type : Corundum( -Al 2 O 3 ) Ø Salts of AB type crystallises in any one of five basic types of crystal structure having 1: 1 Stoichiometry. 39
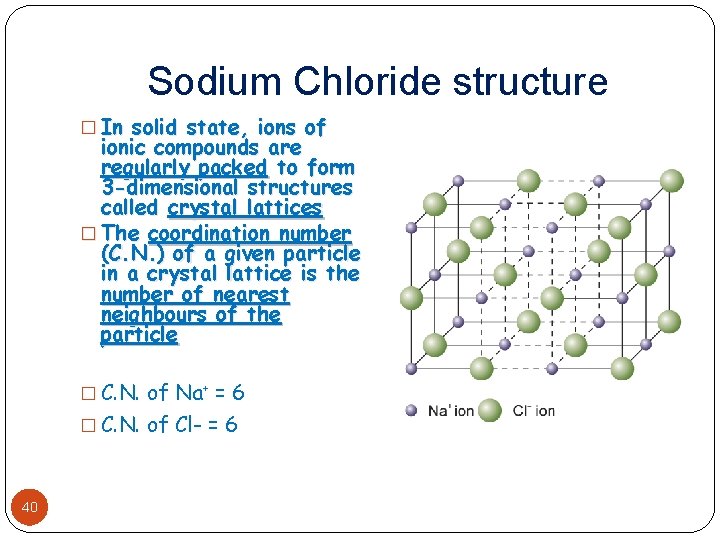
Sodium Chloride structure � In solid state, ions of ionic compounds are regularly packed to form 3 -dimensional structures called crystal lattices � The coordination number (C. N. ) of a given particle in a crystal lattice is the number of nearest neighbours of the particle � C. N. of Na+ = 6 � C. N. of Cl- = 6 40

Sodium Chloride structure � Radius ratio r+/r- lies between 0. 414 -0. 732 , C. N. 4 or 6. � Shape should be either square planar or octahedron. � X-ray study prove that structure is octhedron. � All octahedral sites are filled by Na+ ions. � Each chloride ion in lattice is surrounded by six sodium ions in octahedral holes. � Each sodium ion in octahedral hole is surrounded by six chloride ions. � Hence cation and anion have coordination number 6: 6. � Ions are at each corners of cube and at centre of cube. � The oppositely charged ions are at centre of edges and at centre of cube. � Nacl crystal lattice thought of being composed of two interlocking face centered cubic sublattices, one Na+ having origin at (0, 0, 0) and the other at midway (1/2, 0, 0). � Exam. Cs. F, Ag. Cl, Ag. Br, KCN etc. 41
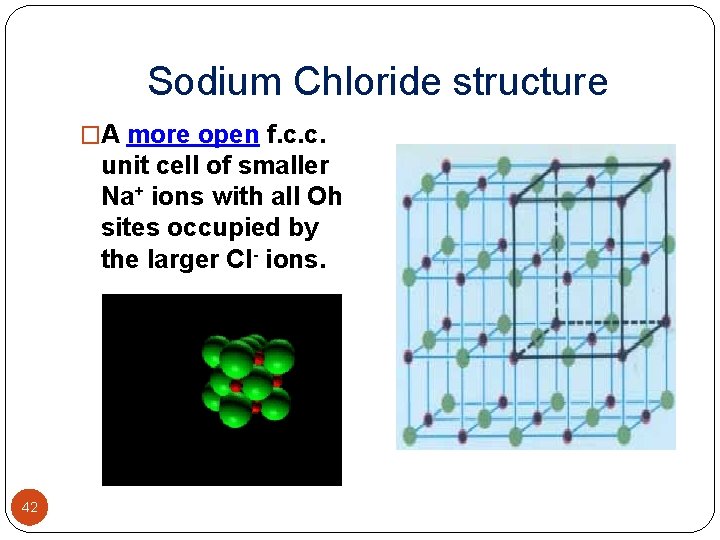
Sodium Chloride structure �A more open f. c. c. unit cell of smaller Na+ ions with all Oh sites occupied by the larger Cl- ions. 42

Caesium Chloride structure Co-ordination number of Cs+ = 8 43 Co-ordination number of Cl- = 8
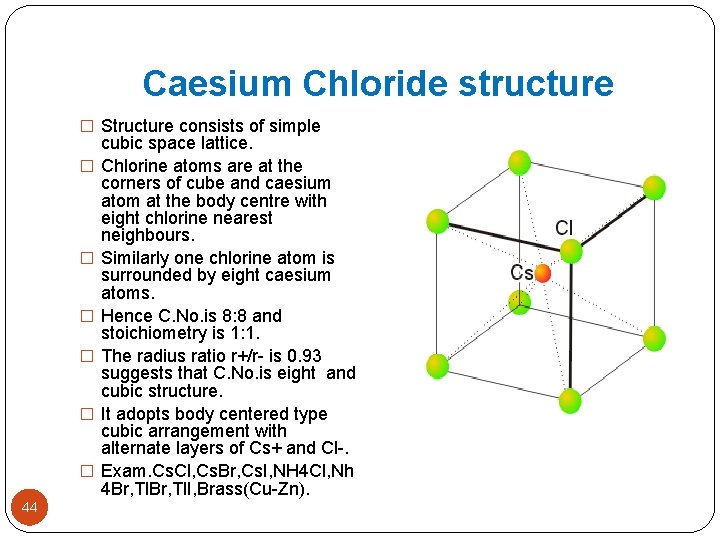
Caesium Chloride structure � Structure consists of simple � � � 44 cubic space lattice. Chlorine atoms are at the corners of cube and caesium atom at the body centre with eight chlorine nearest neighbours. Similarly one chlorine atom is surrounded by eight caesium atoms. Hence C. No. is 8: 8 and stoichiometry is 1: 1. The radius ratio r+/r- is 0. 93 suggests that C. No. is eight and cubic structure. It adopts body centered type cubic arrangement with alternate layers of Cs+ and Cl-. Exam. Cs. Cl, Cs. Br, Cs. I, NH 4 Cl, Nh 4 Br, Tl. I, Brass(Cu-Zn).
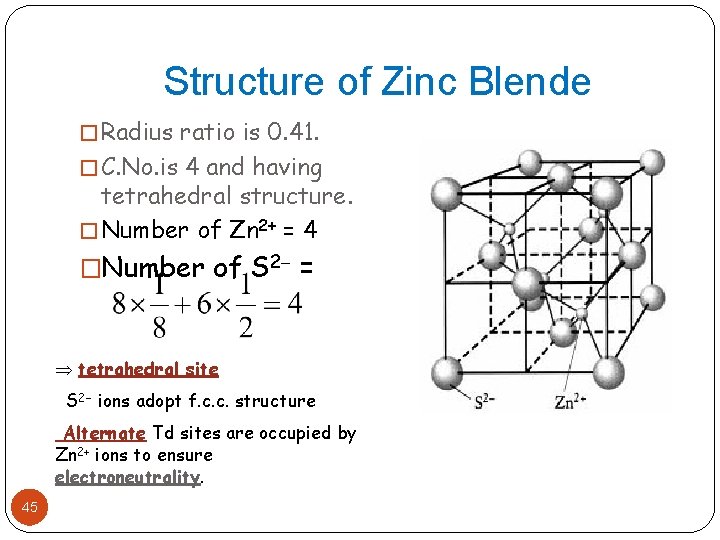
Structure of Zinc Blende � Radius ratio is 0. 41. � C. No. is 4 and having tetrahedral structure. � Number of Zn 2+ = 4 �Number of S 2 = tetrahedral site S 2 ions adopt f. c. c. structure Alternate Td sites are occupied by Zn 2+ ions to ensure electroneutrality. 45

Structure of Zinc Blende � In Zinc blende, the sulphur atoms form cubic close 46 packing(ccp) lattice and zinc atoms occupy alternate tetrahedral voids. � In fact, zinc atoms in a zinc blend separately forms a ccp lattice if we connect zinc atoms from adjoining unit cell. � Hence they are called as interpenetration of two ccp sublattices which are called as sublattices, one of zinc atoms and another of sulphur atoms. � Exa. Cu. Br, Cd. Se, Al. Sb etc. � Generally, when radius ratio between 0. 414 -0. 732, expected crystal structure is octahedral. � But Zn. S is not six coordinated but it is four coordinated. Hence radius ratio fails.
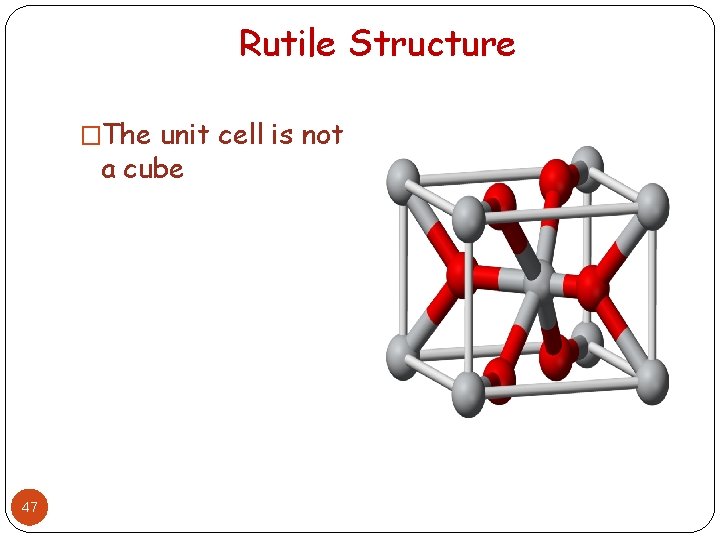
Rutile Structure �The unit cell is not a cube 47

Lattice Enthalpy Calculations �Considering only Coulombic contributions �The electrostatic potential energy between each pair. z. A, z. B = ionic charges in electron units r 0 = distance between ion centers e = electronic charge 4 o = permittivity of a vacuum e 2/ 4 o = 2. 307 10 -28 J m Calculation would be performed on each cation/anion pair (nearest neighbor). 48

Lattice Enthalpy Calculations �A more accurate equation depicts the Coulombic interactions over the entire crystal. NA = Avogadro’s constant A = Madelung’s constant, value specific to a crystal type (in table). This is a sum of all the geometric factors carried out until the interaction become infinitesimal. 49
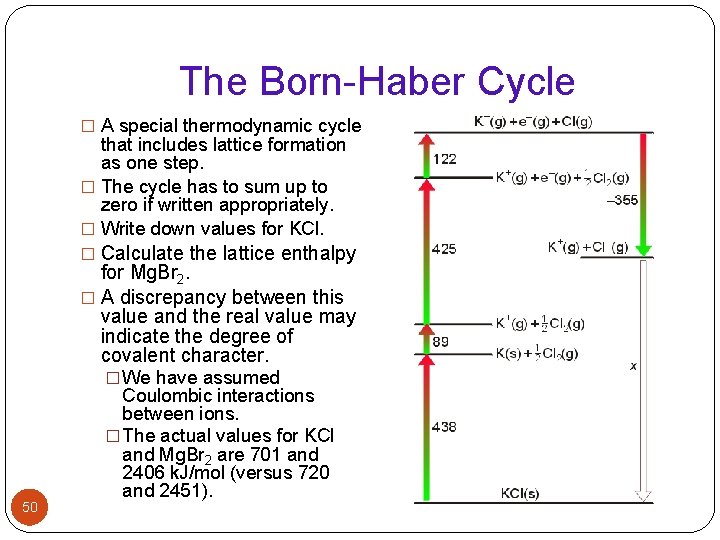
The Born-Haber Cycle � A special thermodynamic cycle that includes lattice formation as one step. � The cycle has to sum up to zero if written appropriately. � Write down values for KCl. � Calculate the lattice enthalpy for Mg. Br 2. � A discrepancy between this value and the real value may indicate the degree of covalent character. � We have assumed 50 Coulombic interactions between ions. � The actual values for KCl and Mg. Br 2 are 701 and 2406 k. J/mol (versus 720 and 2451).

References �College Inorganic Chemistry for S. Y. B. Sc. by Himalaya Publishing House, 23 rd Edition, (2015). �College Inorganic and Industrial Chemistry for S. Y. B. Sc. by Sheth Publishing House, (2015). �A Concise Inorganic Chemistry by J. D. Lee. �Inorganic Chemistry by Alan G. Sharpe, University of Cambridge. 3 rd edition(2013). 51

THANK YOU 52
- Slides: 52