Ionic and Covalent Bonds ALLISON AND ALLISON Ionic

Ionic and Covalent Bonds ALLISON AND ALLISON

Ionic Bonding Definition “Ionic bonding is the transfer of electrons from one atom to another, causing one atom to have a positive charge, and the other a negative charge. ” Basically, two atoms hook up and exchange electrons and one ion is positive and the other is negative. Metal loses electrons to become positively charged Nonmetal gets electrons to become negatively charged

Discovery of Ionic Bonding It was discovered by Joseph John Thomson. He went to college at 14 years old, and Thomson published a paper about his discovery of the electron. He also theorized the possible uses of them. That’s where he suggested that electrons can be transferred from one atom to another to form a type of magnetic bonds. Linus Pauling, however, is the one who defined covalent ionic bonds, after he discovered the difference between the two.

Covalent Bonding Definition A covalent bond involves the sharing of electron pairs between atoms. These pairs are known as bonding pairs, and the balance between attractive and repulsive forces between atoms when they have bonding pairs is covalent bonding. Basically, two atoms exchange an electron with each other. Always happens with nonmetals. A lot of covalent bonding involves hydrogen.

Covalent Bonding History Gilbert Lewis first described covalent bonding by using the Lewis diagrams (the dot model). The term covalence used for bonding was first used in 1919 by Irving Langmuir. He said that “we shall denote by the term covalence the number of pairs of electrons that a given atom shares with its neighbors. ” Lewis proposed that an atom forms enough covalent bonds to form a full outer electron shell/ full valence shell. Quantum mechanics is needed to fully understand covalent bonds. Walter Heitler and Fritz London have the first successful quantum mechanical explanation of a chemical bond in 1927.
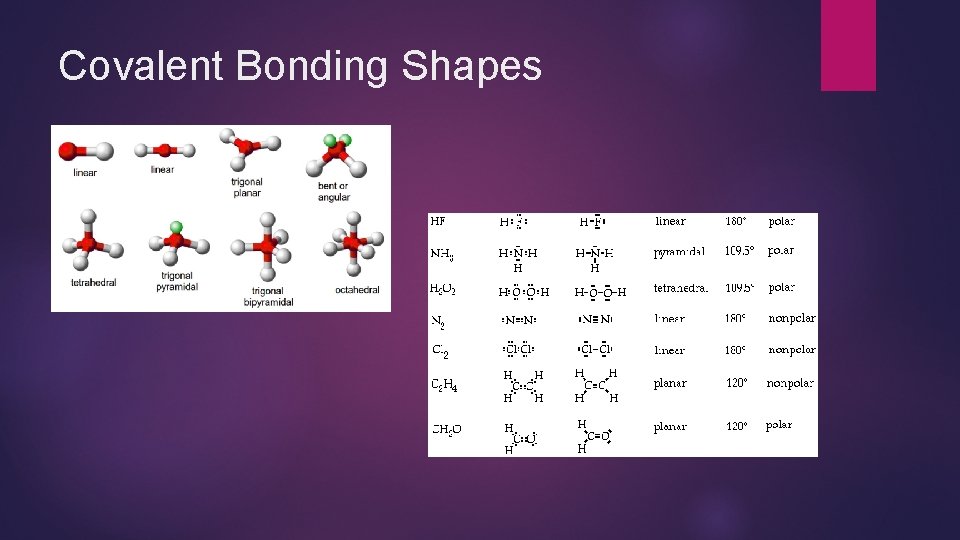
Covalent Bonding Shapes

More on Lewis Dot Structure Also known as Lewis Diagram, Dot Structure, Dot Thingy, No more than two on each side Can only have up to eight Can have multiple bonds on the same side Double bonds, Triple bonds, all covalent The dots are the valence electrons/ it shows the valence shell The one with the most valence electrons are generally in the center You generally want to include the charges of each atom

Important Vocab Ionic Bond – bond formed by attraction between ions Lone pairs – unshared pairs of electrons Covalent Bond - the additional force of attraction that results from valence electrons being shared Multiple Bonds - when two atoms share more than 1 pair of electrons with each other Lewis Dot Structure – used to show atoms valence electrons and bonding Chemical Bond - a strong attractive force Electron Pair - two valence electrons close to one another Unpaired Electrons - single valence electrons that are used for sharing with another atom in a covalent bond Polar Covalent Bond - a covalent bond in which the electrons are shared unequally Ionic Compound – Metal + Nonmetal Covalent Compound – Nonmetal + Nonmetal (or Metalloids) Alloys – Metal + Metals Bonding Pairs – shared pairs of electrons

Practice Questions!

Water, H 2 O, is a what type of bond? Covalent.

Write the Dot Structure for Lithium Chloride and identify its bond type. Ionic
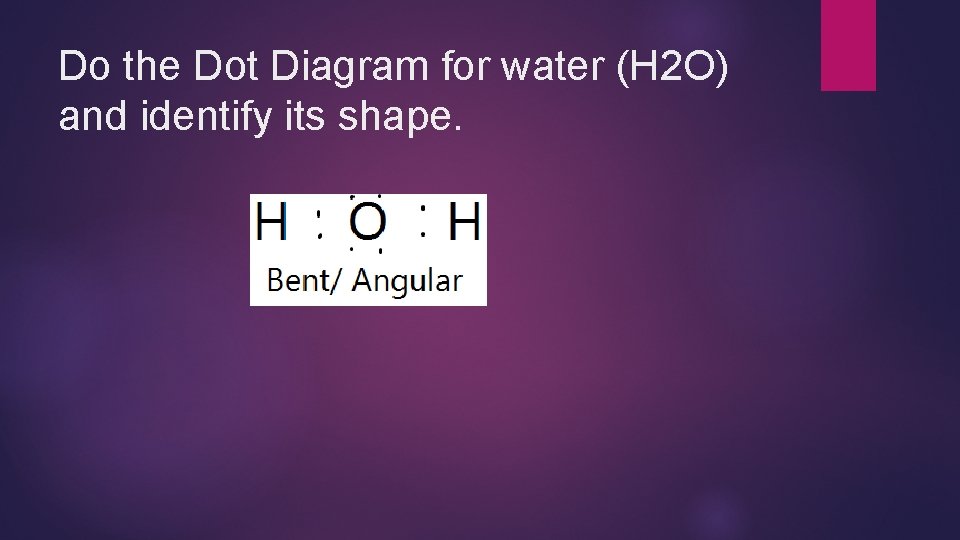
Do the Dot Diagram for water (H 2 O) and identify its shape.

Do the Dot Diagram for Ammonia (H 3 N) and identify its shape.

Do the Dot Diagram for Ammonia (H 3 N) and identify its shape.
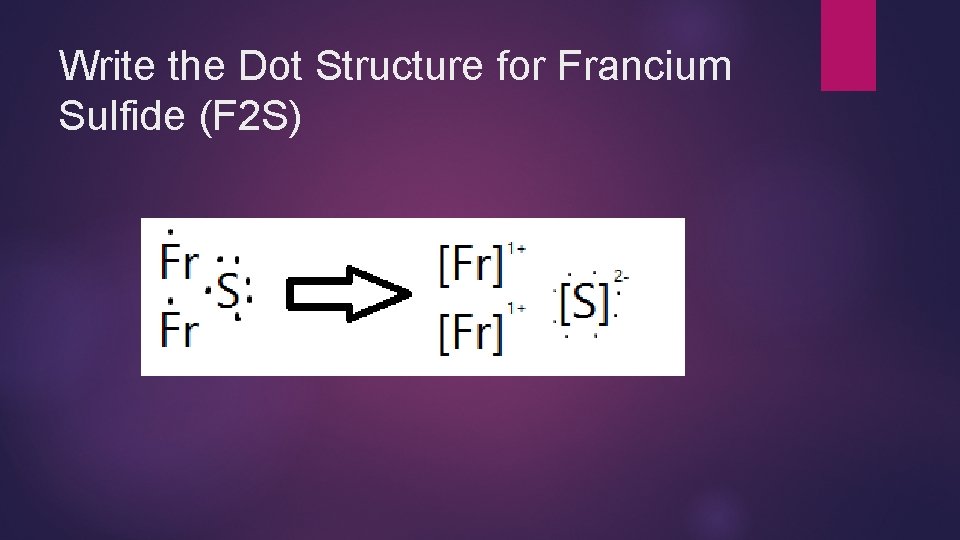
Write the Dot Structure for Francium Sulfide (F 2 S)
- Slides: 15