ION FORMATION group B elements Practice 1 Write

ION FORMATION group “B” elements Practice 1. Write the ion formed by these element. n a. iodine I-1 b. potassium c. sulfur S-2 d. cobalt (II) Co+2 K+1 e. gallium Ga+3 f. chromium (III) Cr+3

ION FORMATION group “B” elements Writing Ions Practice
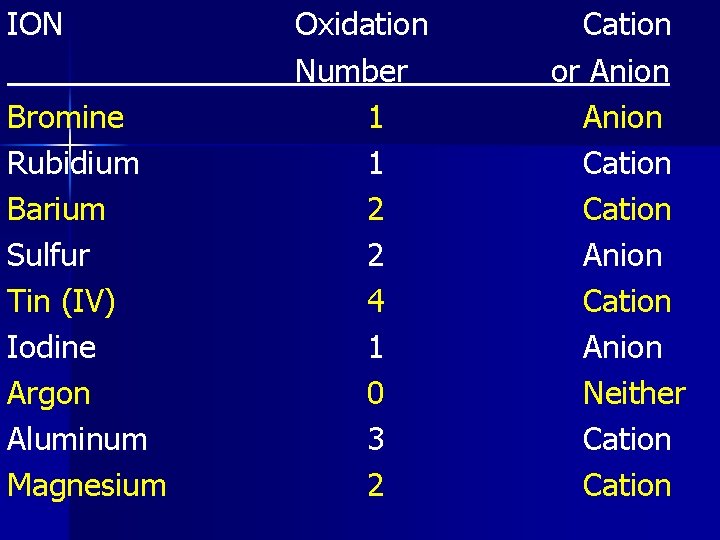
ION Bromine Rubidium Barium Sulfur Tin (IV) Iodine Argon Aluminum Magnesium Oxidation Number 1 1 2 2 4 1 0 3 2 Cation or Anion Cation Anion Neither Cation
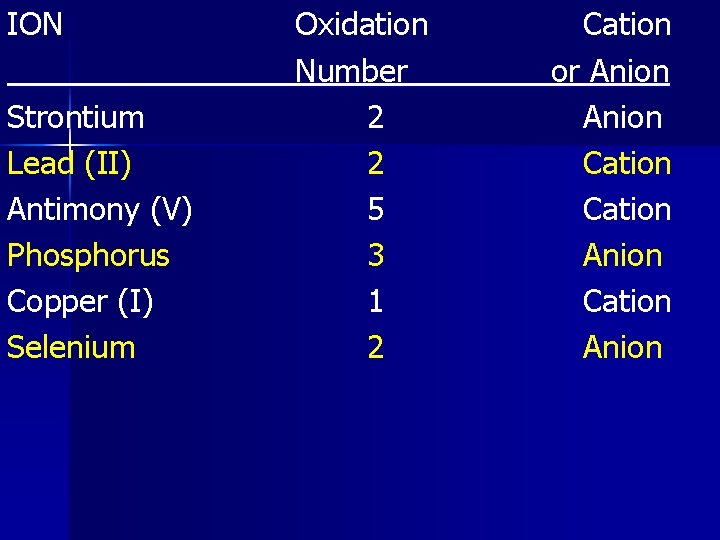
ION Strontium Lead (II) Antimony (V) Phosphorus Copper (I) Selenium Oxidation Number 2 2 5 3 1 2 Cation or Anion Cation Anion

Binary Compounds with Polyatomic Ions n Covalently-bonded groups of atoms that behave as a unit and carry a charge NO 3 -1
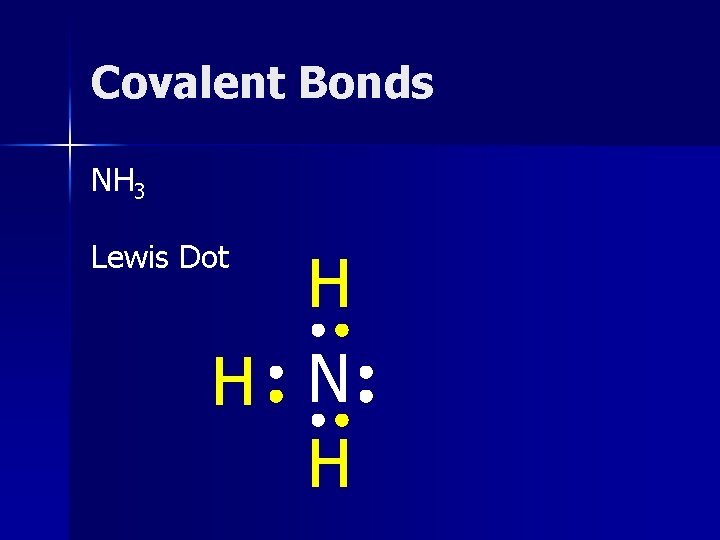
Covalent Bonds NH 3 Lewis Dot H H N H
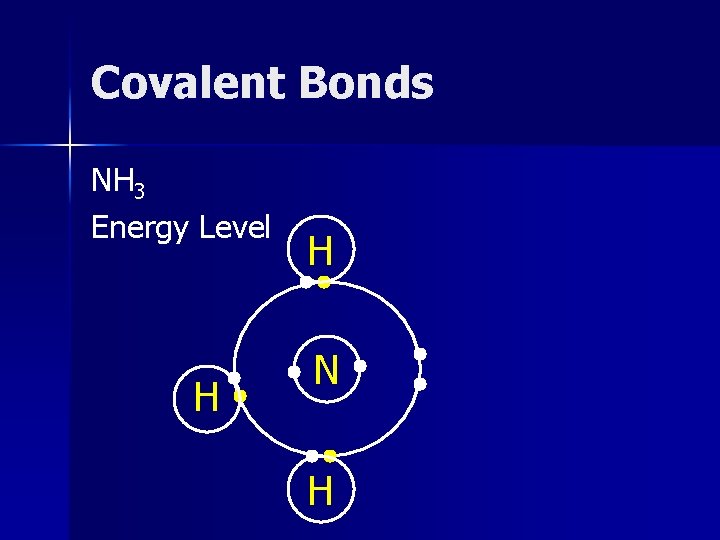
Covalent Bonds NH 3 Energy Level H H N H
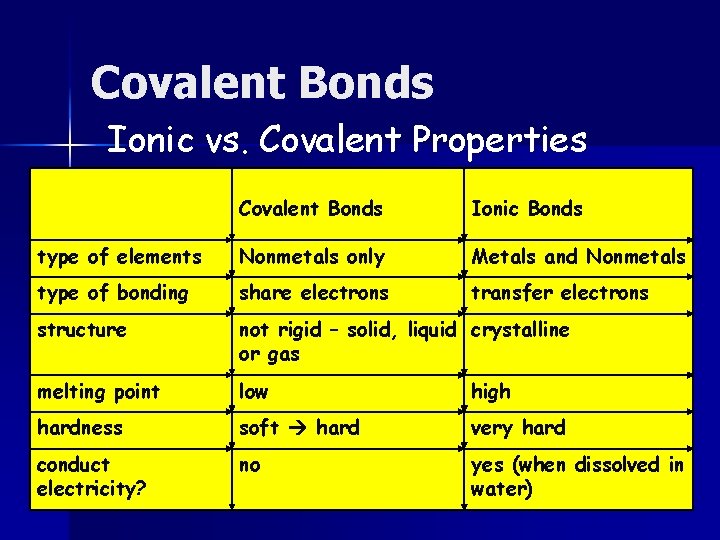
Covalent Bonds Ionic vs. Covalent Properties Covalent Bonds Ionic Bonds type of elements Nonmetals only Metals and Nonmetals type of bonding share electrons transfer electrons structure not rigid – solid, liquid crystalline or gas melting point low high hardness soft hard very hard conduct electricity? no yes (when dissolved in water)

Covalent Bonds: Diatomic Atoms There are 7 elements that can form covalent bonds between its atoms Hydrogen Atoms H· ·H Molecule H: H

Covalent Bonds: Diatomic Atoms Nitrogen Atoms · · : N· ·N: · · Molecule : N: : : N: Forms a triple bond
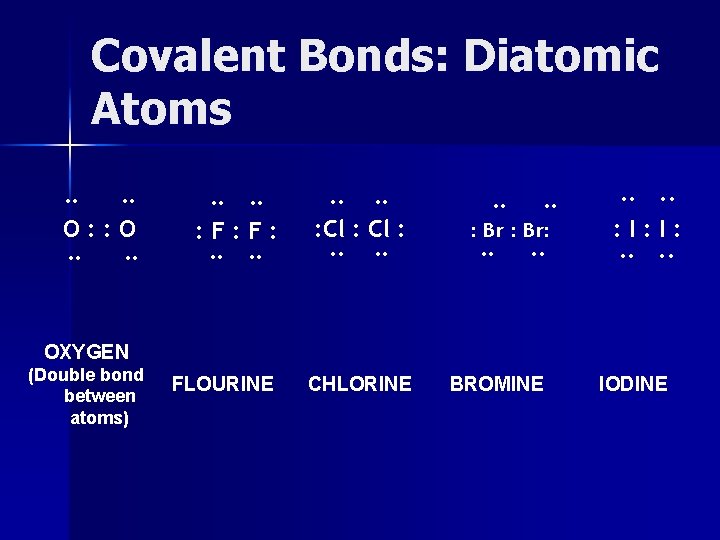
Covalent Bonds: Diatomic Atoms ·· ·· O: : O ·· ·· : F: F: ·· ·· : Cl : ·· ·· : Br: ·· ·· : I: I: ·· ·· OXYGEN (Double bond between atoms) FLOURINE CHLORINE BROMINE IODINE

Determine ionic or covalent bonds N 2 O 5 Covalent (nonmetals) A liquid at toom temp Covalent (liquid) Pb. NO 3 Ionic (Metal with nonmetal)

Determine ionic or covalent bonds A salt that conducts electricity in solution Ionic (conducts electricity) KF Ionic (metal with nonmetal) Ag. Cl Ionic (Metal with nonmetal)

Determine ionic or covalent bonds Gasoline Covalent (liquid) PCl 3 Covalent (metal to metal) O 2 Covalent (share electrons)

Naming Covalent Compounds Oxidation numbers are not used in determining the formula unit Two nonmetals can combine in more than 1 way We use prefixes to name and indicate the number of atoms of each type
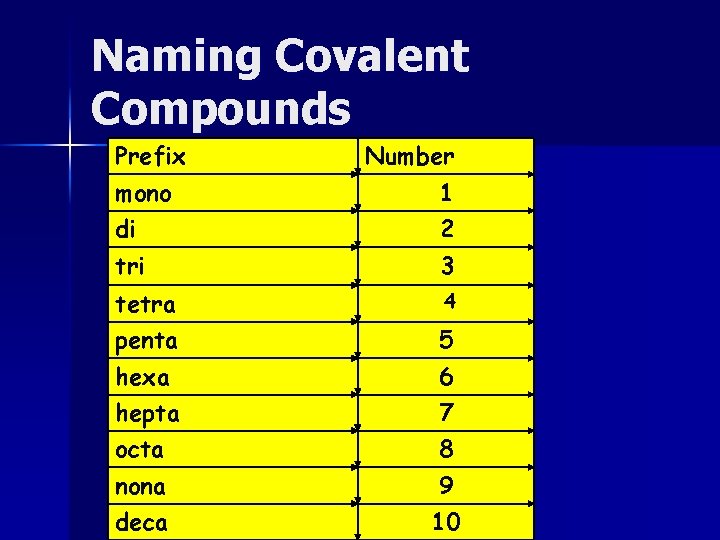
Naming Covalent Compounds Prefix Number mono 1 di 2 tri 3 tetra 4 penta 5 hexa 6 hepta 7 octa 8 nona 9 deca 10

Naming Covalent Compounds Write the name of the element that is farthest left first Write the other element second and change the ending to –ide Add prefixes to the two elements Do not use mono on first element

Naming Covalent Compounds CO 2 Element to left first, then the second element with an –ide ending Carbon Oxide Add prefixes Monocarbon Dioxide Drop Mono from first element Carbon dioxide

Naming Covalent Compounds CF 4 Element to left first, then the second element with an –ide ending Carbon Flouride Add prefixes Monocarbon Tetraflouride Drop Mono from first element Carbon Tetraflouride

Naming Covalent Compounds Si. Cl 3 Element to left first, then the second element with an –ide ending Silicone Chloride Add prefixes Monosilicone trichloride Drop Mono from first element Silicon Trichloride

Naming Covalent Compounds CO 2 Element to left first, then the second element with an –ide ending Carbon Oxide Add prefixes Monocarbon Dioxide Drop Mono from first element Carbon dioxide

Naming Covalent Compounds CO 2 Element to left first, then the second element with an –ide ending Carbon Oxide Add prefixes Monocarbon Dioxide Drop Mono from first element Carbon dioxide

Naming Covalent Compounds H 2 O Element to left first, then the second element with an –ide ending Hydrogen Oxide Add prefixes Dihydrogen oxide Drop Mono from first element Dihydrogen oxide

Naming Covalent Compounds Common names to remember H 2 O = Water NH 3 = Ammonia CH 4 = Methane

Writing Formulas for Binary Covalent Compounds List the symbol of the first element in the name first and then second element next Use the subscripts to identify the prefix in the name for that atom

Writing Formulas for Binary Covalent Compounds Carbon Tetrachloride C Cl Tetra= 4 CCl 4

Writing Formulas for Binary Covalent Compounds Dinitrogen Trioxide N Di = 2 N 2 O 3 O Tri = 3

Writing Formulas for Binary Covalent Compounds Iodine Pentaflouride I F Penta = 5 IF 5

Writing Formulas for Binary Covalent Compounds Hexaboron Silicide B Hexa = 6 B 6 Si Si

Writing Formulas for Binary Covalent Compounds Diantomny Pentoxide Sb Di = 2 Sb 2 O 5 O Penta = 5

Writing Formulas for Binary Covalent Compounds Ammonia NH 3

Polar Covalent Bonds A type of covalent bond where electrons are shared unequally between atoms Electrons spend more time with one atom than the other The stronger atom will be slightly negative and the other will be slightly positive

Polar covalent Bonds Water: the electrons hang around the oxygen atom more than the hydrogen atom Example of Polar Covalent Bond in Action
- Slides: 33