Ion Exchange Chromatography Introduction Adsorption chromatography Reversible electrostatic
Ion Exchange Chromatography

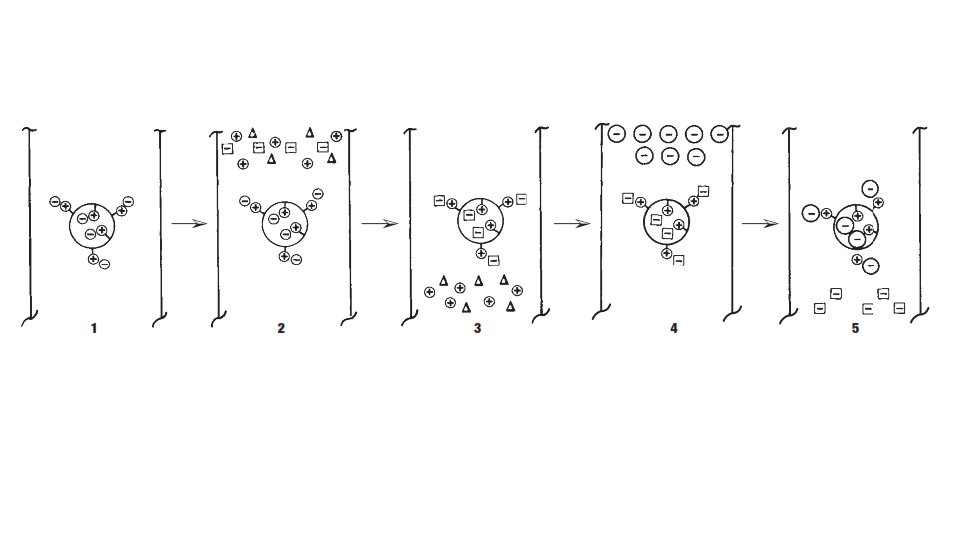
Introduction • Adsorption chromatography • Reversible electrostatic interactions • Ionic interaction

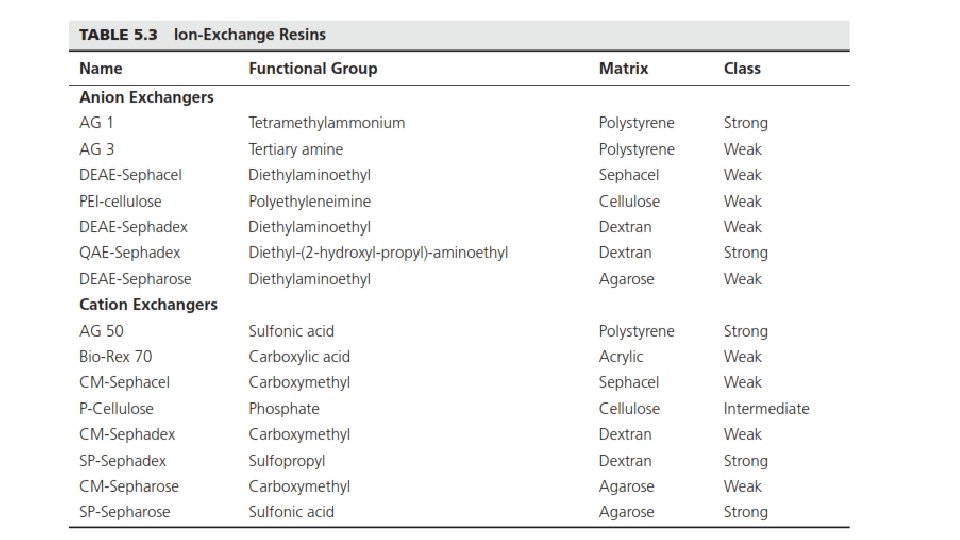
Ion-Exchange Resins • Polystyrene, acrylic resins, polysaccharides (dextrans), agarose, and celluloses • Cationic or anionic • Strong or weak • Strongly basic anion exchanger • Weakly basic anion exchanger • Strongly acidic cation exchanger
Selection of the Ion Exchanger • Nature of a molecules • Polystyrene – amino acids, lipids, nucleotides, carbohydrates, pigments, etc. • Cellulosic ion exchangers and low-percent cross-linked dextran or acrylic exchangers - peptides, proteins, nucleic acids, polysaccharides, and other large biomolecules • Charge based – p. H • p. H staility, isoelectric point
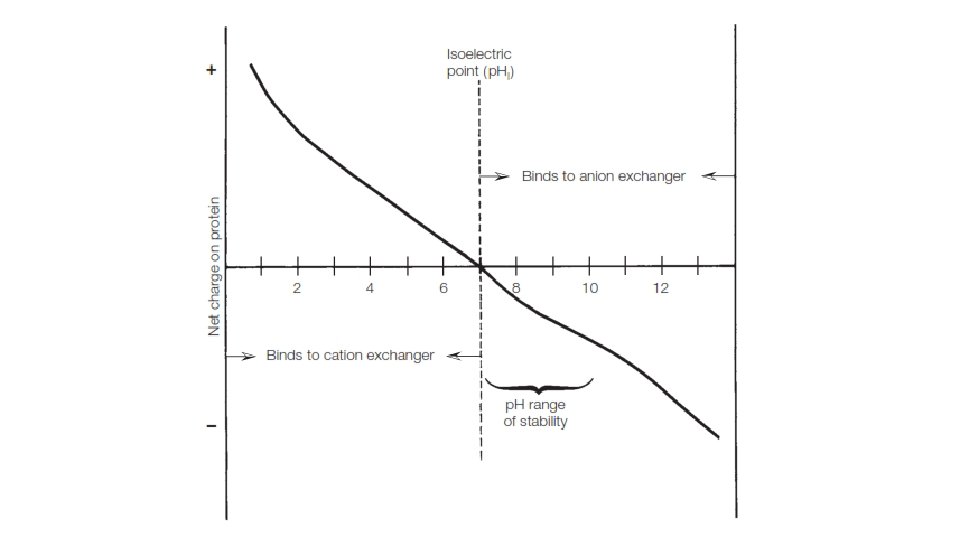

Choice of Buffer • p. H and ionic strength • 0. 05 to 0. 1 M are recommended

Preparation of the Ion Exchanger • Instructions • Pretreatment • Fines removal • 8 -10 times washings

Using the Ion-Exchange Resin • Column packing • Batch separation • Chromatofocusing – protein separation on p. H basis

Storage of Resins • Dry form are stable for many years • Aqueous slurried ion exchangers are still useful after several months • Microbial growth • Sodium azide (0. 02%) is suitable for cation exchangers, • Phenylmercuric salts (0. 001%) are effective for anion exchangers
- Slides: 11