Ion beam Analysis Joele Mira from UWC and

Ion beam Analysis Joele Mira from UWC and i. Themba LABS Tinyiko Maluleke from US 10/10/2008 Supervisor: Summer School 2008 Dr. SA-JINR Alexander Kobzev

Contents ØDescriptions of Van de Graaf ØRutherford back-scattering (RBS) ØRBS and Elastic recoil detection (ERD) ØRBS and Proton induced X-ray emission (PIXE) ØConclusion 10/10/2008 SA-JINR Summer School 2008 2
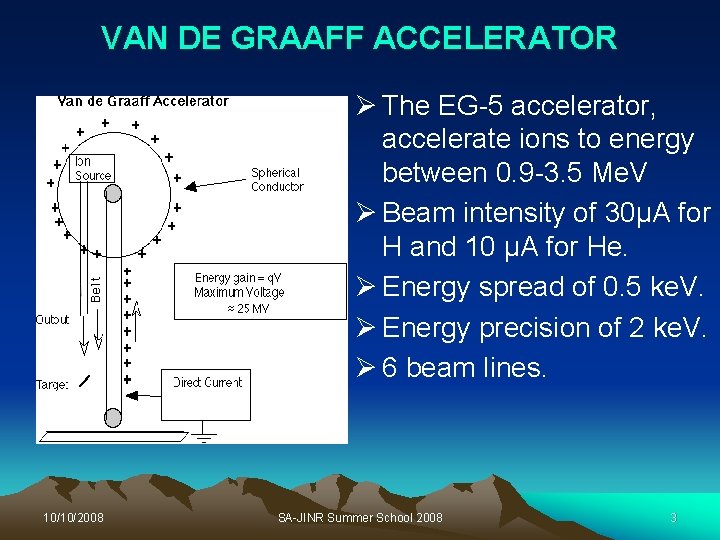
VAN DE GRAAFF ACCELERATOR Ø The EG-5 accelerator, accelerate ions to energy between 0. 9 -3. 5 Me. V Ø Beam intensity of 30μA for H and 10 μA for He. Ø Energy spread of 0. 5 ke. V. Ø Energy precision of 2 ke. V. Ø 6 beam lines. 10/10/2008 SA-JINR Summer School 2008 3

Introduction to RBS Ø The use of RBS is to provide information on concentration vs depth for heavy element in a light material. + Ø A beam of 2 -3 Me. V He ions are directed at different angles on a sample surface. Ø The ion loses energy due to collision with electrons. Ø The ion will scatter elastically with the atomic nucleus and lead to a kinematic factor K, 10/10/2008 SA-JINR Summer School 2008 4
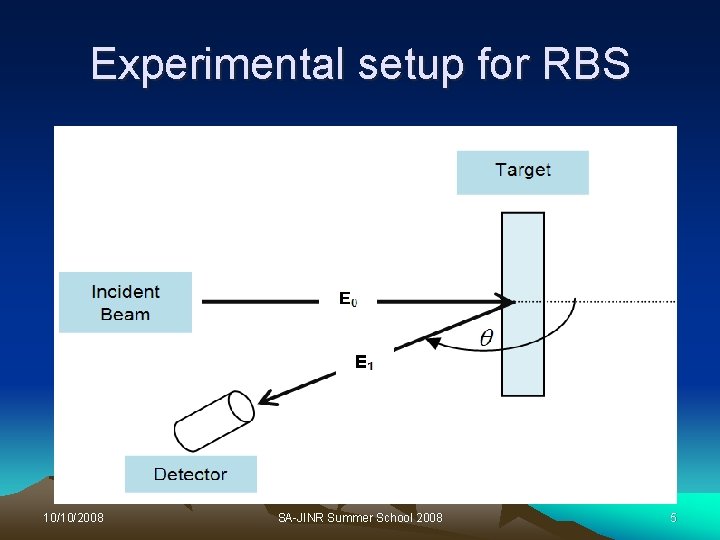
Experimental setup for RBS 10/10/2008 SA-JINR Summer School 2008 5
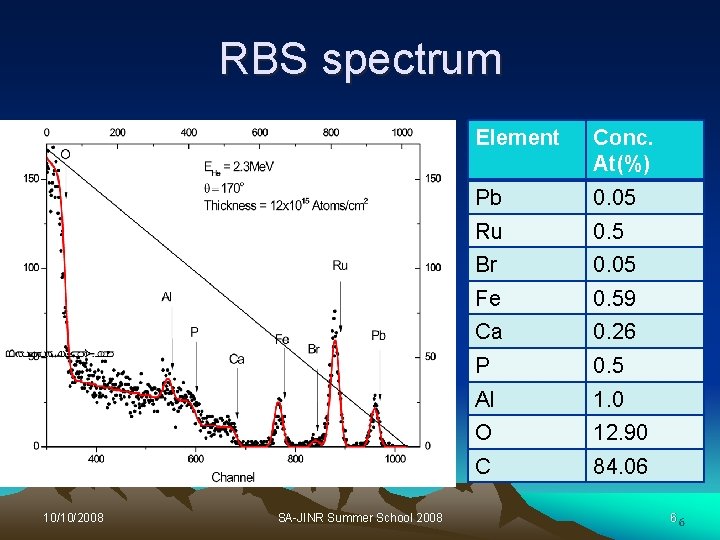
RBS spectrum 10/10/2008 SA-JINR Summer School 2008 Element Conc. At(%) Pb 0. 05 Ru 0. 5 Br 0. 05 Fe 0. 59 Ca 0. 26 P 0. 5 Al 1. 0 O 12. 90 C 84. 06 66

10/10/2008 SA-JINR Summer School 2008 7

Elastic Recoil Detection (ERD) Ø ERD is a complimentary technique to RBS Ø It is used to measure concentration of H atoms in the thin layers, and in the near surface region of material. Ø The incident beam is directed at a grazing angle onto the sample surface. Ø The recoiling atoms are ejected and detected at forward angle. Ø A thin foil is placed in front of the detector to stop elastically scattered incident ion beam and all atoms with mass heavier than the beam. 10/10/2008 SA-JINR Summer School 2008 8
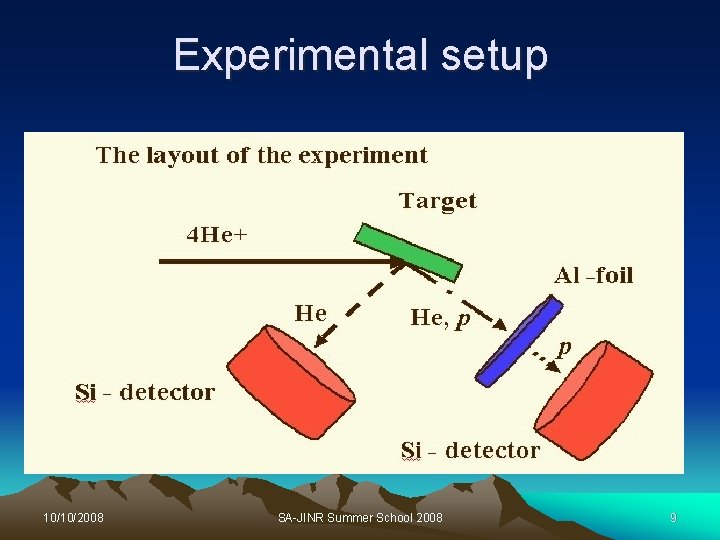
Experimental setup 10/10/2008 SA-JINR Summer School 2008 9
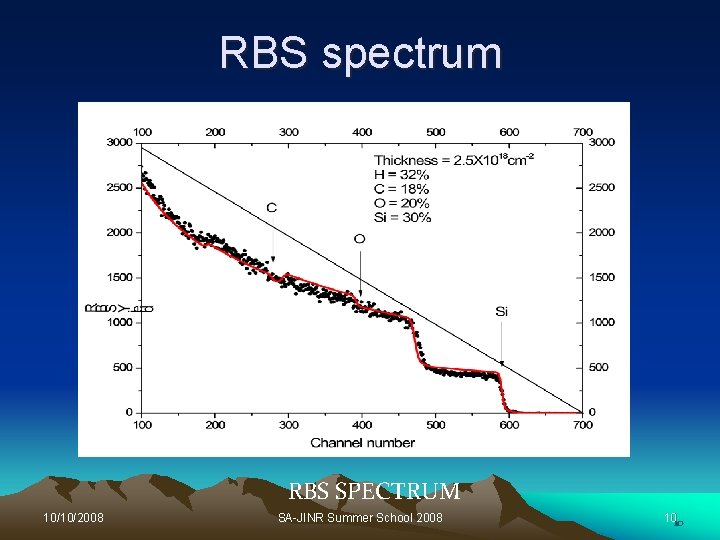
RBS spectrum RBS SPECTRUM 10/10/2008 SA-JINR Summer School 2008 1010
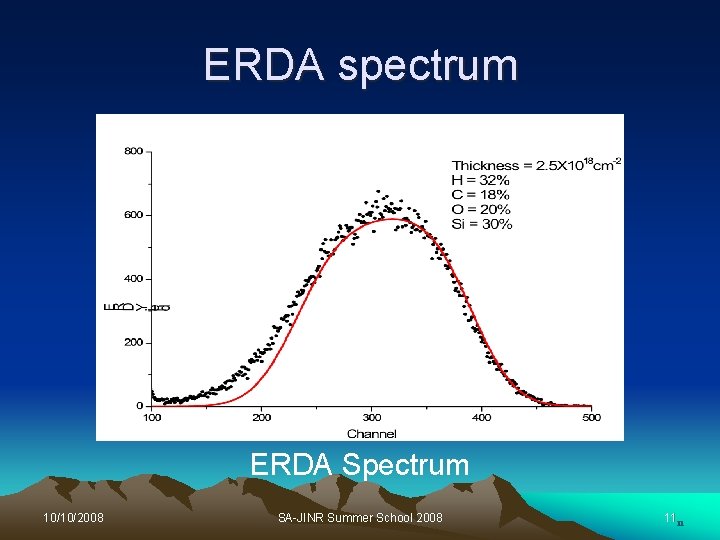
ERDA spectrum ERDA Spectrum 10/10/2008 SA-JINR Summer School 2008 1111

Proton Induced X-ray Emission (PIXE) Ø Occurs when a sample is bombarded with the beam, the proton interact with the electrons in the atoms of the sample, creating an inner shell vacancy Ø The X-ray is emitted when an electron from outer shell fills the hole left by an electron. Ø The energy of the X-rays emitted are characteristic of the element from which they originate. Ø The number of emitted X-rays is proportional to the amount of the corresponding element within the sample. 10/10/2008 SA-JINR Summer School 2008 12
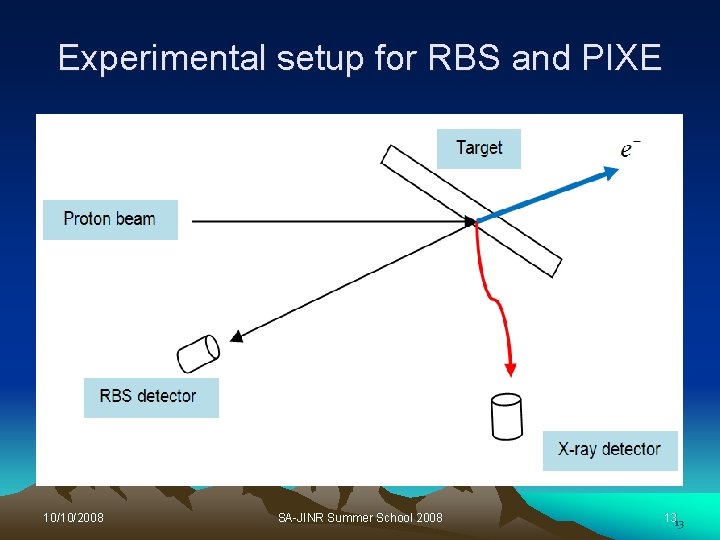
Experimental setup for RBS and PIXE 10/10/2008 SA-JINR Summer School 2008 1313
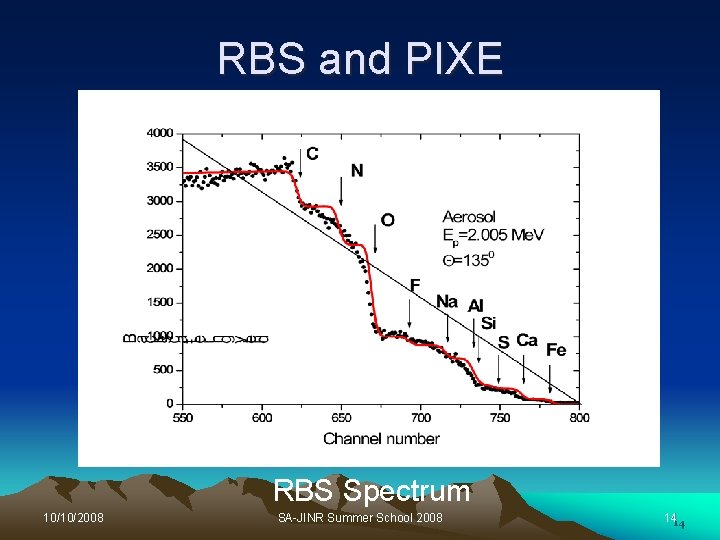
RBS and PIXE RBS Spectrum 10/10/2008 SA-JINR Summer School 2008 1414
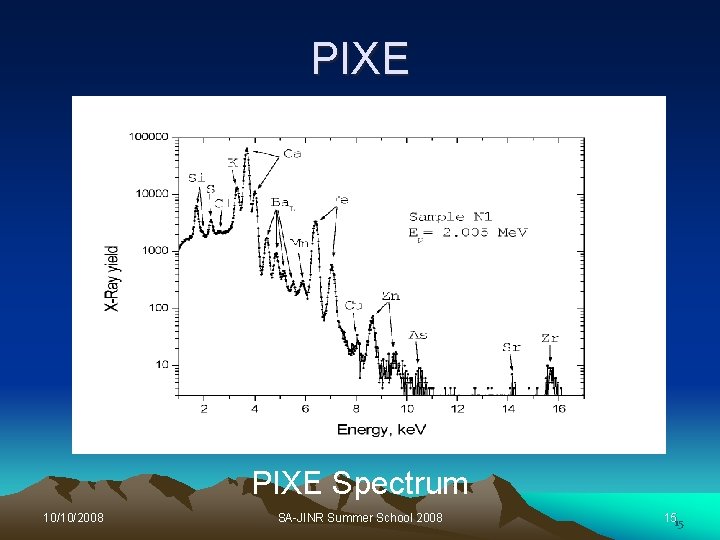
PIXE Spectrum 10/10/2008 SA-JINR Summer School 2008 1515
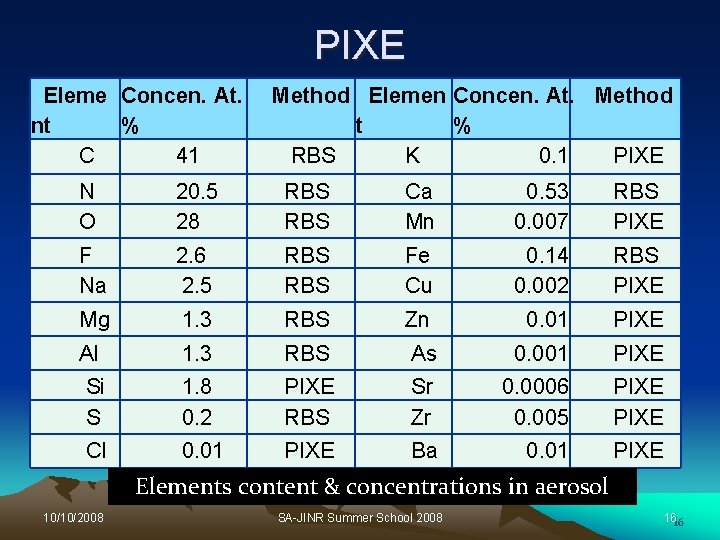
PIXE Eleme Concen. At. nt % C 41 Method Elemen Concen. At. Method t % RBS K 0. 1 PIXE N O 20. 5 28 RBS Ca Mn 0. 53 0. 007 RBS PIXE F Na 2. 6 2. 5 RBS Fe Cu 0. 14 0. 002 RBS PIXE Mg 1. 3 RBS Zn 0. 01 PIXE Al 1. 3 RBS As 0. 001 PIXE Si S 1. 8 0. 2 PIXE RBS Sr Zr 0. 0006 0. 005 PIXE Cl 0. 01 PIXE Ba 0. 01 PIXE Elements content & concentrations in aerosol 10/10/2008 SA-JINR Summer School 2008 1616

Conclusion Ø The use of ion beam analysis is non-destructive, high accuracy and easy to interpret the experimental results. Ø The use of these models allow the determination of different elements from Hydrogen to heavy elements concentrated in samples. Ø It also allow the analysis of very thin sample of about 10 nm. Ø Ion beam analysis is applied in various fields such as microelectronics, environmental monitoring etc. 10/10/2008 SA-JINR Summer School 2008 17

Thanks for your attention!! 10/10/2008 SA-JINR Summer School 2008 18
- Slides: 18