INVESTOR DECK A USbased Biotechnology Company A unique

INVESTOR DECK A US-based Biotechnology Company

• A unique natural extract was discovered in a remote tropical region of Australia around 2007 by Government scientists • Intense subsequent research for over a decade both in Australia and overseas confirms its remarkable medicinal properties • World’s first strong –and only natural antibiotic – totally unique 2019 EXECUTIVE SUMMARY • Ground-breaking USA research in 2015 confirms its “never seen this before” status and “Fabulous” anti-inflammatory effects • USA research also confirmed the natural extracts ability to treat multiple neural pathways –again a world first • Inflammatory capability indicates potential superiority to any existing inflammatory drugs, including the world’s best-selling drug for arthritis • Gen. Bio aims to be the first company to undertake research and identify the core molecule(s) driving these amazing results - and will make huge capital gains • Gen. Bio’s new natural molecules, based on this remarkable new natural extract, is aimed at enhancing the existing processes used in the treatment of arthritis, pain relief, inflammation, MRSA, obesity, anti-aging, high blood pressure, UTI, asthma and Alzheimer’s • The Company also possesses trade secrets for developing a range of infusion-based technology food /nutraceutical goods associated with the novel extract • Gen. Bio Inc. has already conducted discovery/assessment research in the medicinal usage and potentiality of the natural extract while gaining a level of unrivaled expertise • Rapid trade sale within 36 months at significant value expected

• Gen. Bio Inc. is seeking $1, 000 U. S. D. to commence first phase research development to eventual Investigational New Drug [IND] approval inflexion point 1 • Series ‘A’ finance to follow rapidly [within 12 months] once initial discovery phase is complete and further research needs to commence • A further three years of IND enabling toxicology studies in the USA will position the company for a trade sale at IND (Investigational New Drug) inflection point. • Specialized USA and international scientific research teams are ready to begin the drug program once funds are secured • Gen. Bio Inc. has expertise in this category, unrivaled by any competitor • Strategy is a trade sale at IND approval / Phase 1 inflection point within 24/36 months • Substantial pre-qualified leads with pharmaceutical companies have been generated through international attendance at overseas Biotech trade shows • Significant ROI. Conservative financial e. NPV modeling-based on independent valuation suggest ROI in excess of 100% annualized, based on three-year projections.

• 1/10/19 Letter of Intent (LOI) signed with a USA based Nutraceutical Company. [California] • 2/13/19 Licensing agreement with payment milestones [and upfront] countersigned and received CURRENT COMMERCIAL DEVELOPMENTS • Gen. Bio is primarily focused on drug research to IND level only. In addition to its main objective, Gen. Bio recognizes that a valuable and lucrative “spin-off’ is its ability to design an entirely new range of novel nutraceuticals with molecular designed and enhanced medical properties through proprietary extraction technology. Such a strategy can offer prospective clients’ clear differentiation in their respective channels and a clear USP [Unique Selling Point] in the crowded marketplace • For media announcement, please refer to our web site at www. Gen. Bioinc. com

OUR MISSION TO BE A WORLD LEADER IN THE BIOTECH ARENA, SPECIALIZING IN THE TRANSFORMATION OF NATURAL TRADITIONAL THERAPIES INTO MODERN, PROVEN MEDICINES. Our Vision To identify then develop a more efficacious, less toxic and disruptive molecule for the treatment of inflammatory conditions.

The Commercial Opportunity Clinical Applications for the Research • The discovery of a group of American scientists in 2014 who have, quite simply, made a remarkable and highly confidential discovery. • This current IP represents up to several million dollars of research investment to date and enables the company to leverage its expertise and execute on highly focused research with minimal time delays. • Gen. Bio Inc. will announce major scientific advances and breakthroughs to the world only after IP protection is secured. Identifying and patenting how the new molecule[s] is the key to commercial success. • A Chief Scientific Officer will be appointed in Davis, California, to oversee US research activities. The same scientific team will be working off a highly potent selective inhibition activity in a botanical extract. • Inhibitors are used to treat pain and inflammation. They are a subclass of the non-steroidal anti-inflammatory drug (NSAID) class, which include well-known compounds such as ibuprofen and aspirin. Selective COX-2 inhibition avoids the gastrointestinal side-effects that limit the clinical applications of most NSAIDs. • Gen. Bio Inc. ’s business model is to advance the lead drug candidate to the value inflection point of Investigational New Drug [IND] approval while concurrently ring-fencing intellectual property with aggressive patenting. Gen. Bio Inc. will then seek to sell the asset to a pharmaceutical company, interested in obtaining a drug to strengthen their portfolio of antibiotic drugs. • The Company has already implemented a marketing program and commenced building a significant database of suitable pharmaceutical companies, both domestically and internationally, that will fit the profile of a trade sale partner. Preliminary contact has occurred at various international trade shows with appropriate multinationals, and more formal negotiations can commence once further research in dosage/toxicology has been successfully completed.
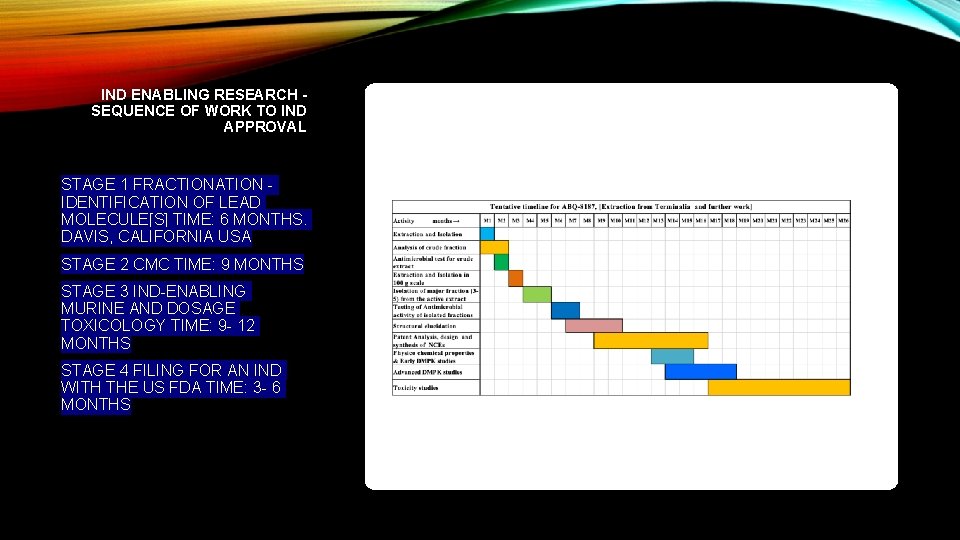
IND ENABLING RESEARCH - SEQUENCE OF WORK TO IND APPROVAL STAGE 1 FRACTIONATION IDENTIFICATION OF LEAD MOLECULE[S] TIME: 6 MONTHS. DAVIS, CALIFORNIA USA STAGE 2 CMC TIME: 9 MONTHS STAGE 3 IND-ENABLING MURINE AND DOSAGE TOXICOLOGY TIME: 9 - 12 MONTHS STAGE 4 FILING FOR AN IND WITH THE US FDA TIME: 3 - 6 MONTHS

MARKET OPPORTUNITY • The Gen. Bio Inc. exit strategy is to be acquired in the next 18 -36 months, at a price that adequately reflects the substantial market size for a patented COX-2 inhibitor. • No other company has such proprietary intellectual property at the current time
- Slides: 8