Investigation strategies and methods Antigen and antibody detection

Investigation strategies and methods Antigen and antibody detection May 2007 Laboratory Training for Field Epidemiologists

Learning objectives At the end of the presentation, participants should • Understand direct and indirect antibody detection • Understand the different methods for detecting antigens or antibodies Laboratory Training for Field Epidemiologists

Detection • Detection of antigen-antibody complex • Antigen-antibody complex requires specific conditions • temperature • p. H • Complex may be directly visible or invisible Laboratory Training for Field Epidemiologists

Detection Directly visible – agglutination Invisible • requires specific probes (enzyme-labelled antiimmunoglobulin, isotope-labelled anti-immunoglobulin, etc. ) • binds Ag-Ab complex and amplifys signals • signals can be measured by naked eyes or specific equipment e. g. in ELISA, RIA, IFA Laboratory Training for Field Epidemiologists

Methods for Ag-Ab detection • Precipitation • Agglutination • Hemagglutination and hemagglutination inhibition • Viral neutralization test • Radio-immunoassays • ELISA • Immunoflourescence • Immunoblotting • Immunochromatography Laboratory Training for Field Epidemiologists

Precipitation Principle • soluble antigen combines with its specific antibody • antigen-antibody complex is too large to stay in solution and precipitates Examples • flocculation test • immuno-diffusion test • counter-immuno-electrophoresis (CIEP) Laboratory Training for Field Epidemiologists

Flocculation test (precipitation reaction) Principle • precipitate, a concentrate of fine particles, is usually visible (macroscopically or microscopically) because the precipitated product is forced to remain suspended Examples • VDRL slide flocculation test • RPR card test • Kahn’s test for syphilis Laboratory Training for Field Epidemiologists

Flocculation test (A precipitation reaction) (1) Non Reactive (2) Weakly Reactive RPR card test Laboratory Training for Field Epidemiologists (3, 4) Reactive

Precipitation: Performance, applications • Advantages • sensitive for antigen detection • Limited applications • Time taken - 10 minutes Laboratory Training for Field Epidemiologists

Direct agglutination Principle • • combination of an insoluble particulate antigen with its soluble antibody • forms antigen-antibody complex • particles clump/agglutinate Ag-Ab complex used for antigen detection Examples • bacterial agglutination tests for sero-typing and sero-grouping e. g. , Vibrio cholerae, Salmonella spp Laboratory Training for Field Epidemiologists Positive Negative

Passive (indirect) agglutination Principle • precipitation reaction converted into agglutination coating antigen onto the surface of carrier particles like red blood cells, latex, gelatin, bentonite • background clears Examples of types • latex agglutination • co-agglutination • passive hemagglutination (treated red blood cells made resistant) Examples of tests - agglutination for leptospirosis Widal test (typhoid fever) Laboratory Training for Field Epidemiologists

Reverse passive agglutination Principle • antigen binds to soluble antibody coated on carrier particles and results in agglutination • detects antigens Example • detecting cholera toxin Laboratory Training for Field Epidemiologists
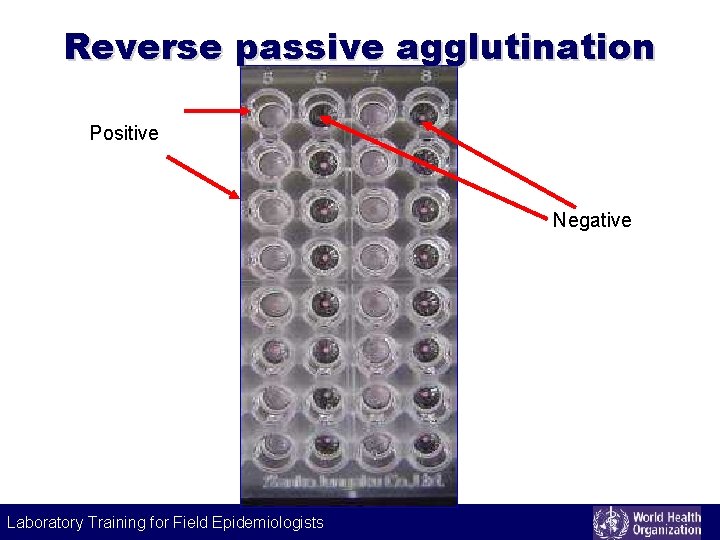
Reverse passive agglutination Positive Negative Laboratory Training for Field Epidemiologists

Agglutination: Performance, applications Advantages • sensitive for antibody detection Limitations • Prozone phenomenon: • requires the right combination of quantities of antigen and antibody • handled through dilution to improve the match Time taken • 10 -30 minutes Laboratory Training for Field Epidemiologists

Hemagglutination Principle • many human viruses have the ability to bind to the surface structures on red blood cells from different species thereby causing agglutination Example • influenza virus binds to fowl’s red blood cells Laboratory Training for Field Epidemiologists
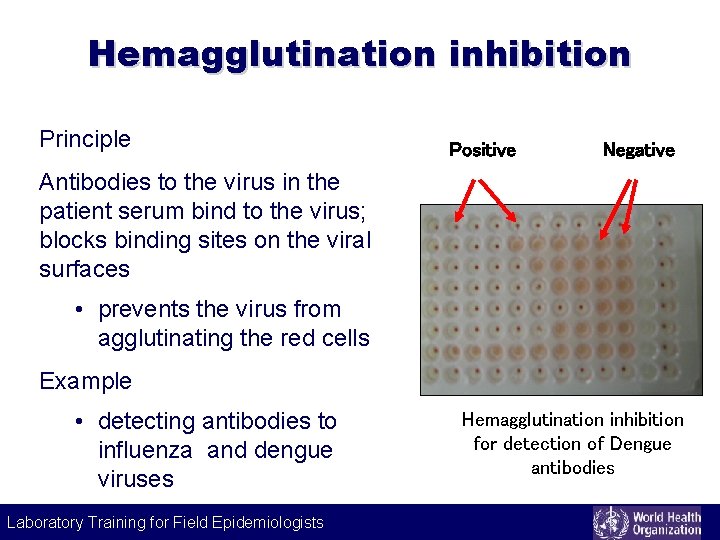
Hemagglutination inhibition Principle Positive Negative Antibodies to the virus in the patient serum bind to the virus; blocks binding sites on the viral surfaces • prevents the virus from agglutinating the red cells Example • detecting antibodies to influenza and dengue viruses Laboratory Training for Field Epidemiologists Hemagglutination inhibition for detection of Dengue antibodies

Hemagglutination: Performance, applications Advantages • highly specific • can be used as gold standard Limitations • technically demanding • time consuming • cannot distinguish Ig. G from Ig. M Time taken • 1 day Laboratory Training for Field Epidemiologists

Principle Neutralization assays • antibodies in serum neutralize antigens on the surface of viruses (neutralizing antibodies) • inhibited viruses cannot infect cell lines Example • plaque neutralization assay for dengue virus, Japanese encephalitis virus • antibodies to bacterial toxins and other extra-cellular products that display measurable activities (e. g. , ASLO, diphtheria toxin, clostridium toxin) Laboratory Training for Field Epidemiologists

Neutralization: Performance, applications • Advantages • Highly specific • Often used as gold standard • Limitations • Technically demanding • Time consuming • Can only be used for viruses that can be grown • Complexity limits the use beyond gold standard • Time taken • 1 week Laboratory Training for Field Epidemiologists

Radio-immunoassays • Principle • Radioactively labelled-antibody (or antigen) competes with the patient’s unlabelled antibody (or antigen) for binding sites on a known amount of antigen (or antibody) • Reduction in radioactivity of the antigen-patient antibody complex compared with control test is used to quantify the amount of patient antibody / antibody bound • Limited use due to the problems with handling radioisotope Example • HBs. Ag • Thyroid function test Response • Antibody Laboratory Training for Field Epidemiologists
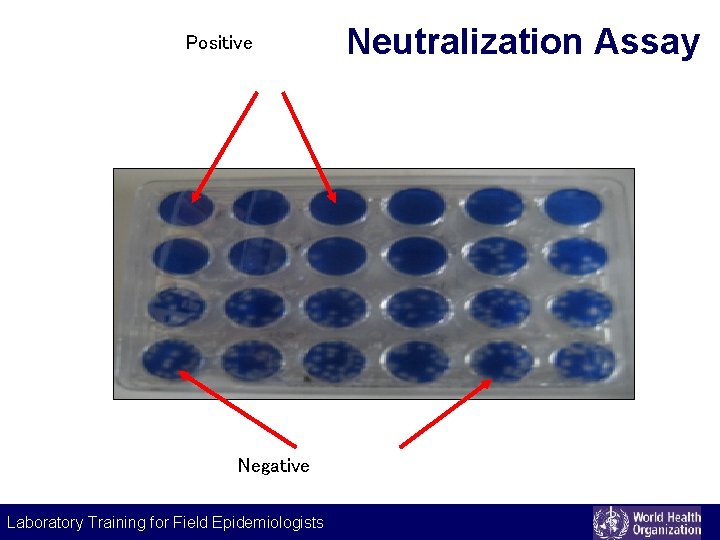
Positive Negative Laboratory Training for Field Epidemiologists Neutralization Assay

Radio-immunoassays: Performance, applications Adantages • highly sensitive • can be used for detection of small quantities • quantification possible Limitations • expensive • requires isotopes Time taken • 1 day Laboratory Training for Field Epidemiologists

Labeling technique Enzyme-linked immunosorbant assay (ELISA) Principle • use of enzyme-labelled immunoglobulin to detect antigens or antibodies • signals are developed by the action of hydrolyzing enzyme on chromogenic substrate • optical density measured by micro-plate reader Examples • Hepatitis A (Anti-HAV-Ig. M, anti-HAV Ig. G) Laboratory Training for Field Epidemiologists
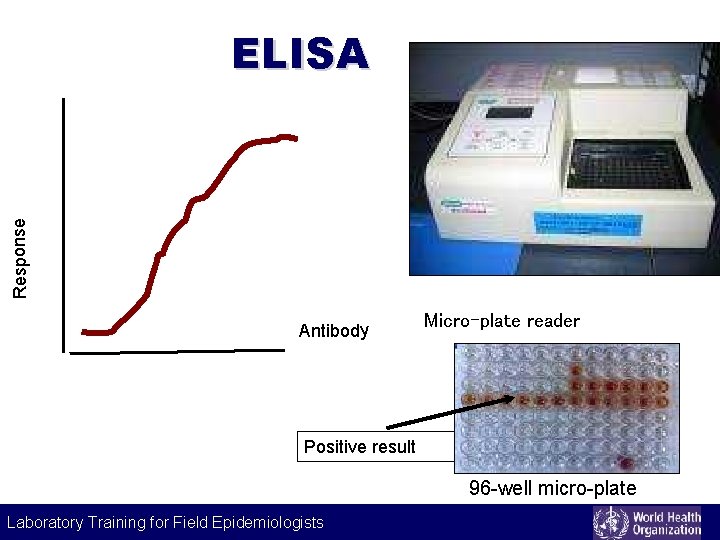
Response ELISA Antibody Micro-plate reader Positive result 96 -well micro-plate Laboratory Training for Field Epidemiologists

Labeling technique Types of ELISA (Ag Ab tests) Competitive • Antigen or antibody are labelled with enzyme and allowed to compete with unlabeled ones (in patient serum) for binding to the same target • Hydrolysis signal from Ag-Ab complex (enzyme-labelled) is measured • Antigen or antibody in serum is then calculated • No need to remove the excess/unbound Ag or Ab from the reaction plate or tubes) Laboratory Training for Field Epidemiologists

Labeling technique Types of ELISA used in the detection of antigens and antibodies • Non-competitive • must remove excess/unbound Ag or Ab before every step of reactions • Direct ELISA • Indirect ELISA • Sandwich ELISA • Ab Capture ELISA (similar to sandwich ELISA but in 1 st step, anti-Ig (M or G) is coated on the plate • Then antibodies in patient serum are allowed to capture in next Laboratory Training for Field Epidemiologists

ELISA: Performance, applications • Advantages • Automated, inexpensive • Objective • Small quantities required • Class specific antibodies measurable • Limitations • Expensive initial investment • Variable sensitivity / specificity of variable tests • Cross contamination Time taken - 1 day • Laboratory Training for Field Epidemiologists

Performance comparison of various ELISAs for antibody detection Performance characteristic Noncompetitive ELISA Capture ELISA Purpose Antibody Best for class specific antibody Sensitivity ++ ++ ++ Specificity ++ ++ +++ Cost + ++ +++ Ease of performance ++ ++ Time taken ++ + +++ Laboratory Training for Field Epidemiologists
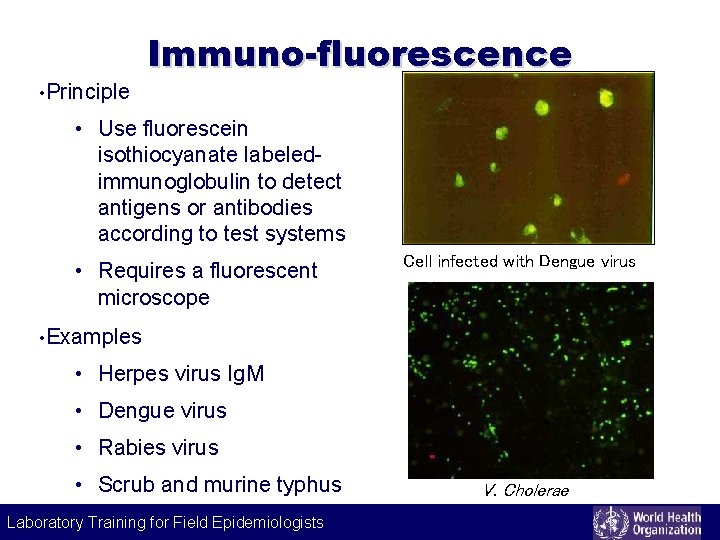
Labeling technique Immuno-fluorescence • Principle • Use fluorescein isothiocyanate labeledimmunoglobulin to detect antigens or antibodies according to test systems • Requires a fluorescent microscope Cell infected with Dengue virus • Examples • Herpes virus Ig. M • Dengue virus • Rabies virus • Scrub and murine typhus Laboratory Training for Field Epidemiologists V. Cholerae

Immuno-fluorescence: Performance, applications • Advantages • Sensitive and specific • Can be used for discrepant analysis • Limitations • Expensive (Reagents and equipment) • Subjective • Cross reactivity • Non-specific immuno-fluorescence • Time taken • Training 1 dayfor Field Epidemiologists Laboratory

Labeling technique Types of immuno-fluorescence • Direct immunofluorescence • Used to detect antigen • Indirect and sandwich immuno-fluorescence • Antigen detection • Antibody detection Laboratory Training for Field Epidemiologists

Western-blot analysis (1) • Principle • Antigens are separated by Poly Acrylomide Gel Electrophoresis (PAGE) and trans-blotted onto nitrocellulose/nylon membranes • Antibodies in serum react with specific antigens • Signals are detected according to the principles of test systems • Antibodies against microbes with numerous cross-reacting antibodies identified more specifically • Examples • T. pallidum, B. burgdorferi, • Herpes simplex virus types 1 and 2 Laboratory Training for Field Epidemiologists Anti HIV-1

Western-blot analysis (2) • Serum, saliva, urine can be tested • Kits are commercially available • Recombinant immuno-blotting assays (RIBA) uses recombinant proteins Anti HIV-2 Laboratory Training for Field Epidemiologists

Immunoblot: Performance, applications • Advantages • Used for discrepant analysis • Highly specific • Rapid kits available • Limitations • Cost • Concern validated data • Time taken • 1 day Laboratory Training for Field Epidemiologists

Immuno-chromatography: Principle (1) • Dye-labelled antibody, specific for target antigen, is present on the lower end of nitrocellulose strip or in a plastic well provided with the strip. • Antibody, also specific for the target antigen, is bound to the strip in a thin (test) line • Either antibody specific for the labelled antibody, or antigen, is bound at the control line Bound AB Lysing agend Labled AB. Test band Control band (bound AB) Free labled AB Nitrocellulose strip Laboratory Training for Field Epidemiologists

Immuno-chromatography: Principle (2) • If antigen is present, some labelled antibody will be trapped on the test line • Excess-labelled antibody is trapped on the control line Captured Ag-labelled Captured labelled Ab-complex Ab Labelled AB-AGcomplex Captured by bound AB of test band Labelled AB-AGcomplex Captured by bound AB of control band Laboratory Training for Field Epidemiologists

Immuno-chromatography: Performance, applications • Advantages • Commercially available • Single use, rapid test • Easy to perform • Can detect antigen or antibody • Can be used in the field • Limitations • Cost • Concern validated data Time taken - 1 hour • Laboratory Training for Field Epidemiologists

Interpretation of antigen detection tests • In general, detection of the antigen denotes a presence of the pathogen • More important in some of parasitic and fungal diseases Antigen test Positive Negative Interpretation • Current or recent infection • No infection • Insufficient number of organisms • Sensitivity of testing is low (Consider test by test) Laboratory Training for Field Epidemiologists

Interpretation of a single, acute Ig. M test Interpretation Negative • No Positive (Newborn) • Congenital current infection Positive (Adult) • Primary infection Laboratory Training for Field Epidemiologists or current

Interpretation of two, acute and convalescent Ig. G tests * Test Negative Interpretation • No current infection • Past infection • Immuno- Positive (4 -fold rise or fall in titer) suppression • Recent infection * Convalescent serum collected 2 -4 weeks after onset Laboratory Training for Field Epidemiologists

Interpretation of a single Ig. G test Test Interpretation Negative • No Positive (Newborn) • Maternal Positive (Adult) • Evidence exposure or immuno-suppression antibodies crossed the placenta of infection at some undetermined time • Infection in some cases (e. g. , rabies, legionella, Ehrlichia) • May be significant if immuno-suppression (e. g. , AIDS) * Collected between onset and convalescence Laboratory Training for Field Epidemiologists

Elements influencing the sensitivity and specificity of a given test kit • Test format • Precipitation versus IFA, Rapid test versus ELISA • Purity of the antigen used • Crude versus purified antigen versus synthetic peptides • Type of the antibody used • Polyclonal versus monoclonal antibodies • Interfering substances in the sample • Presence of rheumatoid factor in the serum of the patient • Similarity in antigenic composition of pathogens • Cross reactivity Laboratory Training for Field Epidemiologists

Investigation strategies and methods Developed by: The Department of Epidemic and Pandemic Alert and Response of the World Health Organization with the assistance of: European Program for Field Epidemiology Canadian Field Epidemiology Programme Thailand Ministry of Health Institut Pasteur Laboratory Training for Field Epidemiologists Training
- Slides: 43