Investigation of the ammonia oxidation over copper with
- Slides: 1

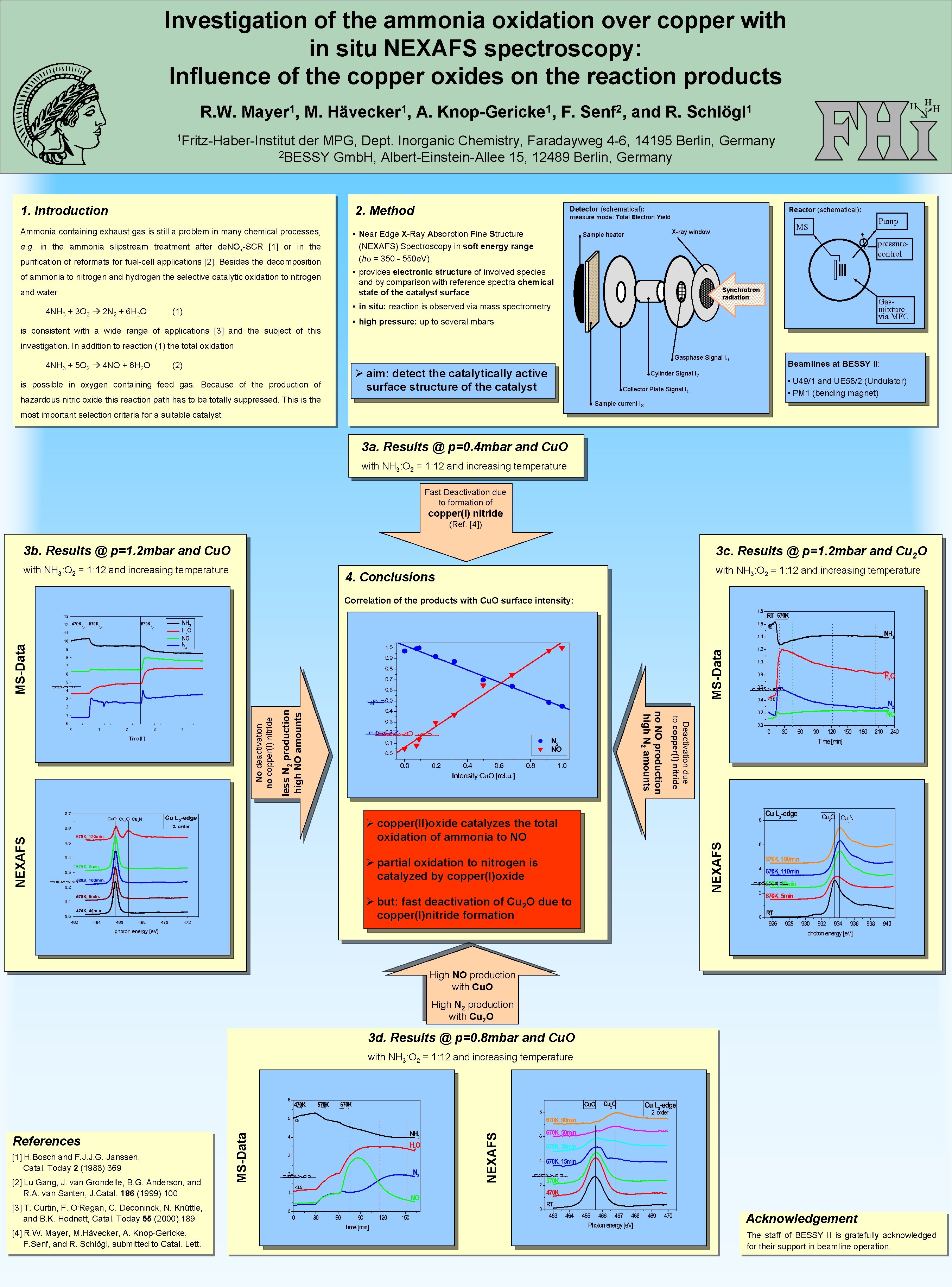
Investigation of the ammonia oxidation over copper with in situ NEXAFS spectroscopy: Influence of the copper oxides on the reaction products R. W. 1 Mayer , M. 1 Hävecker , A. 1 Knop-Gericke , F. 2 Senf , and R. 1 Schlögl 1 Fritz-Haber-Institut der MPG, Dept. Inorganic Chemistry, Faradayweg 4 -6, 14195 Berlin, Germany 2 BESSY Gmb. H, Albert-Einstein-Allee 15, 12489 Berlin, Germany 2. Method 1. Introduction Ammonia containing exhaust gas is still a problem in many chemical processes, e. g. in the ammonia slipstream treatment after de. NOx-SCR [1] or in the purification of reformats for fuel-cell applications [2]. Besides the decomposition of ammonia to nitrogen and hydrogen the selective catalytic oxidation to nitrogen and water 4 NH 3 + 3 O 2 2 N 2 + 6 H 2 O Detector (schematical): Reactor (schematical): measure mode: Total Electron Yield • Near Edge X-Ray Absorption Fine Structure (NEXAFS) Spectroscopy in soft energy range (hu = 350 - 550 e. V) MS X-ray window Sample heater pressurecontrol • provides electronic structure of involved species and by comparison with reference spectra chemical state of the catalyst surface Synchrotron radiation Gasmixture via MFC • in situ: reaction is observed via mass spectrometry (1) is consistent with a wide range of applications [3] and the subject of this Pump • high pressure: up to several mbars investigation. In addition to reaction (1) the total oxidation 4 NH 3 + 5 O 2 4 NO + 6 H 2 O Gasphase Signal IG (2) is possible in oxygen containing feed gas. Because of the production of Ø aim: detect the catalytically active surface structure of the catalyst hazardous nitric oxide this reaction path has to be totally suppressed. This is the Cylinder Signal IZ Beamlines at BESSY II: • U 49/1 and UE 56/2 (Undulator) • PM 1 (bending magnet) Collector Plate Signal IC Sample current IS most important selection criteria for a suitable catalyst. 3 a. Results @ p=0. 4 mbar and Cu. O with NH 3: O 2 = 1: 12 and increasing temperature Fast Deactivation due to formation of copper(I) nitride (Ref. [4]) 3 b. Results @ p=1. 2 mbar and Cu. O 3 c. Results @ p=1. 2 mbar and Cu 2 O with NH 3: O 2 = 1: 12 and increasing temperature 4. Conclusions less N 2 production high NO amounts Ø partial oxidation to nitrogen is catalyzed by copper(I)oxide NEXAFS Ø copper(II)oxide catalyzes the total oxidation of ammonia to NO Deactivation due to copper(I) nitride no NO production high N 2 amounts No deactivation no copper(I) nitride MS-Data Correlation of the products with Cu. O surface intensity: Ø but: fast deactivation of Cu 2 O due to copper(I)nitride formation High NO production with Cu. O High N 2 production with Cu 2 O 3 d. Results @ p=0. 8 mbar and Cu. O [1] H. Bosch and F. J. J. G. Janssen, Catal. Today 2 (1988) 369 [2] Lu Gang, J. van Grondelle, B. G. Anderson, and R. A. van Santen, J. Catal. 186 (1999) 100 NEXAFS References MS-Data with NH 3: O 2 = 1: 12 and increasing temperature [3] T. Curtin, F. O‘Regan, C. Deconinck, N. Knüttle, and B. K. Hodnett, Catal. Today 55 (2000) 189 Acknowledgement [4] R. W. Mayer, M. Hävecker, A. Knop-Gericke, F. Senf, and R. Schlögl, submitted to Catal. Lett. The staff of BESSY II is gratefully acknowledged for their support in beamline operation.