Investigating Specific Heat Capacity 27 February 2021 LO

Investigating Specific Heat Capacity 27 February 2021 LO: Investigate the specific heat capacity of different metals Do now: Convert the following from Kilo Joules into Joules 1. 8. 2 KJ 2. 9. 1 KJ 3. 108 KJ 4. 548 KJ 5. 640 KJ Now Convert these from Joules in Kilo Joules 1. 98 000 J 2. 102 000 J 3. 12 000 J 4. 980 000 J 5. 802 000 J 1. 2. 3. 4. 5. 8 200 J 9 100 J 108 000 J 548 000 J 640 000 J 1. 2. 3. 4. 5. 98 KJ 102 KJ 12 KJ 908 KJ 802 KJ
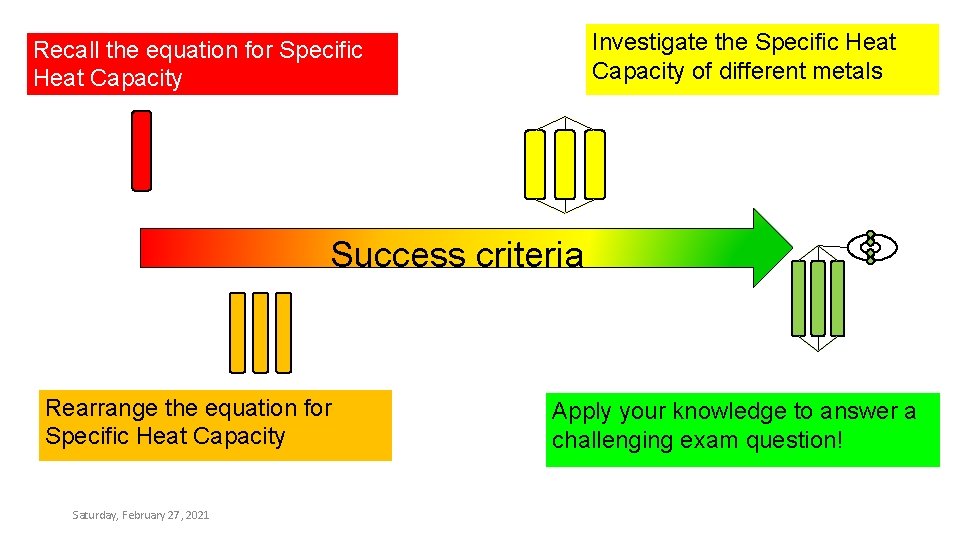
Investigate the Specific Heat Capacity of different metals Recall the equation for Specific Heat Capacity Success criteria Rearrange the equation for Specific Heat Capacity Saturday, February 27, 2021 Apply your knowledge to answer a challenging exam question!
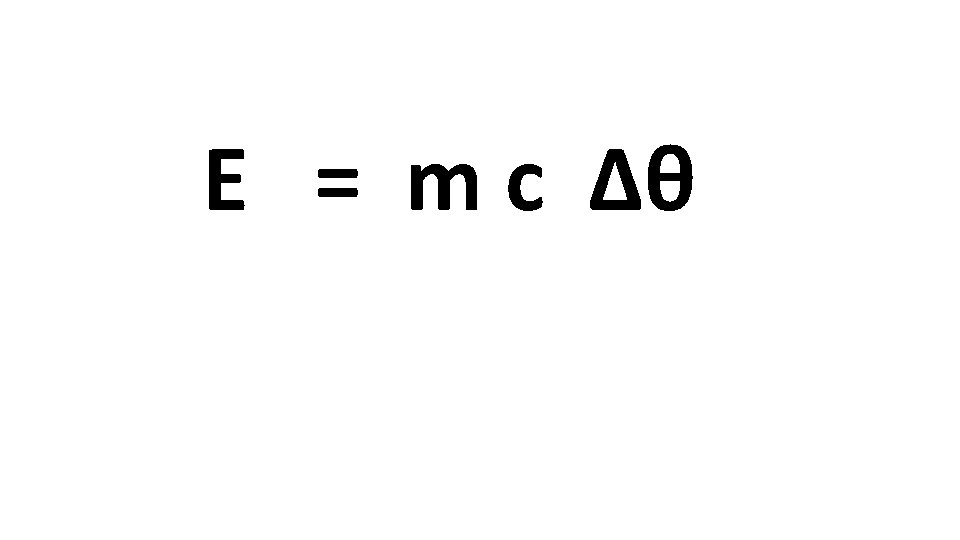

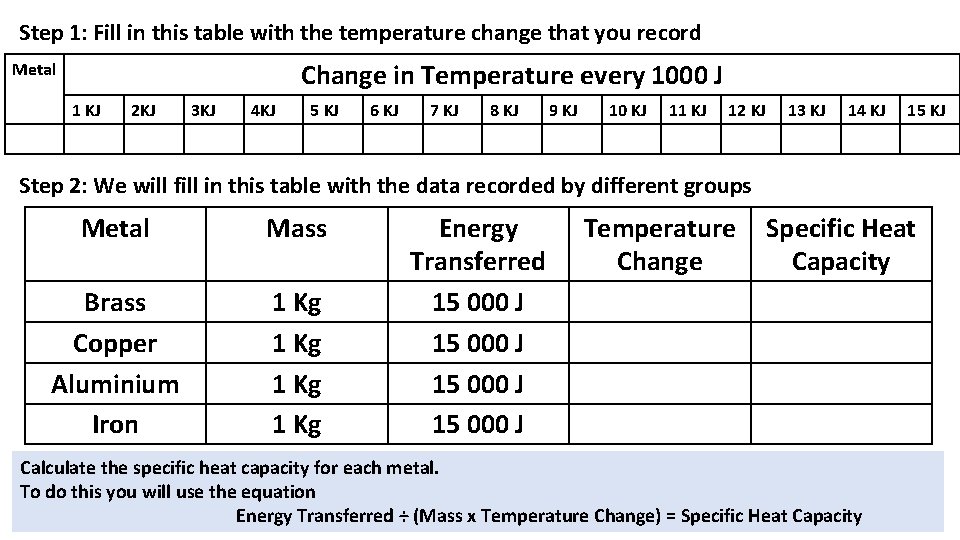
Step 1: Fill in this table with the temperature change that you record Change in Temperature every 1000 J Metal 1 KJ 2 KJ 3 KJ 4 KJ 5 KJ 6 KJ 7 KJ 8 KJ 9 KJ 10 KJ 11 KJ 12 KJ 13 KJ 14 KJ 15 KJ Step 2: We will fill in this table with the data recorded by different groups Metal Mass Brass Copper Aluminium Iron 1 Kg Energy Transferred 15 000 J Temperature Change Specific Heat Capacity Calculate the specific heat capacity for each metal. To do this you will use the equation Energy Transferred ÷ (Mass x Temperature Change) = Specific Heat Capacity

Tasks: Finished? Collect some graph paper and draw a graph of your results

Exam Question Plenary Self Assess: 1 a). Specific heat capacity is the amount of energy required to heat 1 kg of a substance by 1ᴼC. b). Specific heat capacity = energy / (mass x change in temperature) Change in temperature = 41. 3 – 13. 1 = 28. 2 ᴼC 23 800 J / (0. 831 x 28. 2) = 1 015. 6 J/ kgᴼC c). Change in temperature: 62. 1 – 41 = 21. 1ᴼC 23 800 J / (0. 831 x 21. 1) = 13 57. 4 J/ kgᴼC d). The reading is greater because there are more energy losses this time because it is hotter (the temperature difference to its surroundings is higher) e). – Insulate the block of metal so that no heat is lost -- Allow the metal to cool back down again to the same start temperature each time.
- Slides: 7