Inversion Motions in the Dimethyl Ether Carbon Disulfide

Inversion Motions in the Dimethyl Ether. Carbon Disulfide Dimer Rebecca A. Peebles, Josh J. Newby and Sean A. Peebles Department of Chemistry Eastern Illinois University Charleston, IL 61920

Introduction • Increasing interest in C-H and other weak hydrogen bonds • Dimethyl ether (DME) complexes -Relatively acidic hydrogen atoms -Oxygen atom with lone pairs • DME-HCCH, 1 DME-CO 2, 2 DME-OCS 3 • DME-HF, 4 DME-HCl, 5 DME-Rg 6 1 Newby, J. J. ; Serafin, M. M. ; Peebles, R. A. ; Peebles, S. A. J. Phys. Chem. A. 2005, 109, 5316. J. J. ; Peebles, R. A. ; Peebles, S. A. J. Phys. Chem. A 2004, 108, 7372. 3 Newby, J. J. ; Peebles, R. A. ; Peebles, S. A. J. Phys. Chem. A 2004, 108, 11234. 4 Ottaviani, P. ; Caminati, W. ; Velino, B. ; Blanco, S. ; Lessarri, A. ; López, J. ; Alonso, J. Chem. Phys. Chem. 2004, 5, 336. 5 Ottaviani, P. ; Caminati, W. ; Velino, B. ; López, J. C. Chem. Phys. Lett. 2004, 394, 262. 6 Multiple references 2 Newby,
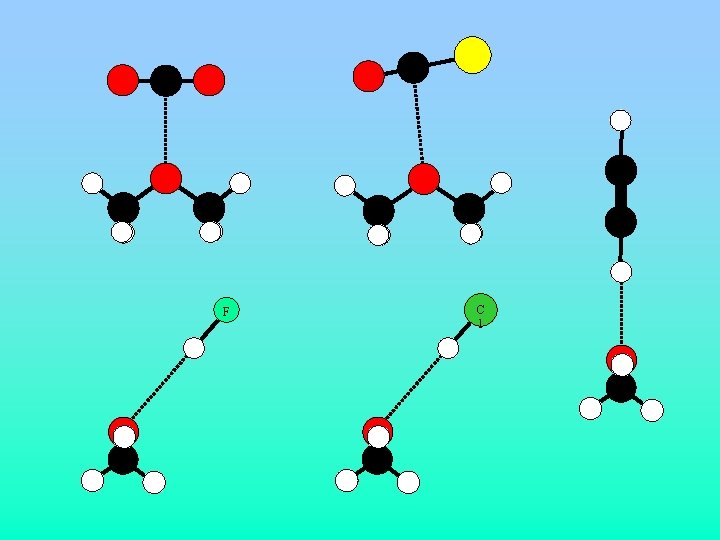
F C l

Experimental Methods • Balle-Flygare Fourier transform microwave spectrometer, 4. 5 – 16 GHz • ~1. 5% each DME and CS 2 pressurized to 2 atm with He/Ne • Stark effects with voltages up to 5 k. V – Calibrated with J = 1 0 of OCS
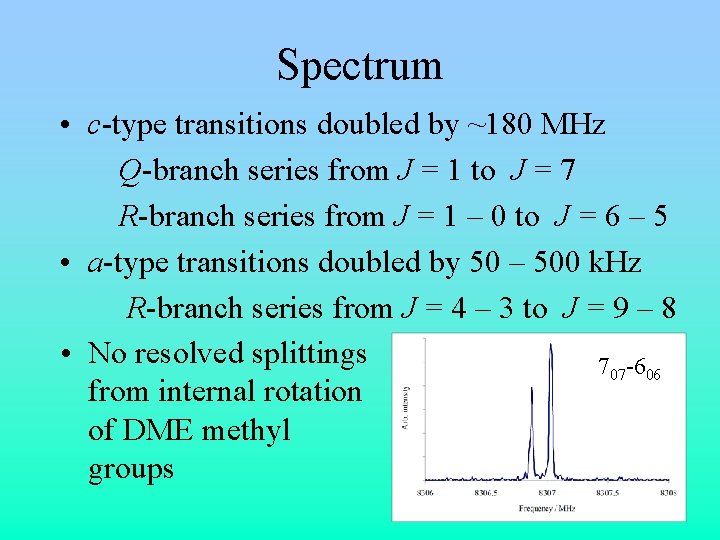
Spectrum • c-type transitions doubled by ~180 MHz Q-branch series from J = 1 to J = 7 R-branch series from J = 1 – 0 to J = 6 – 5 • a-type transitions doubled by 50 – 500 k. Hz R-branch series from J = 4 – 3 to J = 9 – 8 • No resolved splittings 707 -606 from internal rotation of DME methyl groups
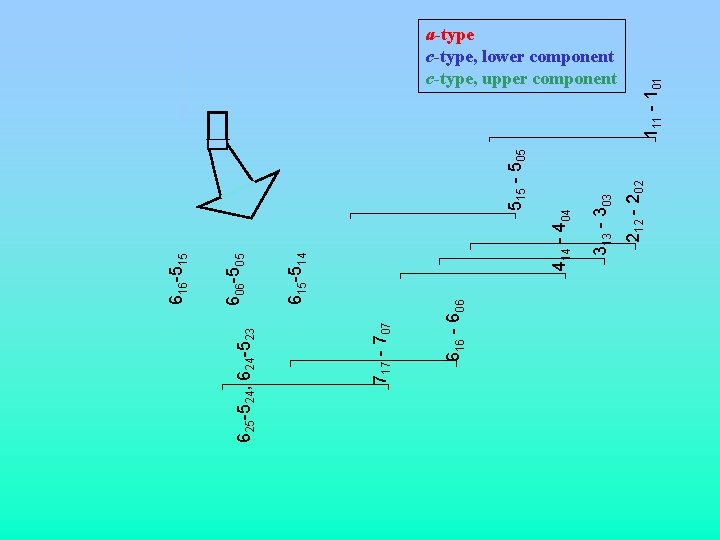
515 - 505 212 - 202 313 - 303 414 - 404 615 -514 606 -505 616 - 606 717 - 707 625 -524, 624 -523 616 -515 111 - 101 a-type c-type, lower component c-type, upper component
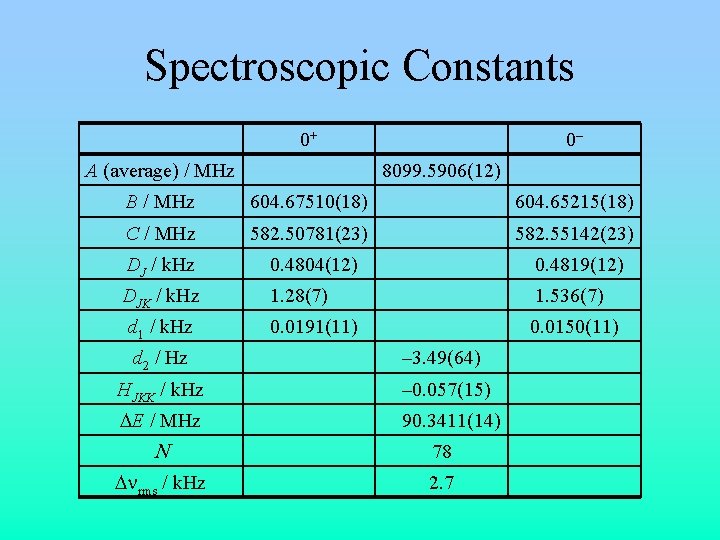
Spectroscopic Constants 0+ A (average) / MHz 0– 8099. 5906(12) B / MHz 604. 67510(18) 604. 65215(18) C / MHz 582. 50781(23) 582. 55142(23) DJ / k. Hz 0. 4804(12) 0. 4819(12) DJK / k. Hz 1. 28(7) 1. 536(7) 0. 0191(11) 0. 0150(11) d 1 / k. Hz d 2 / Hz – 3. 49(64) HJKK / k. Hz – 0. 057(15) DE / MHz 90. 3411(14) N 78 Dnrms / k. Hz 2. 7

Structural Considerations Monomer Dimer Paa or Pbb (u 47. 04660(3) 47. 07653(5) Å2) Pcc (u Å2) 3. 207326(3 15. 31910(4) ) ma = 0. 924(8) D, mc = 1. 101(21) D, mtotal = 1. 438(17) D • CS 2 must lie in the plane bisecting the COC angle of the monomer
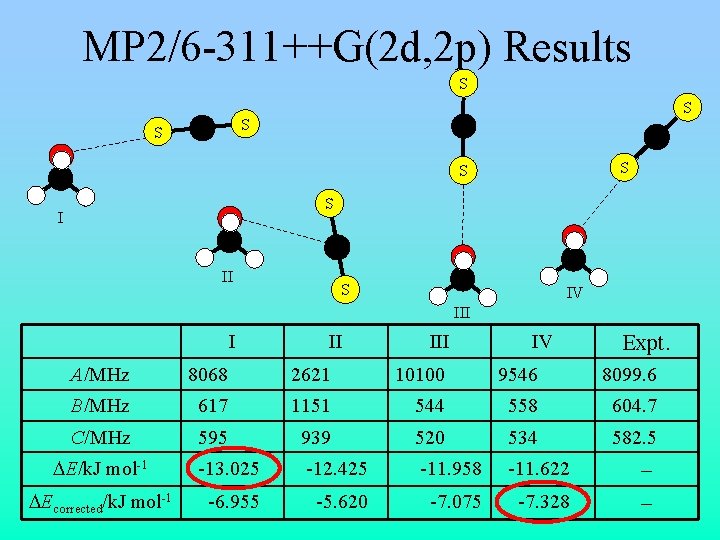
MP 2/6 -311++G(2 d, 2 p) Results S S C C C S S S I C II S IV III IV Expt. A/MHz 8068 2621 10100 9546 8099. 6 B/MHz 617 1151 544 558 604. 7 C/MHz 595 939 520 534 582. 5 DE/k. J mol-1 -13. 025 -12. 425 -11. 958 -11. 622 – DEcorrected/k. J mol-1 -6. 955 -5. 620 -7. 075 -7. 328 –
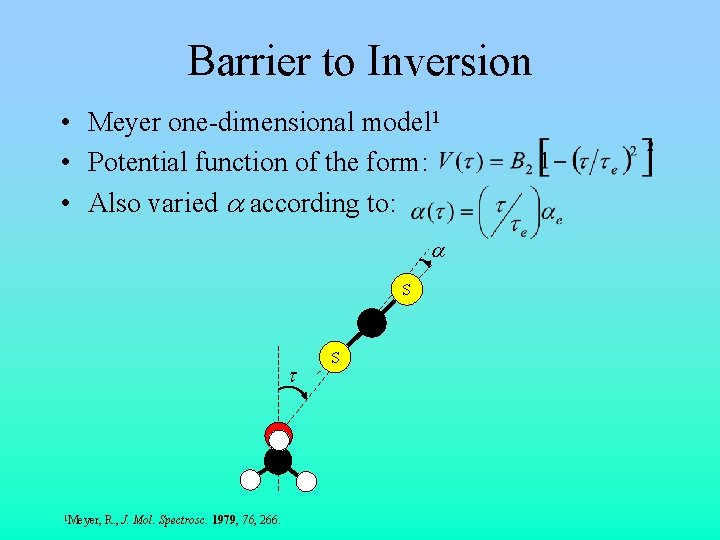
Barrier to Inversion • Meyer one-dimensional model 1 • Potential function of the form: • Also varied a according to: a S C t 1 Meyer, R. , J. Mol. Spectrosc. 1979, 76, 266. S
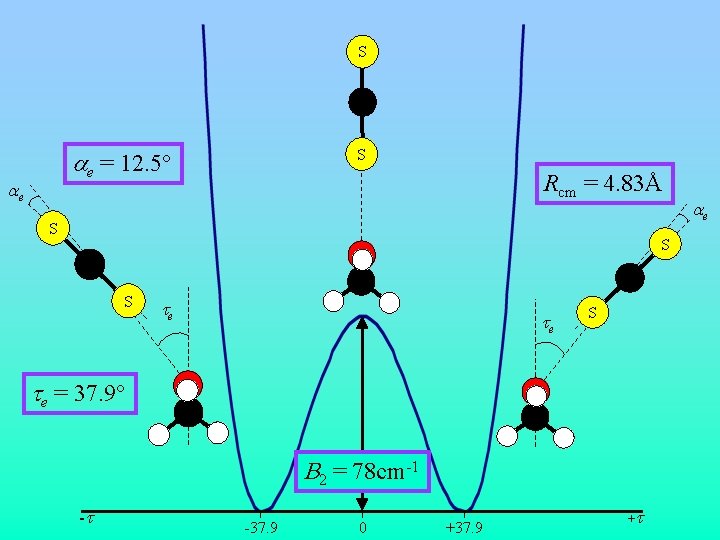
S C ae = 12. 5° ae S Rcm = 4. 83Å S S C C S te te S te = 37. 9° B 2 = 78 cm-1 -t -37. 9 0 +37. 9 +t ae

Intermolecular Interaction • Force constant, ks, for intermolecular bond: • Binding energy, EB, estimated from: • ks = 2. 57(6) N m-1, EB = 5. 0(2) k. J mol-1 – Similar to DME-rare gas complexes
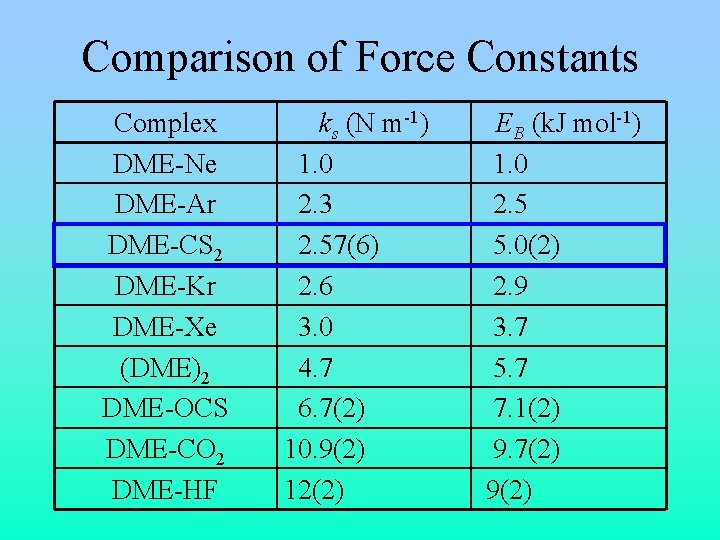
Comparison of Force Constants Complex DME-Ne DME-Ar DME-CS 2 DME-Kr DME-Xe (DME)2 DME-OCS DME-CO 2 DME-HF ks (N m-1) 1. 0 2. 3 2. 57(6) 2. 6 3. 0 4. 7 6. 7(2) 10. 9(2) 12(2) EB (k. J mol-1) 1. 0 2. 5 5. 0(2) 2. 9 3. 7 5. 7 7. 1(2) 9. 7(2) 9(2)

Conclusions • Theory and experiment consistent with Cs symmetry; CS 2 along lone pair of DME • Tunneling of CS 2 between DME lone pairs – Barrier ~78(1) cm-1 – Tunneling frequency 90. 3411(14) MHz • MP 2/6 -311++G(2 d, 2 p) calculations provide useful structural predictions • BSSE and ZPE corrections are essential for determining energy ordering

Acknowledgements • The American Chemical Society Petroleum Research Fund, #39752 -GB 6 • Prof. Walther Caminati • Prof. Robert Kuczkowski • Michal Serafin

Evidence for/against Structure I • Planar moments of I seem to agree better with experiment (but IV is also close) • Dipole moment components of I agree better with experiment • Tunneling pathway and barrier calculation make more sense in light of IV • Both I and IV can be brought into good agreement with experiment via minor structural modifications • I is similar to other linear molecules; IV is similar to noble gases
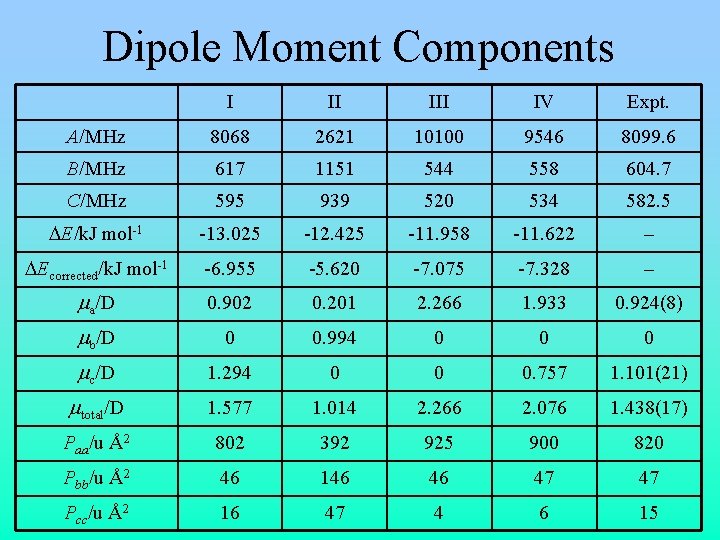
Dipole Moment Components I II IV Expt. A/MHz 8068 2621 10100 9546 8099. 6 B/MHz 617 1151 544 558 604. 7 C/MHz 595 939 520 534 582. 5 DE/k. J mol-1 -13. 025 -12. 425 -11. 958 -11. 622 – DEcorrected/k. J mol-1 -6. 955 -5. 620 -7. 075 -7. 328 – ma/D 0. 902 0. 201 2. 266 1. 933 0. 924(8) mb/D 0 0. 994 0 0 0 mc/D 1. 294 0 0 0. 757 1. 101(21) mtotal/D 1. 577 1. 014 2. 266 2. 076 1. 438(17) Paa/u Å2 802 392 925 900 820 Pbb/u Å2 46 146 46 47 47 Pcc/u Å2 16 47 4 6 15
- Slides: 17