INTRODUCTORY CHEMISTRY Concepts Connections Fifth Edition by Charles

INTRODUCTORY CHEMISTRY Concepts & Connections Fifth Edition by Charles H. Corwin Chapter 4 Matter and Energy Christopher G. Hamaker, Illinois State University, Normal IL © 2008, Prentice Hall

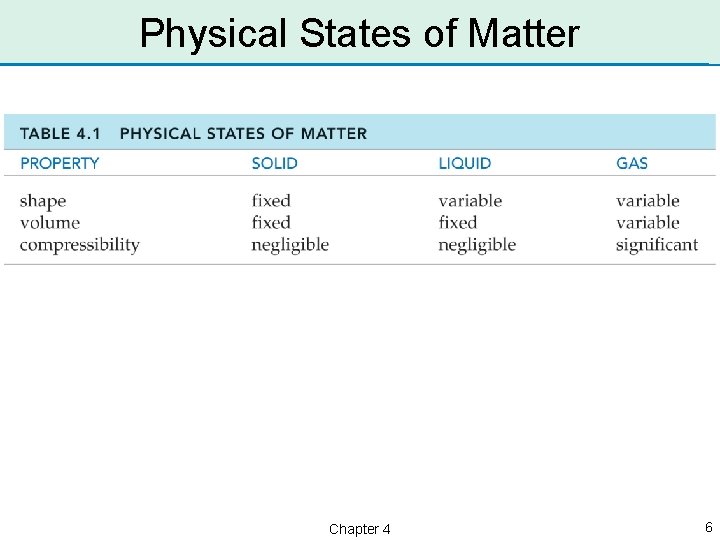
Matter • Matter is any substance that has mass and occupies volume. • Matter exists in one of three physical states: – solid – liquid – gas Chapter 4 2

Gaseous State • In a gas, the particles of matter are far apart and uniformly distributed throughout the container. • Gases have an indefinite shape and assume the shape of their container. • Gases can be compressed and have an indefinite volume. • Gases have the most energy of the three states of matter. Chapter 4 3

Liquid State • In a liquid, the particles of matter are loosely packed and are free to move past one another. • Liquids have an indefinite shape and assume the shape of their container. • Liquids cannot be compressed and have a definite volume. • Liquids have less energy than gases but more energy than solids. Chapter 4 4

Solid State • In a solid, the particles of matter are tightly packed together. • Solids have a definite, fixed shape. • Solids cannot be compressed and have a definite volume. • Solids have the least energy of the three states of matter. Chapter 4 5
Physical States of Matter Chapter 4 6

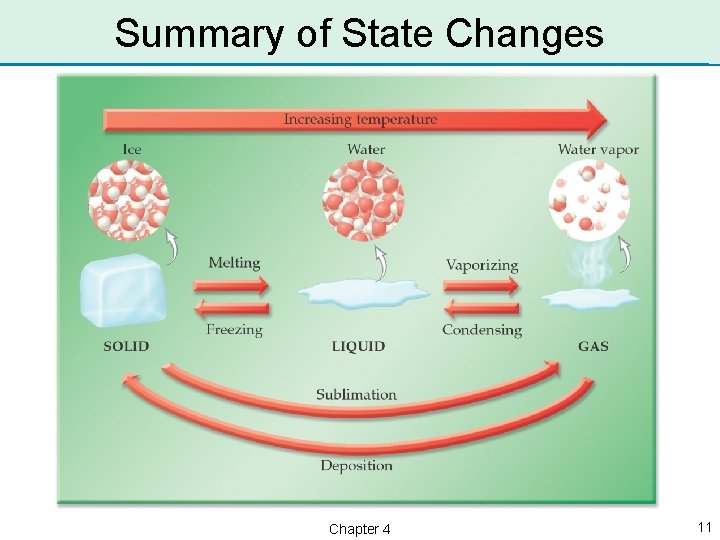
Changes in Physical State • Most substances can exist as either a solid, liquid, or gas. • Water exists as a solid below 0 °C; as a liquid between 0 °C and 100 °C; and as a gas above 100 °C. • A substance can change physical states as the temperature changes. Chapter 4 7

Solid ↔ Liquid Phase Changes • When a solid changes to a liquid, the phase change is called melting. • A substance melts as the temperature increases. • When a liquid changes to a solid, the phase change is called freezing. • A substance freezes as the temperature decreases. Chapter 4 8

Liquid ↔ Gas Phase Changes • When a liquid changes to a gas, the phase change is called vaporization. • A substance vaporizes as the temperature increases. • When a gas changes to a the phase change is condensation. called liquid, • A substance condenses as the temperature decreases. Chapter 4 9

Solid ↔ Gas Phase Changes • When a solid changes directly to a gas, the phase change is called sublimation. • A substance sublimes as the temperature increases. • When a gas changes directly to a solid, the phase change is deposition. • A substance undergoes as the Chapter 4 called deposition temperature decreases. 10
Summary of State Changes Chapter 4 11

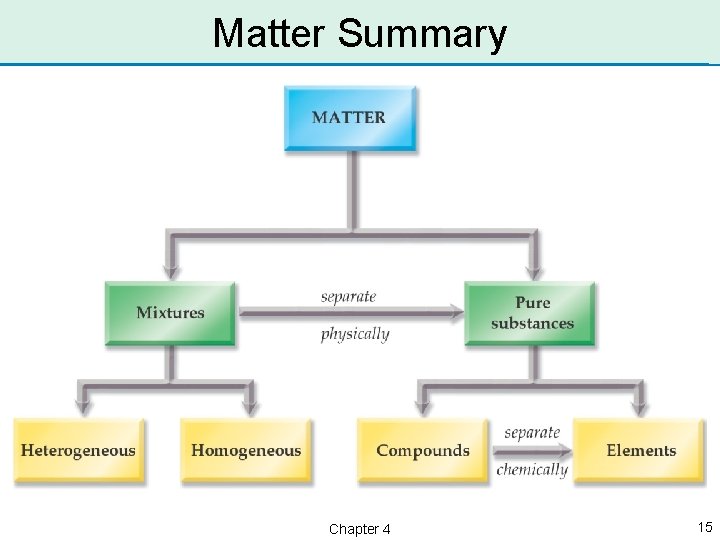
Classifications of Matter • Matter can be divided into two classes: – mixtures – pure substances • Mixtures are composed of more than one substance and can be physically separated into its component substances. • Pure substances are composed of only one substance and cannot be physically separated. Chapter 4 12

Mixtures • There are two types of mixtures: – homogeneous mixtures – heterogeneous mixtures • Homogeneous mixtures have uniform properties throughout. – Salt water is a homogeneous mixture. • Heterogeneous mixtures do not have uniform properties throughout. – Sand water is a heterogeneous mixture. Chapter 4 13

Pure Substances • There are two types of pure substances: – compounds – elements • Compounds can be chemically separated into individual elements. – Water is a compound that can be separated into hydrogen and oxygen. • An element cannot be broken down further by chemical reactions. Chapter 4 14
Matter Summary Chapter 4 15

Occurrence of the Elements • There are over 100 elements that occur in nature; 81 of those elements are stable. • Only 10 elements account for 95% of the mass of the Earth’s crust: Chapter 4 16

Elements In the Human Body • Oxygen is the most common element in both the Earth’s crust and in the human body. • While silicon is the second most abundant element in the crust, carbon is the second most abundant in the body. Chapter 4 17

Names of the Elements • Each element has a unique name. • Names have several origins: – hydrogen is derived from Greek – carbon is derived from Latin – scandium is named for Scandinavia – nobelium is named for Alfred Nobel – yttrium is named for the town of Ytterby, Sweden Chapter 4 18

Element Symbols • Each element is abbreviated using a chemical symbol. • The symbols are 1 or 2 letters long. • Most of the time, the symbol is derived from the name of the element. – C is the symbol for carbon – Cd is the symbol for cadmium • When a symbol has two letters, the first is capitalized and the second is lowercase. Chapter 4 19

Other Element Symbols • For some elements, the chemical symbol is derived from the original Latin name. Gold – Au Sodium – Na Silver – Ag Antimony – Sb Copper – Cu Tin – Sn Mercury – Hg Iron – Fe Potassium – K Tungsten – W Chapter 4 20

Types of Elements • Elements can be divided into three classes: – metals – nonmetals – semimetals or metalloids • Semimetals have properties midway between those of metals and nonmetals. Chapter 4 21

Metal Properties • Metals are typically solids with high melting points and high densities and have a bright, metallic luster. • Metals are good conductors of heat and electricity. • Metals can be hammered into thin sheets and are said to be malleable. • Metals can be drawn into fine wires and are said to be ductile. Chapter 4 22

Nonmetal Properties • Nonmetals typically have low melting points and low densities and have a dull appearance. • Nonmetals are poor conductors of heat and electricity. • Nonmetals are not malleable or ductile and crush into a powder when hammered. • 11 nonmetals occur naturally in the gaseous state. Chapter 4 23

Summary of Properties Chapter 4 24

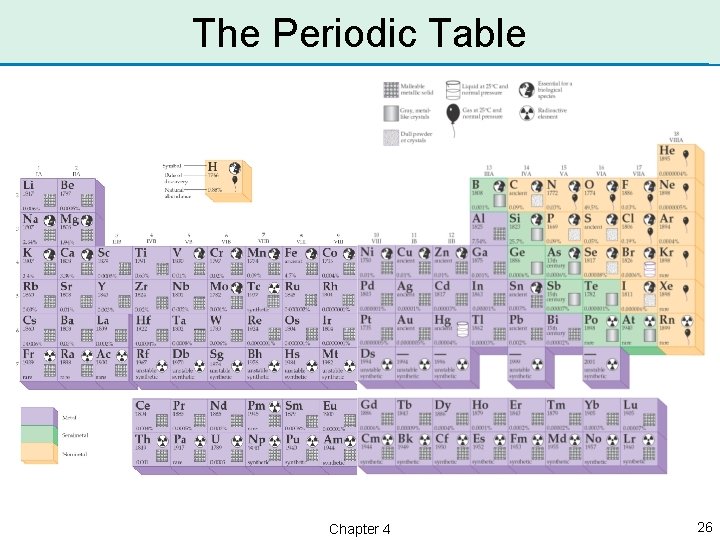
Periodic Table of the Elements • Each element is assigned a number to identify it. It is called the atomic number. • Hydrogen is 1; helium is 2; up to uranium, which is 92. • The elements are arranged by atomic number on the periodic table. Chapter 4 25
The Periodic Table Chapter 4 26
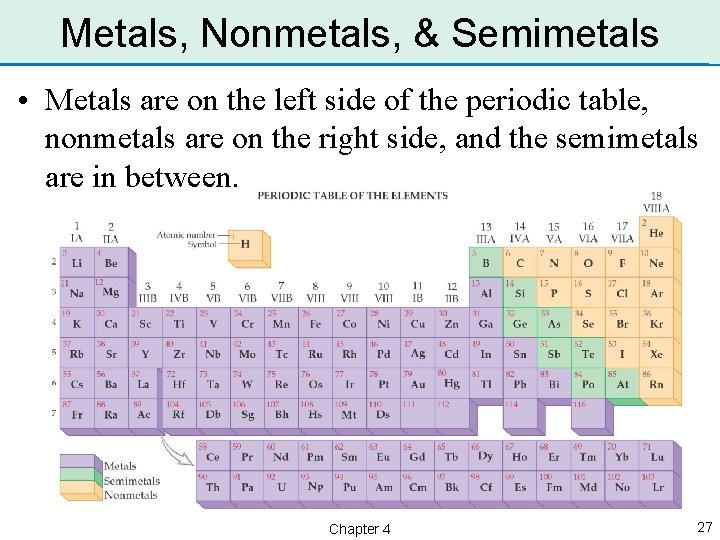
Metals, Nonmetals, & Semimetals • Metals are on the left side of the periodic table, nonmetals are on the right side, and the semimetals are in between. Chapter 4 27
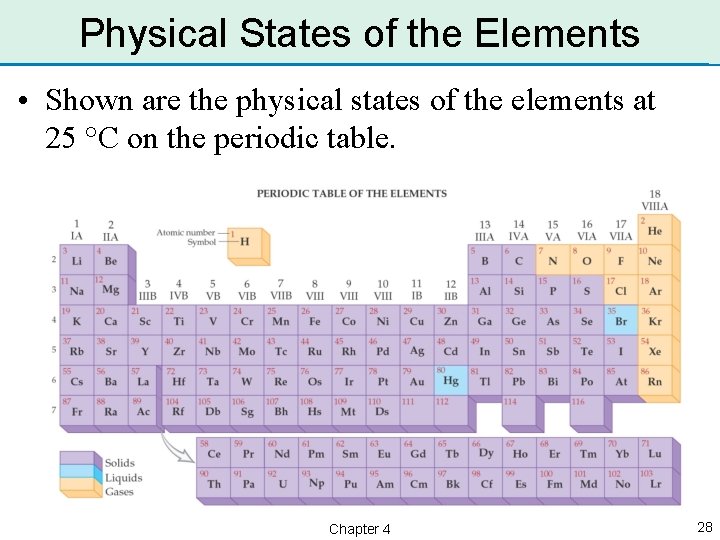
Physical States of the Elements • Shown are the physical states of the elements at 25 °C on the periodic table. Chapter 4 28

Law of Definite Composition • The law of definite composition states that “Compounds always contain the same elements in a constant proportion by mass. ” • Water is always 11. 19% hydrogen and 88. 81% oxygen by mass, no matter what its source. • Ethanol is always 13. 13% hydrogen, 52. 14% carbon, and 34. 73% oxygen by mass. Chapter 4 29
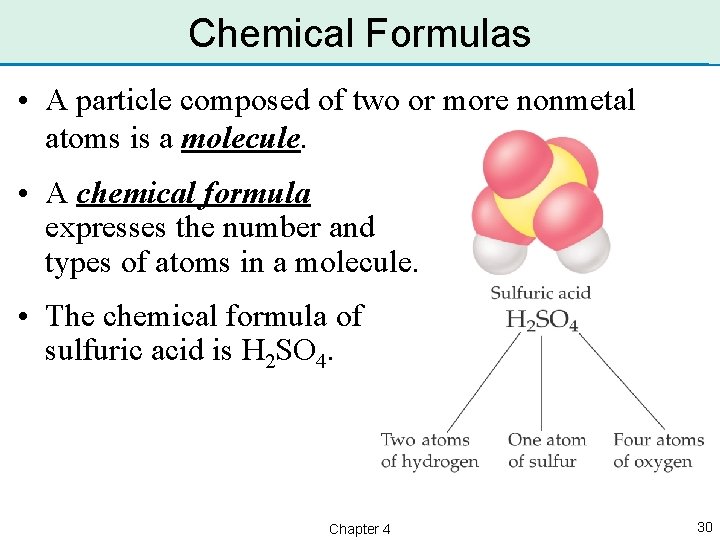
Chemical Formulas • A particle composed of two or more nonmetal atoms is a molecule. • A chemical formula expresses the number and types of atoms in a molecule. • The chemical formula of sulfuric acid is H 2 SO 4. Chapter 4 30

Writing Chemical Formulas • The number of each type of atom in a molecule is indicated with a subscript in a chemical formula. • If there is only one atom of a certain type, no ‘ 1’ is used. • A molecule of the vitamin niacin has 6 carbon atoms, 6 hydrogen atoms, 2 nitrogen atoms, and 1 oxygen atom. What is the chemical formula? C 6 H 6 N 2 O Chapter 4 31

Interpreting Chemical Formulas • Some chemical formulas use parentheses to clarify atomic composition. • Ethylene glycol, a component of some antifreezes, has a chemical formula of C 2 H 4(OH)2. There are 2 carbon atoms, 4 hydrogen atoms, and 2 OH units, giving a total of 6 hydrogen atoms and 2 oxygen atoms. How many total atoms are in ethylene glycol? • Ethylene glycol has a total of 10 atoms. Chapter 4 32

Physical & Chemical Properties • A physical property is a characteristic of a pure substance that we can observe without changing its composition. • Physical properties include appearance, melting and boiling points, density, conductivity, and physical state. • A chemical property describes the chemical reactions of a pure substance. Chapter 4 33

Physical & Chemical Change • A physical change is a change where the chemical composition of the substance is not changed. • These include changes in physical state or shape of a pure substance. • A chemical change is a chemical reaction. • The composition of the substances changes during a chemical change. Chapter 4 34

Evidence for Chemical Changes • gas release (bubbles) • light or release of heat energy • formation of a precipitate • a permanent color change Chapter 4 35

Conservation of Mass • Antoine Lavoisier found that the mass of substances before a chemical change was always equal to the mass of substances after a chemical change. • This is the law of conservation of mass. • Matter is neither created nor destroyed in physical or chemical processes. Chapter 4 36

Conservation of Mass Example • If 1. 0 gram of hydrogen combines with 8. 0 grams of oxygen, 9. 0 grams of water is produced. • Consequently, 3. 0 grams of hydrogen combine with 24. 0 grams of oxygen to produce 27. 0 grams of water. • If 50. 0 grams of water decompose to produce 45. 0 grams of oxygen, how many grams of hydrogen are produced? 50. 0 g water – 45. 0 g oxygen = 5. 0 g hydrogen Chapter 4 37

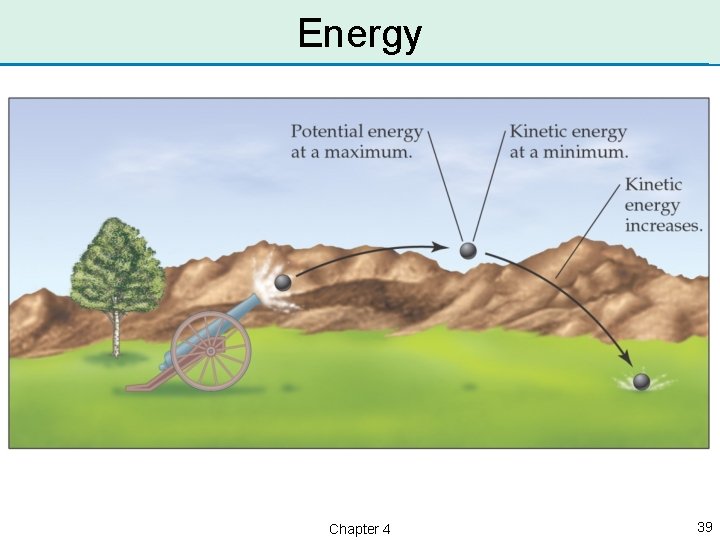
Potential & Kinetic Energy • Potential energy, PE, is stored energy; it results from position or composition. • Kinetic energy, KE, is the energy matter has as a result of motion. • Energy can be converted between the two types. • A boulder at the top of the hill has potential energy; if you push it down the hill, the potential energy is converted to kinetic energy. Chapter 4 38
Energy Chapter 4 39

KE, Temperature, and Physical State • All substances have kinetic energy no matter what physical state they are in. • Solids have the lowest kinetic energy, and gases have the greatest kinetic energy. • As you increase the temperature of a substance, its kinetic energy increases. Chapter 4 40

Law of Conservation of Energy • Just like matter, energy cannot be created or destroyed, but it can be converted from one form to another. • This is the law of conservation of energy. • There are six forms of energy: heat, light, electrical, mechanical, chemical, and nuclear. Chapter 4 41
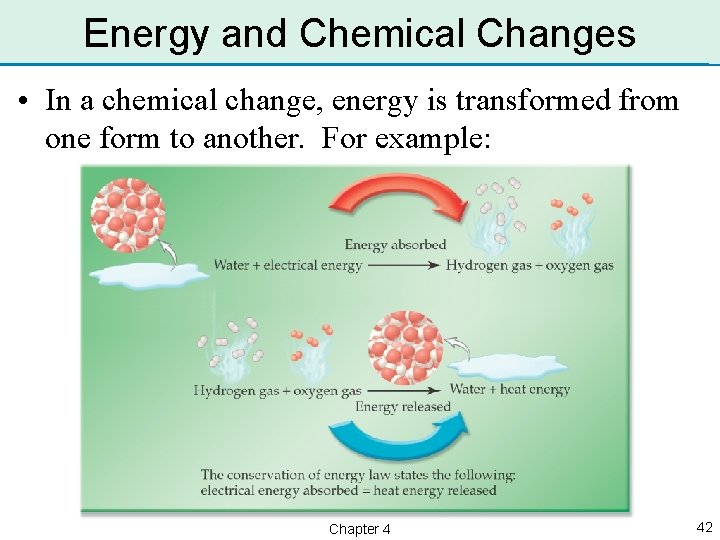
Energy and Chemical Changes • In a chemical change, energy is transformed from one form to another. For example: Chapter 4 42

Law of Conservation of Mass & Energy • Mass and energy are related by Einstein’s theory of relativity, E = mc 2. • Mass and energy can be interchanged. • The law of conservation of and energy states that mass and energy of is constant. Chapter 4 mass the total the universe 43
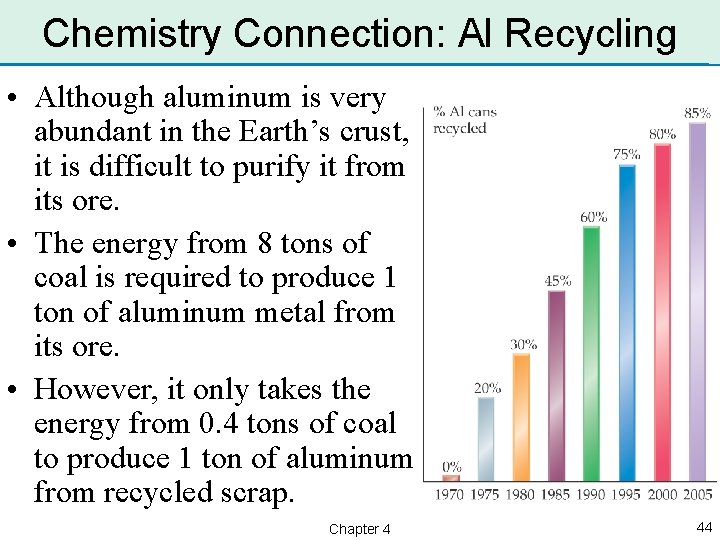
Chemistry Connection: Al Recycling • Although aluminum is very abundant in the Earth’s crust, it is difficult to purify it from its ore. • The energy from 8 tons of coal is required to produce 1 ton of aluminum metal from its ore. • However, it only takes the energy from 0. 4 tons of coal to produce 1 ton of aluminum from recycled scrap. Chapter 4 44

Chapter Summary • Matter exists in three physical states: – solid – liquid – gas • Substances can be converted between the three states. • Substances can be mixtures or pure substances. Chapter 4 45

Chapter Summary, continued • Pure substances can be either compound or elements. • The elements are arranged in the periodic table. • Each element has a name and a 1 - or 2 -letter symbol. • Elements are classified as either metals, nonmetals, or semimetals. Chapter 4 46

Chapter Summary, continued • A physical change is a change in physical state or shape. • A chemical change is a change in the chemical composition of a substance. • Both mass and energy are conserved in chemical and physical changes. Chapter 4 47
- Slides: 47