INTRODUCTORY CHEMISTRY Concepts Connections Fifth Edition by Charles

INTRODUCTORY CHEMISTRY Concepts & Connections Fifth Edition by Charles H. Corwin Chapter 20 Biochemistry Christopher G. Hamaker, Illinois State University, Normal IL © 2008, Prentice Hall

Introduction to Biochemistry • There are 30 elements that are essential for life. • The study of the chemistry of living things is biochemistry. • Biological compounds are often large and complex with molar masses greater than 1, 000 g/mol. • These large molecules are polymers of smaller molecules. Chapter 20 2
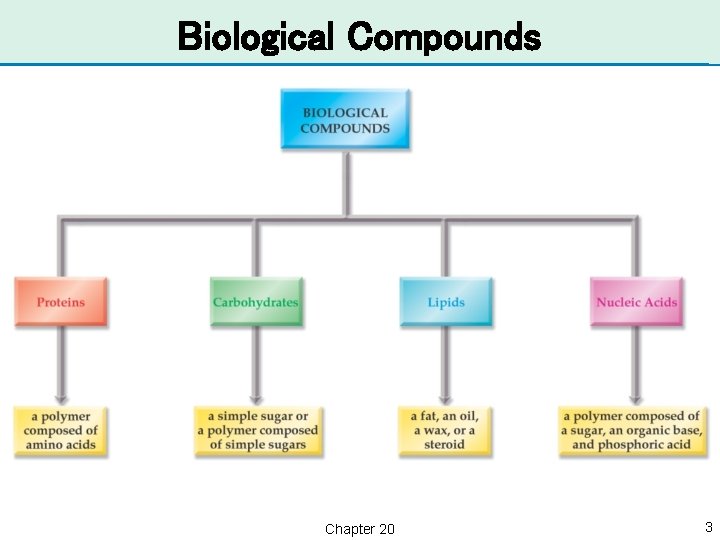
Biological Compounds Chapter 20 3
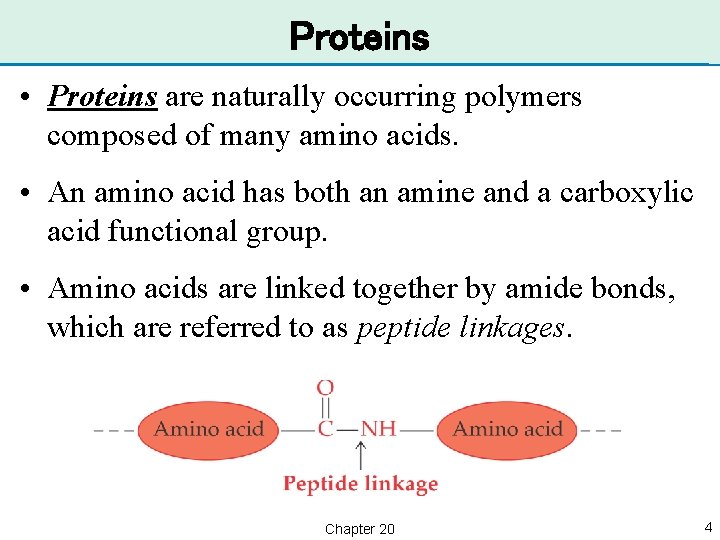
Proteins • Proteins are naturally occurring polymers composed of many amino acids. • An amino acid has both an amine and a carboxylic acid functional group. • Amino acids are linked together by amide bonds, which are referred to as peptide linkages. Chapter 20 4

Sources of Protein • Meat, fish, poultry, cheese, milk products, and many beans are rich in protein. Chapter 20 5
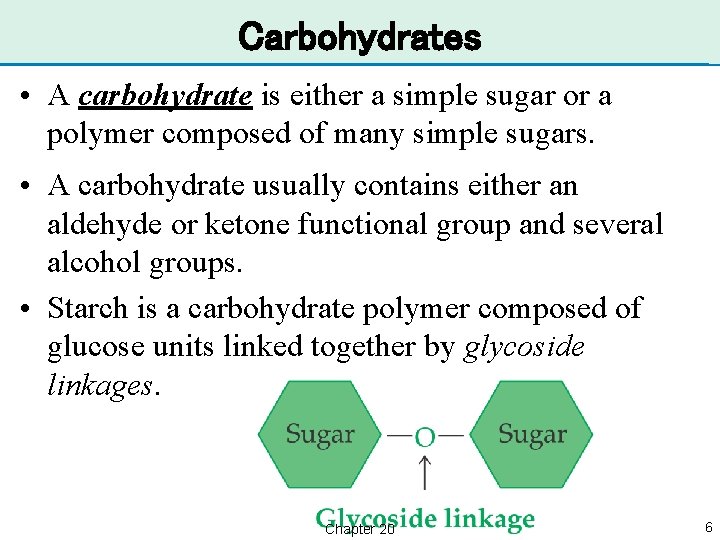
Carbohydrates • A carbohydrate is either a simple sugar or a polymer composed of many simple sugars. • A carbohydrate usually contains either an aldehyde or ketone functional group and several alcohol groups. • Starch is a carbohydrate polymer composed of glucose units linked together by glycoside linkages. Chapter 20 6

Lipids • A lipid is a water-insoluble compound usually composed of an alcohol and one or more carboxylic acid molecules. • Fats and oils are esters of glycerol, an alcohol that has three –OH groups. • As a result, each molecule of a fat or an oil contains three ester groups from three carboxylic acid molecules joined to one glycerol molecule. Chapter 20 7
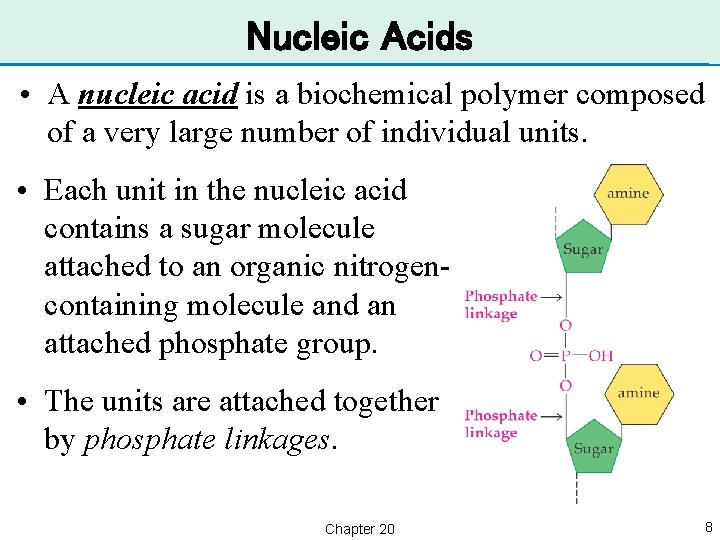
Nucleic Acids • A nucleic acid is a biochemical polymer composed of a very large number of individual units. • Each unit in the nucleic acid contains a sugar molecule attached to an organic nitrogencontaining molecule and an attached phosphate group. • The units are attached together by phosphate linkages. Chapter 20 8

Amino Acids • Proteins are composed of amino acids. • An amino acid has an amine (-NH 2) and a carboxy group (-COOH) attached to a carbon atom with a side chain (R) attached to the a-carbon. • There are 20 naturally occurring amino acids in human proteins. Each one has a different side chain (R). Chapter 20 9
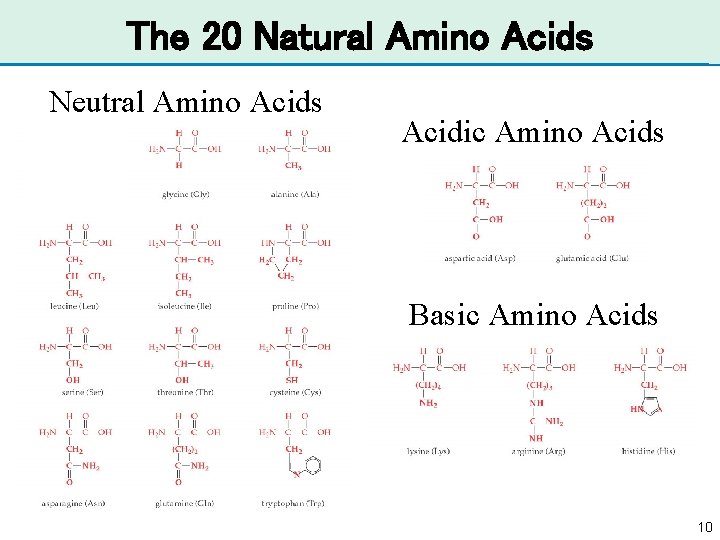
The 20 Natural Amino Acids Neutral Amino Acids Acidic Amino Acids Basic Amino Acids 10
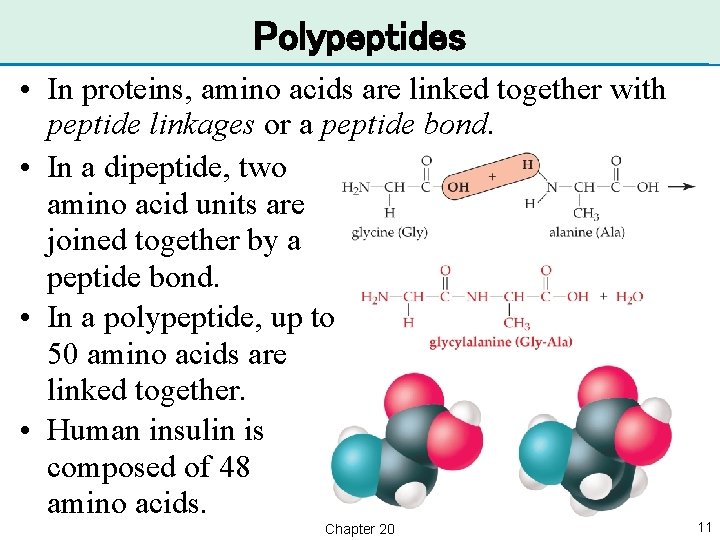
Polypeptides • In proteins, amino acids are linked together with peptide linkages or a peptide bond. • In a dipeptide, two amino acid units are joined together by a peptide bond. • In a polypeptide, up to 50 amino acids are linked together. • Human insulin is composed of 48 amino acids. Chapter 20 11

Primary Structure of Proteins • Proteins are polypeptides composed of hundreds or thousands of amino acid units. • The primary structure of a protein is the sequence of amino acids. Individual amino acids are represented by the ovals in the figure below. • The replacement of one amino acid by another in the primary structure of a protein can completely alter its biological activity. Chapter 20 12
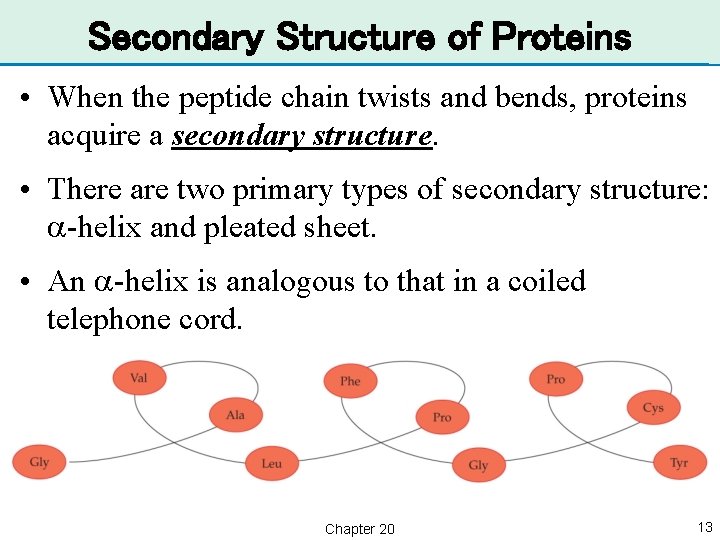
Secondary Structure of Proteins • When the peptide chain twists and bends, proteins acquire a secondary structure. • There are two primary types of secondary structure: a-helix and pleated sheet. • An a-helix is analogous to that in a coiled telephone cord. Chapter 20 13
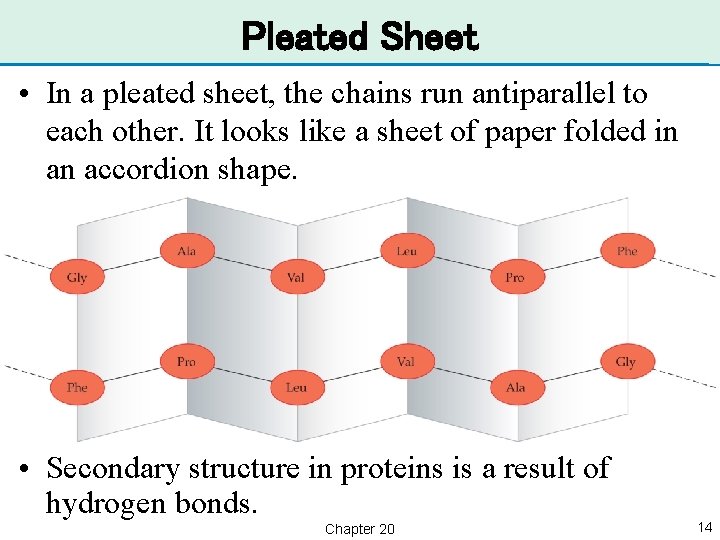
Pleated Sheet • In a pleated sheet, the chains run antiparallel to each other. It looks like a sheet of paper folded in an accordion shape. • Secondary structure in proteins is a result of hydrogen bonds. Chapter 20 14
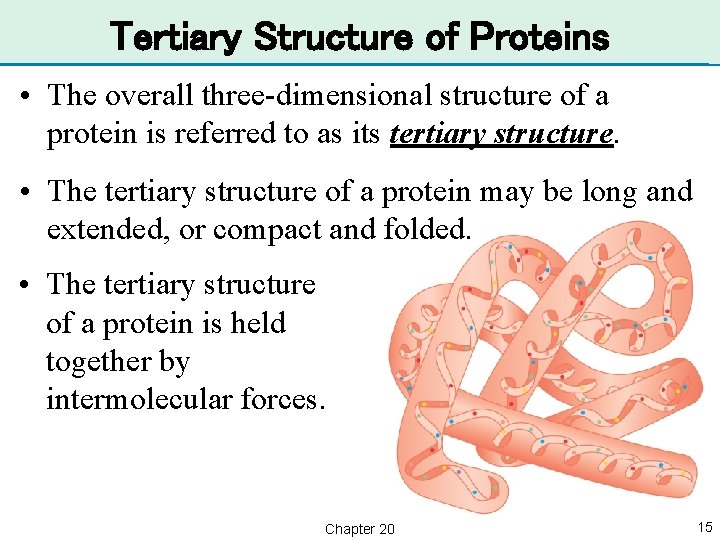
Tertiary Structure of Proteins • The overall three-dimensional structure of a protein is referred to as its tertiary structure. • The tertiary structure of a protein may be long and extended, or compact and folded. • The tertiary structure of a protein is held together by intermolecular forces. Chapter 20 15

Enzymes • An enzyme is a protein that acts as a biological catalyst. • Enzymes are incredibly selective for specific molecules. • An enzyme can speed up a biochemical reaction so that the rate is a million times faster than it would be in the absence of the enzyme. • Many reactions catalyzed by enzymes would be too slow without the enzyme to sustain life. Chapter 20 16
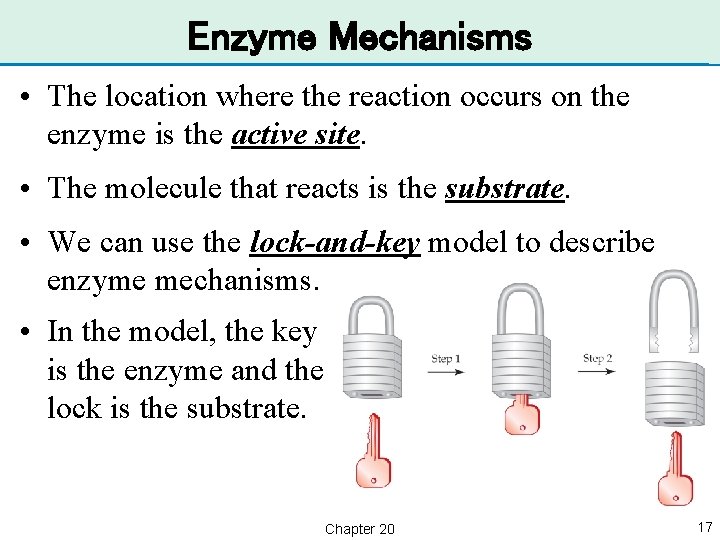
Enzyme Mechanisms • The location where the reaction occurs on the enzyme is the active site. • The molecule that reacts is the substrate. • We can use the lock-and-key model to describe enzyme mechanisms. • In the model, the key is the enzyme and the lock is the substrate. Chapter 20 17

Enzyme Reactions • An enzyme reaction takes place in two steps. • First, the substrate (S) binds to the active site on the enzyme (E). Step 1: E + S → ES • Second, the enzyme releases two or more products (P 1 and P 2). Step 2: ES → E + P 1 + P 2 Chapter 20 18
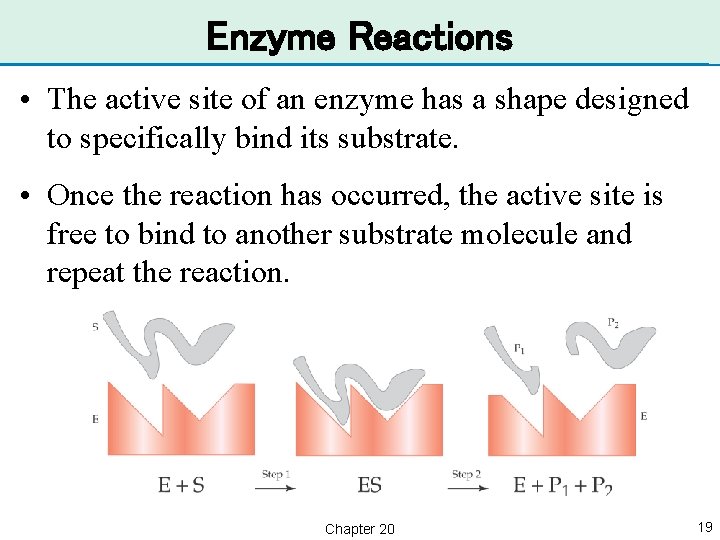
Enzyme Reactions • The active site of an enzyme has a shape designed to specifically bind its substrate. • Once the reaction has occurred, the active site is free to bind to another substrate molecule and repeat the reaction. Chapter 20 19
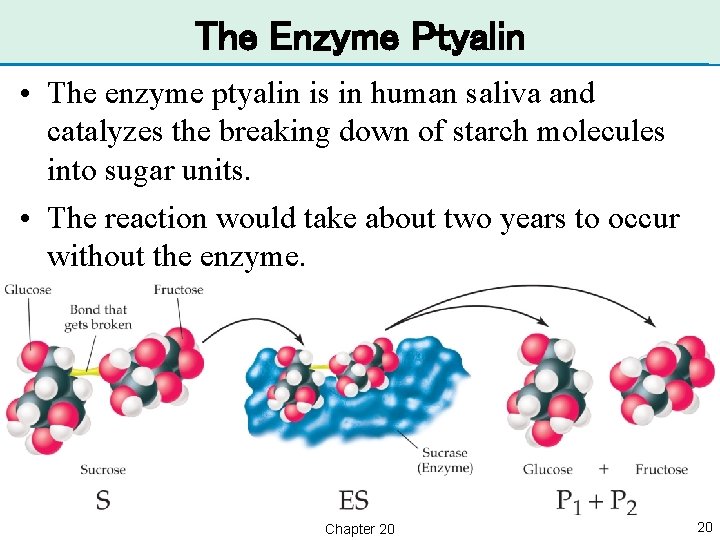
The Enzyme Ptyalin • The enzyme ptyalin is in human saliva and catalyzes the breaking down of starch molecules into sugar units. • The reaction would take about two years to occur without the enzyme. Chapter 20 20

Carbohydrates • The word carbohydrate means “hydrates of carbon, ” and many have the empirical formula CH 2 O. • Carbohydrate names usually end in the suffix -ose. • Carbohydrates have an aldehyde or ketone functional group and several hydroxyl groups. Chapter 20 21

Simple Sugars • A simple sugar molecule typically has three to six carbons, an aldehyde or ketone group, and a few hydroxyl groups. • A monosaccharide with an aldehyde group is an aldose, and one with a ketone group is a ketose. Chapter 20 22
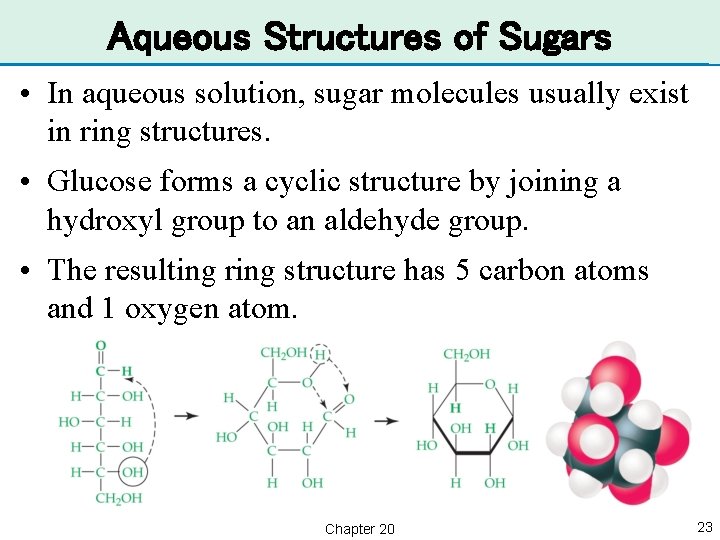
Aqueous Structures of Sugars • In aqueous solution, sugar molecules usually exist in ring structures. • Glucose forms a cyclic structure by joining a hydroxyl group to an aldehyde group. • The resulting ring structure has 5 carbon atoms and 1 oxygen atom. Chapter 20 23
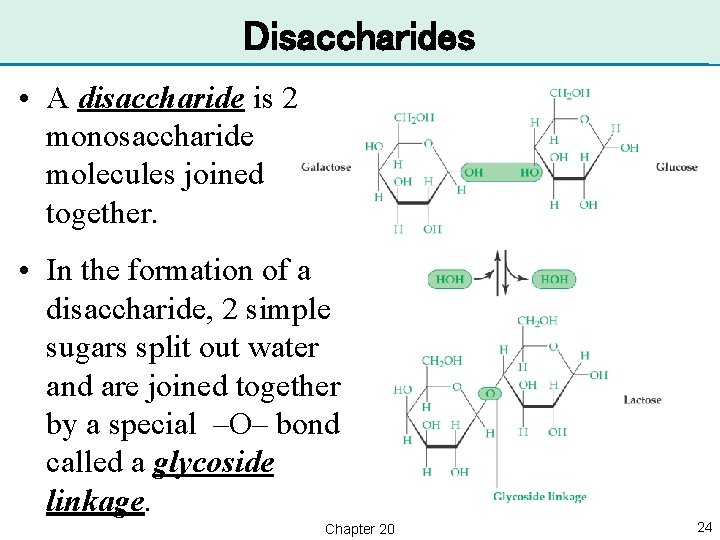
Disaccharides • A disaccharide is 2 monosaccharide molecules joined together. • In the formation of a disaccharide, 2 simple sugars split out water and are joined together by a special –O– bond called a glycoside linkage. Chapter 20 24

Polysaccharides • A polysaccharide is a polymer of many monosaccharide linkages joined by glycoside linkages. • Starch and cellulose are both polysaccharides composed only of glucose units, but with slightly different 3 -dimentional structures. starch cellulose Chapter 20 25

Lipids • Unlike most other biological compounds, lipids are not water soluble. • Lipids include – fats and oils – waxes – steroids – the water insoluble A, D, E, and K vitamins Chapter 20 26

Fatty Acids • A fatty acid is a carboxylic acid with a long hydrocarbon chain. • Fatty acids can be saturated or unsaturated. Chapter 20 27
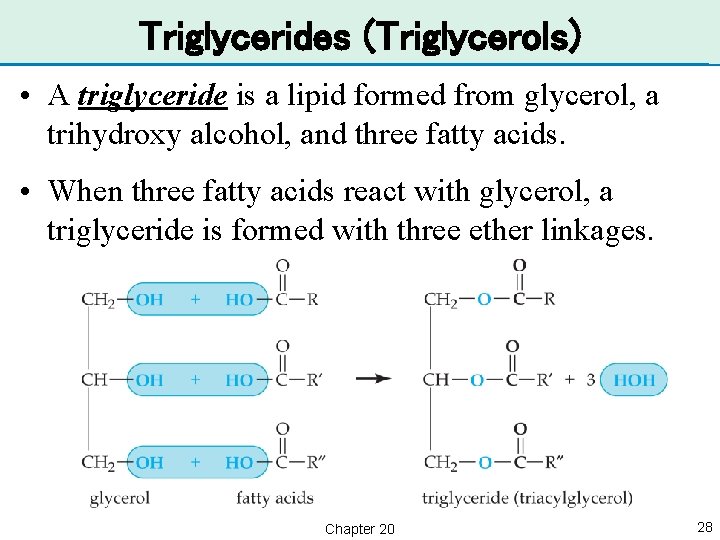
Triglycerides (Triglycerols) • A triglyceride is a lipid formed from glycerol, a trihydroxy alcohol, and three fatty acids. • When three fatty acids react with glycerol, a triglyceride is formed with three ether linkages. Chapter 20 28

Fats • In triglycerides obtained from animals, the fatty acids are mostly saturated and have few double bonds. • A semisolid lipid obtained from an animal source is a fat. • The following structure is an example of a saturated triglyceride from animal fat. Chapter 20 29

Oils • A liquid lipid obtained from a plant source is an oil. • Triglycerides obtained from plants have mostly unsaturated fatty acid chains. • The following structure is an example of an unsaturated triglyceride from a vegetable oil. Chapter 20 30

Saponification of Fats and Oils • The ester linkage in a triglyceride can be broken by treatment with aqueous sodium hydroxide. • The products of the reaction are glycerol and the sodium salt of the fatty acid. • The sodium salts of fatty acids are soaps. Chapter 20 31

Soaps • Soap molecules have a polar “head” (COO-Na+) and a long nonpolar “tail” (R, R’, R” ). • The soap molecules are able to dissolve oil with the tail, and the heads stick into the water. Chapter 20 32

Phospholipids • A phospholipid is a glyceride found in living cells and membranes. • Phospholipids have two fatty acids esterified to the glycerol. • The third –OH is linked to a phosphoric acid. Chapter 20 33
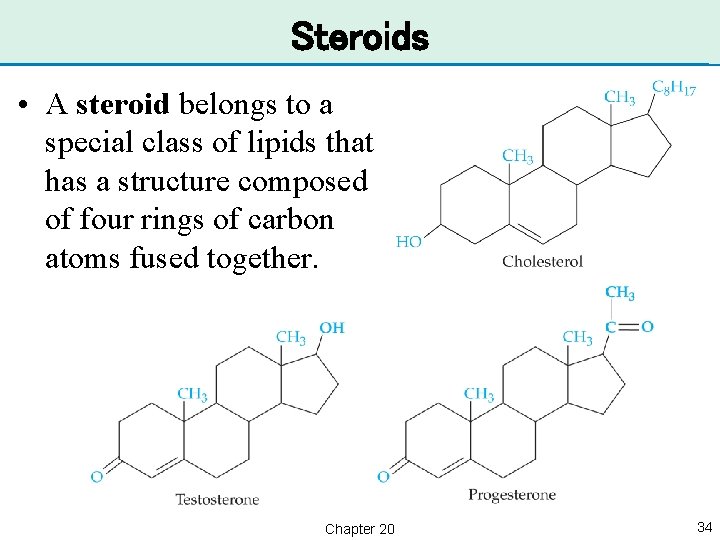
Steroids • A steroid belongs to a special class of lipids that has a structure composed of four rings of carbon atoms fused together. Chapter 20 34

Chemistry Connection: Vitamins • Vitamins are biochemical compounds required for normal metabolism. • Vitamins are not synthesized in the human body, and therefore must be included in the diet. • Excess water-soluble vitamins are easily excreted from the body. • Excess doses of fat-soluble vitamins (A, D, E, and K) accumulate in fatty tissue and can be toxic. Chapter 20 35

Nucleic Acids • Nucleic acids are biochemical compounds found in every living cell. • Nucleic acids contain the genetic information responsible for the reproduction of a species. • There are two basic types of nucleic acids: deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). • A nucleic acid is a polymer composed of many repeating units, each of which is called a nucleotide. Chapter 20 36
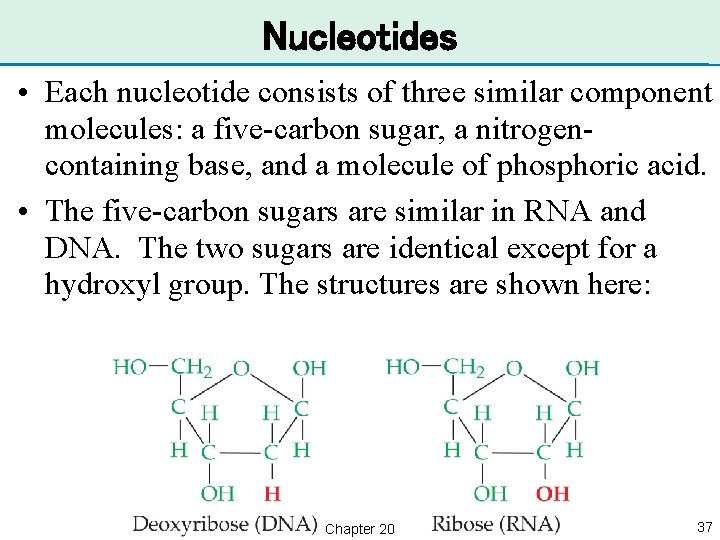
Nucleotides • Each nucleotide consists of three similar component molecules: a five-carbon sugar, a nitrogencontaining base, and a molecule of phosphoric acid. • The five-carbon sugars are similar in RNA and DNA. The two sugars are identical except for a hydroxyl group. The structures are shown here: Chapter 20 37
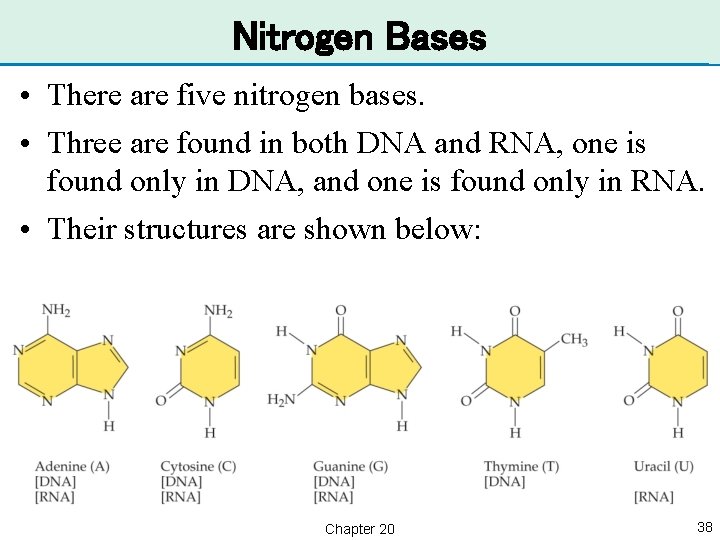
Nitrogen Bases • There are five nitrogen bases. • Three are found in both DNA and RNA, one is found only in DNA, and one is found only in RNA. • Their structures are shown below: Chapter 20 38
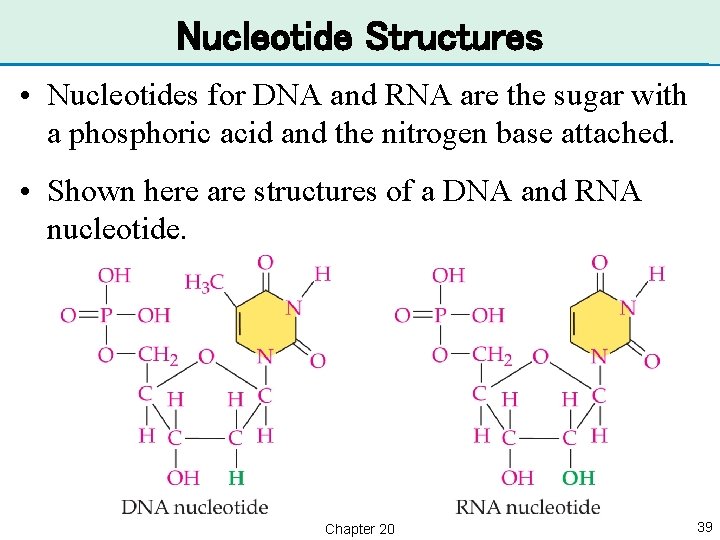
Nucleotide Structures • Nucleotides for DNA and RNA are the sugar with a phosphoric acid and the nitrogen base attached. • Shown here are structures of a DNA and RNA nucleotide. Chapter 20 39
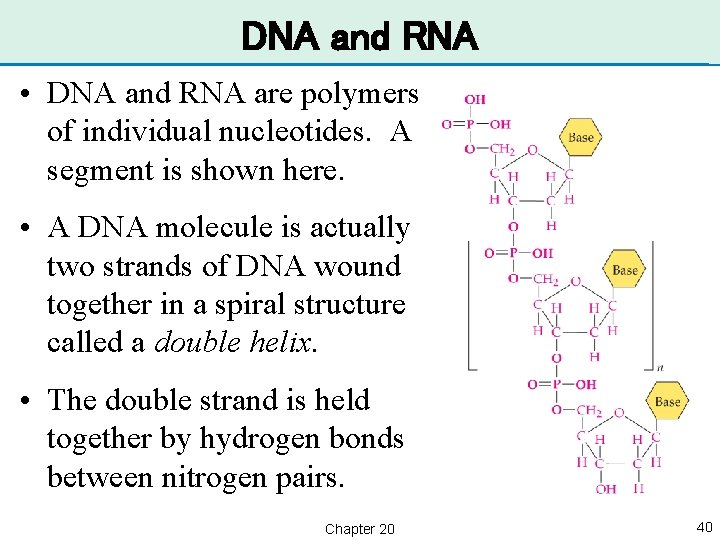
DNA and RNA • DNA and RNA are polymers of individual nucleotides. A segment is shown here. • A DNA molecule is actually two strands of DNA wound together in a spiral structure called a double helix. • The double strand is held together by hydrogen bonds between nitrogen pairs. Chapter 20 40
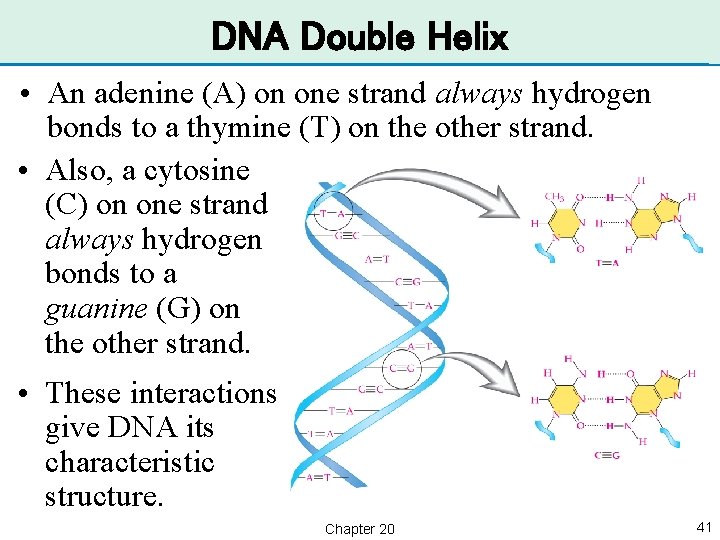
DNA Double Helix • An adenine (A) on one strand always hydrogen bonds to a thymine (T) on the other strand. • Also, a cytosine (C) on one strand always hydrogen bonds to a guanine (G) on the other strand. • These interactions give DNA its characteristic structure. Chapter 20 41

Chapter Summary • There are 4 major types of biological molecules: – Proteins are composed of amino acids. – Carbohydrates are composed of simple sugar molecules. – A lipid is a water-insoluble biological molecule. – A nucleic acid is a polymer composed of a sugar molecule, a nitrogen base, and a phosphoric acid. Chapter 20 42

Chapter Summary, continued • An enzyme is a catalyst for biological reactions. • Enzymes work by a lock-and-key mechanism where only a specific substrate fits into the enzyme to react. • Reactions catalyzed by enzymes can be completed in a matter of seconds where the reaction would normally take many years. Chapter 20 43

Chapter Summary, continued • A triglyceride is an ester of glycerol and three different fatty acids. • A fat is a triglyceride from an animal source. An oil is a triglyceride from a plant source. • A nucleic acid is a molecule that carries genetic information. • DNA and RNA are the two basic types of nucleic acids. Chapter 20 44
- Slides: 44