Introductory Chemistry Concepts Connections 4 th Edition by

Introductory Chemistry: Concepts & Connections 4 th Edition by Charles H. Corwin Chapter 3 The Metric System Christopher G. Hamaker, Illinois State University, Normal IL © 2005, Prentice Hall

The Metric System • The English System was used primarily in the British Empire and wasn’t very standardized. • The French organized a committee to devise a universal measuring system. • After about 10 years, the committee designed and agreed on the metric system. • The metric system offers simplicity with a single base unit for each measurement. Chapter 3 2
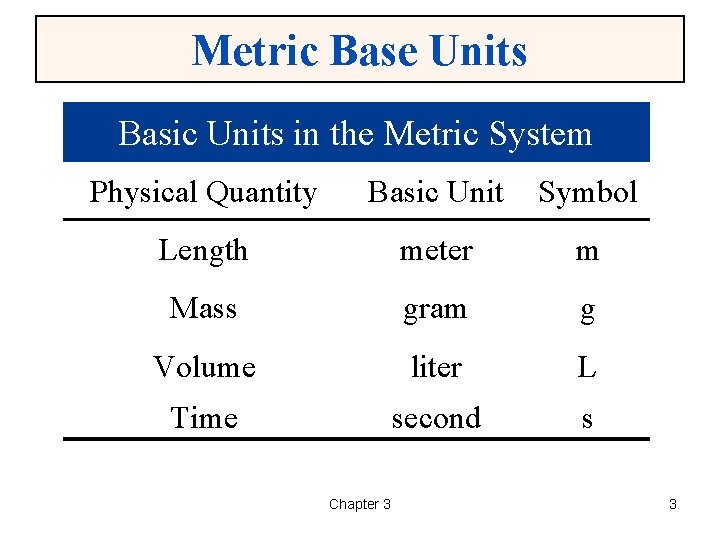
Metric Base Units Basic Units in the Metric System Physical Quantity Basic Unit Symbol Length meter m Mass gram g Volume liter L Time second s Chapter 3 3

Unit Definitions • A meter was defined as 1/10, 000 of the distance from the North Pole to the equator. • A kilogram (1000 grams) was equal to the mass of a cube of water measuring 0. 1 m on each side. • A liter was set equal to the volume of one kilogram of water at 4 C. Chapter 3 4

Metric System Advantage • Another advantage of the metric system is that it is a decimal system. • It uses prefixes to enlarge or reduce the basic units. • For example: – A kilometer is 1000 meters. – A centimeter is 1/100 of a meter. Chapter 3 5

Metric System Prefixes • The following table lists the common prefixes used in the metric system: Chapter 3 6

Metric Prefixes Continued • For example, the prefix kilo- increases a base unit by 1000: – 1 kilogram is 1000 grams. • The prefix centi- decreases a base unit by a factor of 100: – 1 centimeter is 0. 01 meters. Chapter 3 7

Metric Symbols • The names of metric units are abbreviated using symbols. Use the prefix symbol followed by the symbol for the base unit, so: – kilometer is abbreviated km. – milligram is abbreviated mg. – microliter is abbreviated m. L. – nanosecond is abbreviated ns. Chapter 3 8

Metric Equivalents • We can write unit equations for the conversion between different metric units. • The prefix kilo- means 1000 basic units, so 1 kilometer is 1000 meters. • The unit equation is 1 km = 1000 m. • Similarly, a centimeter is 1/100 of a meter, so the unit equation is 1 cm = 0. 01 m. Chapter 3 9

Metric Unit Factors • Since 1000 m = 1 km, we can write the following unit factors for converting between meters and kilometers: 1 km 1000 m or 1000 m 1 km • Since 1 cm = 0. 01 m, we can write the following unit factors. 1 cm 0. 01 m or Chapter 3 0. 01 m 1 cm 10

Metric-Metric Conversions • We will use the unit analysis method we learned in Chapter 2 to do metric-metric conversion problems. • Remember, there are three steps – Write down the unit asked for in the answer – Write down the given value related to the answer – Apply unit factor(s) to convert the given unit to the units desired in the answer. Chapter 3 11

Metric-Metric Conversion Problem • What is the mass in grams of a 325 mg aspirin tablet? • Step 1: We want grams. • Step 2: We write down the given: 325 mg. • Step 3: We apply a unit factor (1 g = 1000 mg) and round to three significant figures. 1. 0 g 325 mg × = 0. 325 g 1000 mg Chapter 3 12

Two Metric-Metric Conversions • A hospital has 125 deciliter bags of blood plasma. What is the volume in milliliters? • Step 1: we want the answer in m. L • Step 2: we have 125 d. L. • Step 3: we need to first convert d. L to L and then convert L to m. L: 1. 0 L 10 d. L and Chapter 3 1000 m. L 1 L. 13

Problem Continued • Apply both unit factors, and round the answer to 3 significant digits. • Notice that both d. L and L units cancel, leaving us with units of m. L. 1000 m. L 1. 0 L 125 d. L × × = 12, 500 m. L 1. 0 L 10 d. L Chapter 3 14

Another Example • The mass of the Earth is 5. 98 × 1024 kg. What is the mass expressed in megagrams, Mg? • We want Mg; we have 5. 98 × 1024 kg • Convert kilograms to grams, and then grams to megagrams. 5. 98 × 1024 1000 g 1 Mg kg × × = 5. 98 × 1021 Mg 1000000 g 1 kg Chapter 3 15

Metric and English Units • The English system is still very common in the United States. • We often have to convert between English and Metric Units. Chapter 3 16

Metric-English Conversion • Which distance is longer, 100 meters or 100 yards? • Lets convert 100. 0 m to 100 yards given that 1 yd = 0. 914 m. • 100 meters is 109 yards, so 100 yards is shorter. 1 yd 100. 0 m × = 109 yd 0. 914 m Chapter 3 17

English-Metric Conversion • A half gallon carton contains 64. 0 fl oz of milk. How many milliliters of milk are in a carton? • We want m. L, we have 64. 0 fl oz. • Use 1 qt = 32 fl oz, and 1 qt = 946 m. L. 1 qt 946 m. L 64. 0 fl oz × × = 1, 890 m. L 32 fl oz 1 qt Chapter 3 18

Compound Units • Some measurements have a ratio of units. • For example, the speed limit on many highways is 55 miles per hour. How would you convert to this to meters per second? • Convert one unit at a time using unit factors. Chapter 3 19

Compound Unit Problem • A Corvette is traveling at 95 km/hour. What is the speed in meters per second? • We have km/h, we want m/s. • Use 1 km = 1000 m and 1 h = 3600 s. 95 km 1 hr 1000 m × × = 26 m/s 1 km hr 3600 s Chapter 3 20

Volume by Calculation • The volume of an object is calculated by multiplying the length (l) by the width (w) by the thickness (t). volume = l × w × t • All three measurements must be in the same units. • If an object measures 3 cm by 2 cm by 1 cm, the volume is 6 cm 3 (cm 3 is cubic centimeters). Chapter 3 21
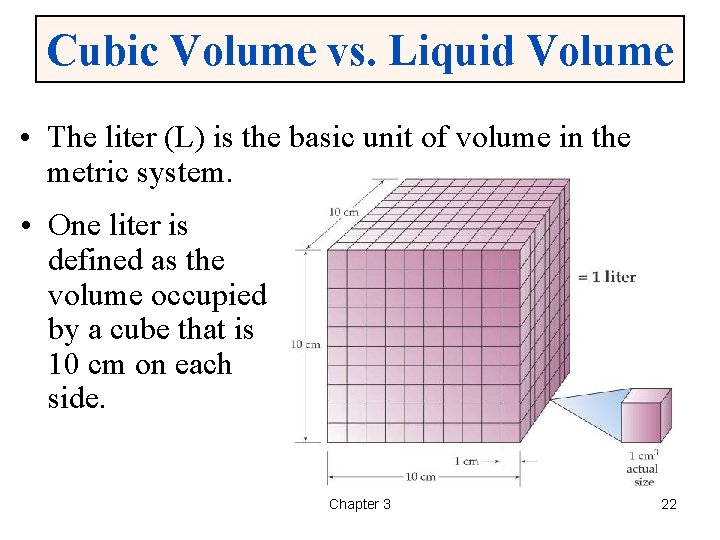
Cubic Volume vs. Liquid Volume • The liter (L) is the basic unit of volume in the metric system. • One liter is defined as the volume occupied by a cube that is 10 cm on each side. Chapter 3 22

Cubic & Liquid Volume Units • 1 liter is equal to 1000 cubic centimeters – 10 cm × 10 cm = 1000 cm 3 • 1000 cm 3 = 1 L = 1000 m. L. • Therefore, 1 cm 3 = 1 m. L. Chapter 3 23

Conclusions Continued • A Volkswagen Beetle engine displaces a volume of 498 cm 3 in each cylinder. What is the displacement in cubic inches, in 3? • We want in 3, we have 498 cm 3. • Use 1 in = 2. 54 cm three times. 498 cm 3 1 in × × × = 30. 4 in 3 2. 54 cm Chapter 3 24

Volume by Displacement • If a solid has an irregular shape, its volume cannot be determined by measuring its dimensions. • You can determine its volume indirectly by measuring the amount of water it displaces. • This technique is call volume by displacement. Chapter 3 25

Volume by Displacement • You want to measure the volume of an irregularly shaped piece of jade. • Partially fill a volumetric flask with water and measure the volume of the water. • Add the jade, and measure the difference in volume. • The volume of the jade is 10. 5 m. L. Chapter 3 26

The Density Concept • The density of an object is a measure of its concentration of mass • Density is defined as the mass of an object divided by the volume of the object. mass = density volume Chapter 3 27

Density • Density is expressed in different units. It is usually grams per milliliter (g/m. L) for liquids, grams per cubic centimeter (g/cm 3) for solids, and grams per liter (g/L) for gases. Chapter 3 28

Estimating Density • We can estimate the density of a substance by comparing it to another object. • A solid object will float on top a liquid with a higher density. • Object S 1 has a density less than that of water, but larger than that of L 1. • Object S 2 has a density less than that of L 2, but larger than that of water. Chapter 3 29

Calculating Density • What is the density of a platinum nugget has a mass of 224. 50 g and a volume of 10. 0 cm 3 ? • Recall, density is mass/volume. 224. 50 g 3 = 22. 5 g/cm 10. 0 cm 3 Chapter 3 30

Density as a Unit Factor • We can use density as a unit factor for conversions between mass and volume. • An automobile battery contains 1275 m. L of acid. If the density of battery acid is 1. 84 g/m. L, how many grams of acid are in an automobile battery? • We have 1275 m. L and we want grams: 1. 84 g 1275 m. L × = 2350 g m. L Chapter 3 31

Temperature • Temperature is a measure of the average kinetic energy of the individual particles in a sample. • There are three temperature scales: – Celsius – Fahrenheit – Kelvin • Kelvin is the absolute temperature scale. Chapter 3 32

Temperature Scales • On the Fahrenheit scale, water freezes at 32°F and boils at 212°F. • On the Celsius scale, water freezes at 0°C and boils at 100°C. These are the reference points for the Celsius scale. • Water freezes at 273 K and boils at 373 K on the Kelvin scale. Chapter 3 33

Temperature Conversions • This is the equation for converting °C to °F. °C × ( 180°F 100°C ) = °F • This is the equation for converting °F to °C. ( ) 100°C = °C (°F - 32°F) × 180°F • To convert from °C to K, add 273. °C + 273 = K Chapter 3 34

Fahrenheit-Celsius Conversions • Body temperature is 98. 6°F. What is body temperature in Celsius? (98. 6°F - 32°F) × ( 100°C 180°F Chapter 3 ) = 37. 0°C 35

Heat • Heat is the flow of energy from an object of higher temperature to an object of lower temperature. • Heat measures the total energy of a system while temperature measures the average energy of particles in a system. • Heat is often expressed in terms of joules (J) or calories (cal). Chapter 3 36

Specific Heat • The specific heat of a substance is the amount of heat required to bring about a change in temperature. • It is expressed with units of calories per gram degree Celsius. • The larger the specific heat, the more heat is required to raise the temperature of the substance. Chapter 3 37

Conclusions • The basic units in the metric system are grams for mass, liters for volume, and meters for distance. • The base units are modified using prefixes to reduce or enlarge the base units by factors of 10. • We can use unit factors to convert between metric units. • We can convert between metric and English units using unit factors. Chapter 3 38

Conclusions Continued • Volume is defined as length × width × thickness. • Volume can also be determined by displacement of water. • Density is mass divided by volume. Chapter 3 39

Conclusions Continued • Temperature is a measure of the average energy of the particles in a sample. • Heat is a measure of the total energy of a substance. • Specific heat is a measure of how much heat is required to raise the temperature of a substance. Chapter 3 40

QUIZE - 3 1. What is the mass in grams of a 325 mg aspirin tablet? 2. What is the density of a platinum nugget has a mass of 224. 50 g and a volume of 10. 0 cm 3 ? Chapter 3 41
- Slides: 41