Introductory Biochemistry BIOC Dr Ebtihaj Jambi Room 2125

Introductory Biochemistry BIOC Dr Ebtihaj Jambi Room 2125

Books

The performance in this course will be evaluated in five areas : home works , lab. Exam and two exams • • • Home works……………………. 5% First exam ……………………. . 15 % Second exam …………………. 15 % Lab. Exam…………………. …. 25% Final exam ……………………. 40 %


The important Dates: Section Sat. Mon. 1 The First Exam ………. 22/11/1433 2 The Second Exam…. . . 12/ 1 /1434 3 The home work…. . .

What is Biochemistry? • Biochemistry = chemistry of life. • Biochemists use physical and chemical principles to explain biology at the molecular level. • Basic principles of biochemistry are common to all living organism

How does biochemistry impact you? • Medicine • Agriculture • Industrial applications • Environmental applications
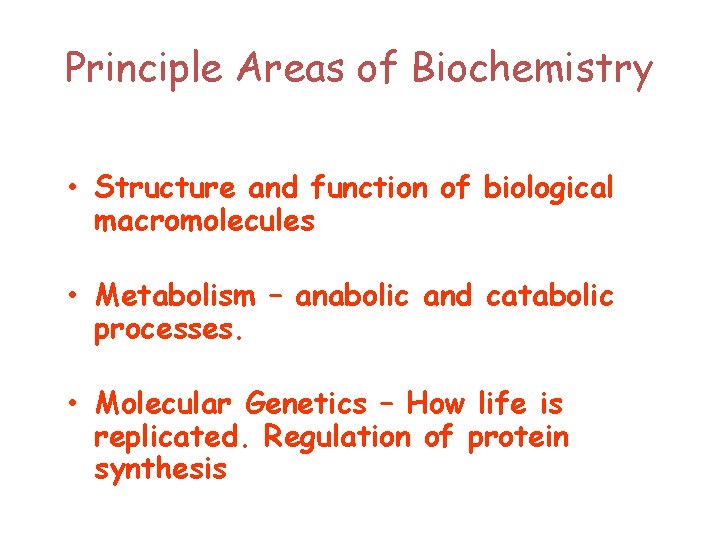
Principle Areas of Biochemistry • Structure and function of biological macromolecules • Metabolism – anabolic and catabolic processes. • Molecular Genetics – How life is replicated. Regulation of protein synthesis

Organization of Life • elements • simple organic compounds (monomers) • macromolecules (polymers) • supramolecular structures • organelles • cells • tissues • organisms
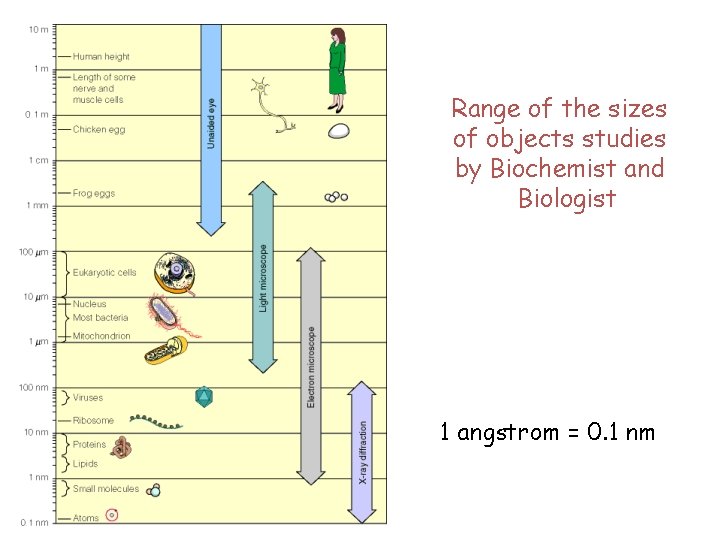
Range of the sizes of objects studies by Biochemist and Biologist 1 angstrom = 0. 1 nm

Elements of Life Most abundant, essential for all organisms: C, N, O, P, S, H Less abundant, essential for all organisms : Na, Mg, K, Ca, Cl Trace levels, essential for all organism: Mn, Fe, Co, Cu, Zn Trace levels, essential for some organisms: V, Cr, Mo, B, Al, Ga, Sn, Si, As, Se, I,
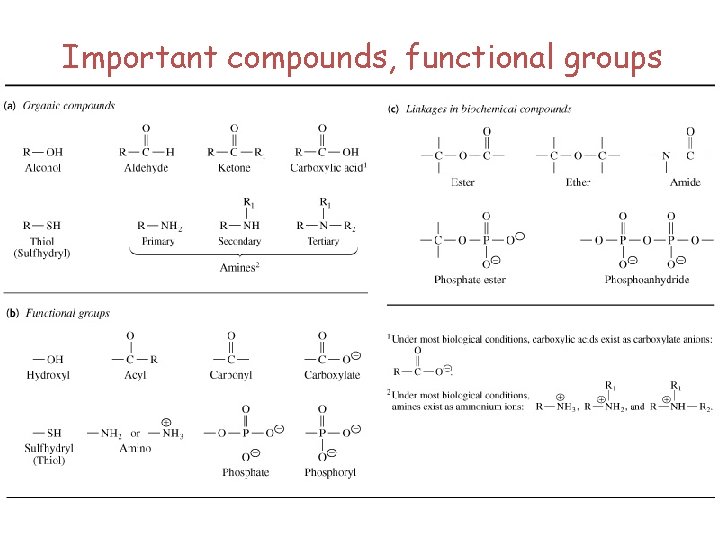
Important compounds, functional groups
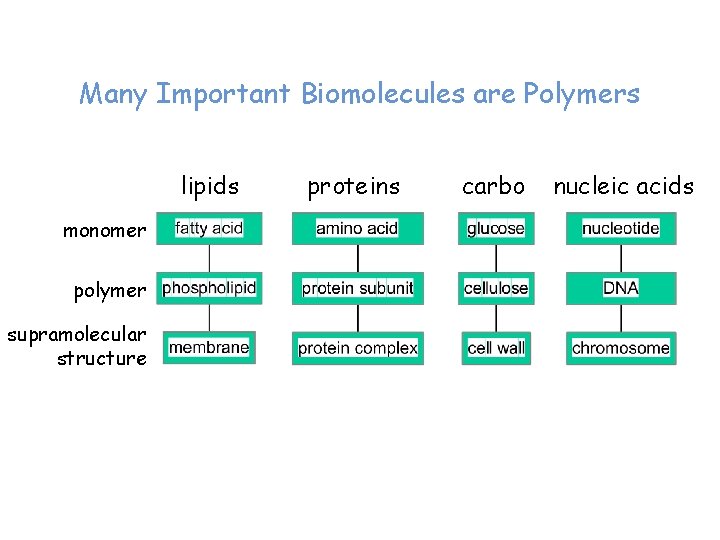
Many Important Biomolecules are Polymers lipids monomer polymer supramolecular structure proteins carbo nucleic acids
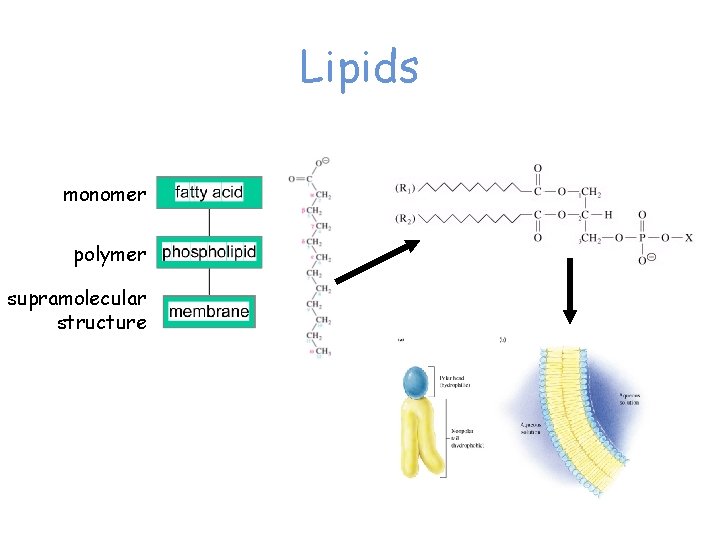
Lipids monomer polymer supramolecular structure
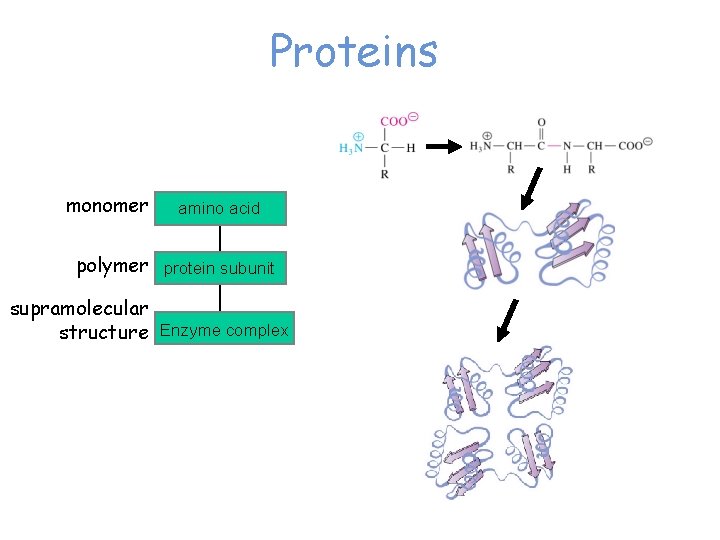
Proteins monomer amino acid polymer protein subunit supramolecular structure Enzyme complex
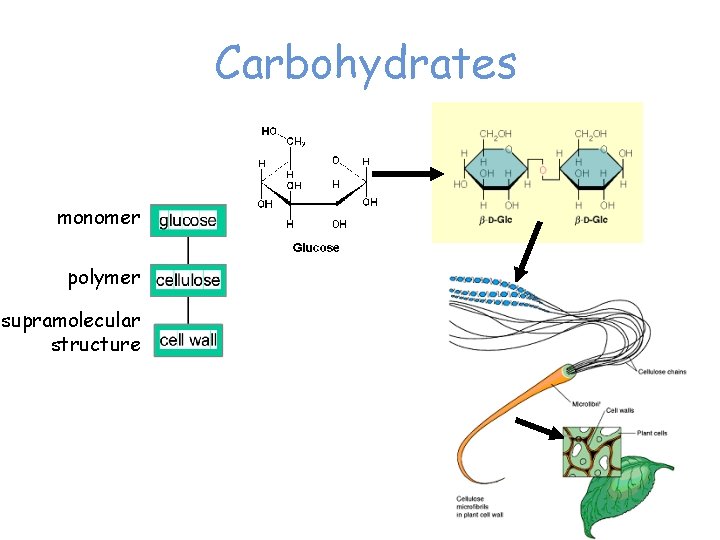
Carbohydrates monomer polymer supramolecular structure
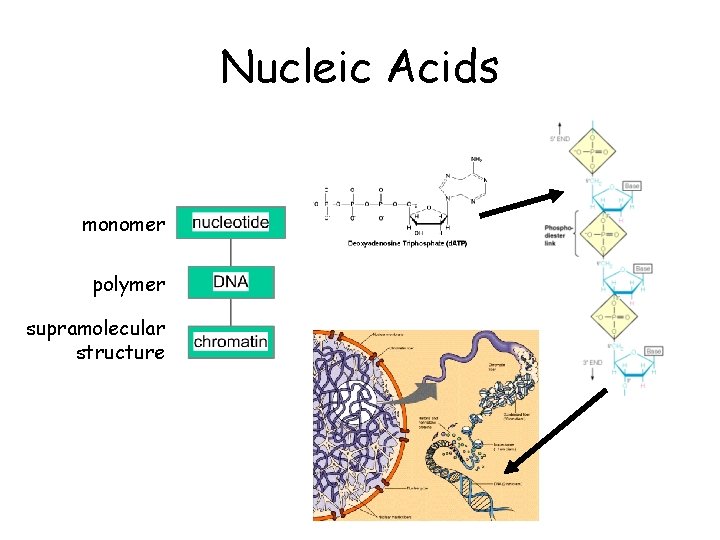
Nucleic Acids monomer polymer supramolecular structure
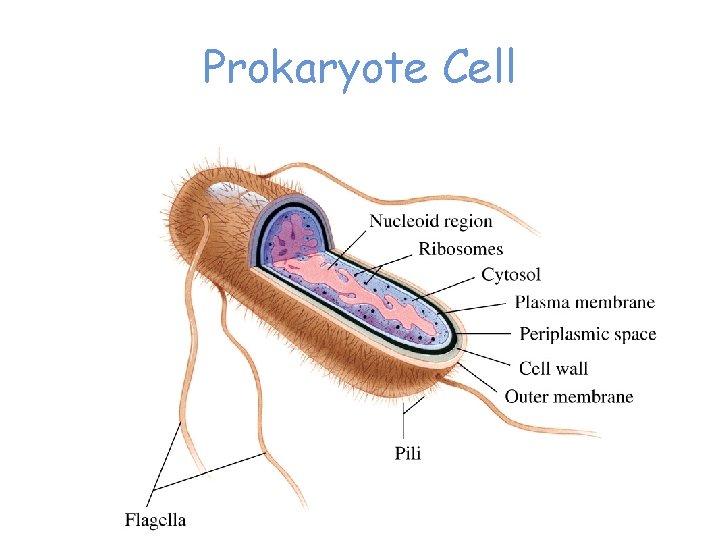
Prokaryote Cell

Cellular Organization of an E. coli Cell 200 – 300 mg protein / m. L cytoplasm
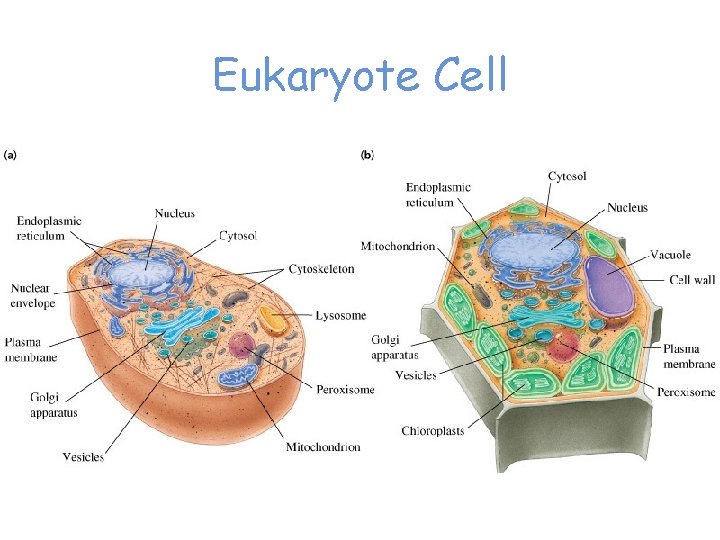
Eukaryote Cell

Carbohydrate

Carbohydrates • Most abundant class of biological molecules on Earth • Originally produced through CO 2 fixation during photosynthesis • Carbohydrate are aldehyde or ketone compounds with multiple hydroxyl group

Function • Function, the primary function of carbohydrates is to provide energy for the body, especially the brain and the nervous system. An enzyme called amylase helps break down carbohydrates into glucose (blood sugar), which is used for energy by the body.

Function • • Energy storage (glycogen, starch) Structural components (cellulose, chitin) Cellular recognition Carbohydrate derivatives include DNA, RNA, co-factors, glycoproteins, glycolipids
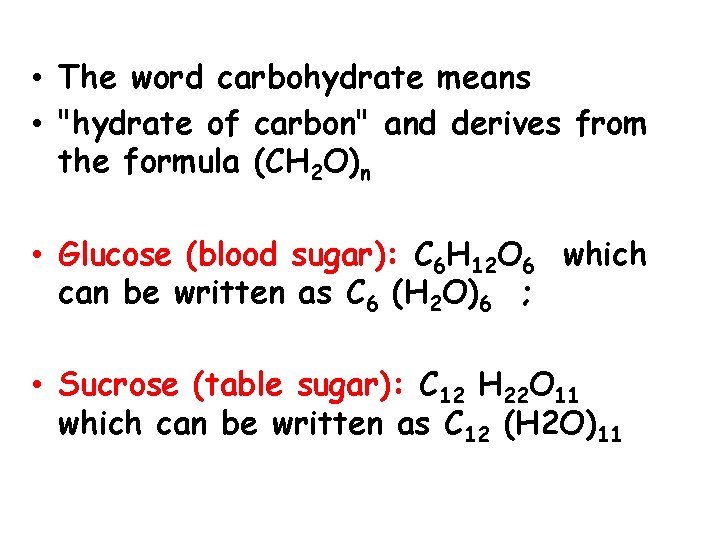
• The word carbohydrate means • "hydrate of carbon" and derives from the formula (CH 2 O)n • Glucose (blood sugar): C 6 H 12 O 6 which can be written as C 6 (H 2 O)6 ; • Sucrose (table sugar): C 12 H 22 O 11 which can be written as C 12 (H 2 O)11

●The simpler members of the carbohydrate family are often referred to as saccharides. ● Carbohydrates are classified as • Monosaccharides n≥ 3 • Oligosaccharides 2 -20 • Polysaccharides ≥ 20 depending on the number of simple sugars they contain.



• Mono saccharides containing an aldehyde group are classified as aldoses; those containing a ketone group are classified as ketoses. ●There are only two trioses: the aldotriose glyceraldehyde the ketotriose dihydroxyacetone.


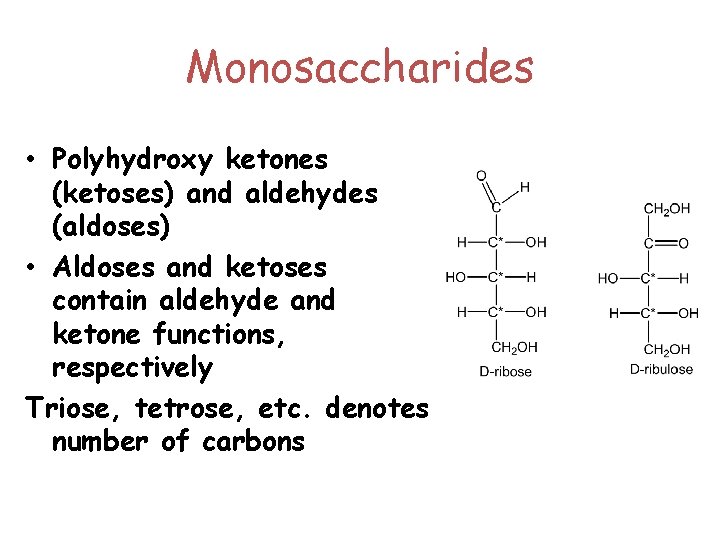
Monosaccharides • Polyhydroxy ketones (ketoses) and aldehydes (aldoses) • Aldoses and ketoses contain aldehyde and ketone functions, respectively Triose, tetrose, etc. denotes number of carbons
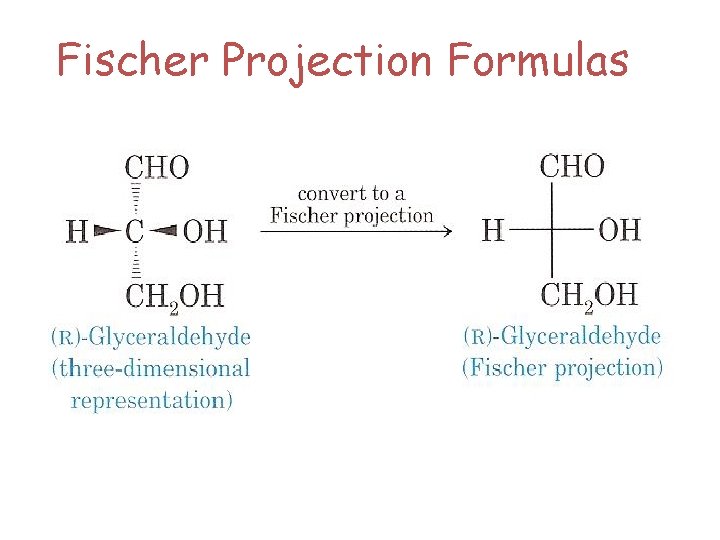
Fischer Projection Formulas
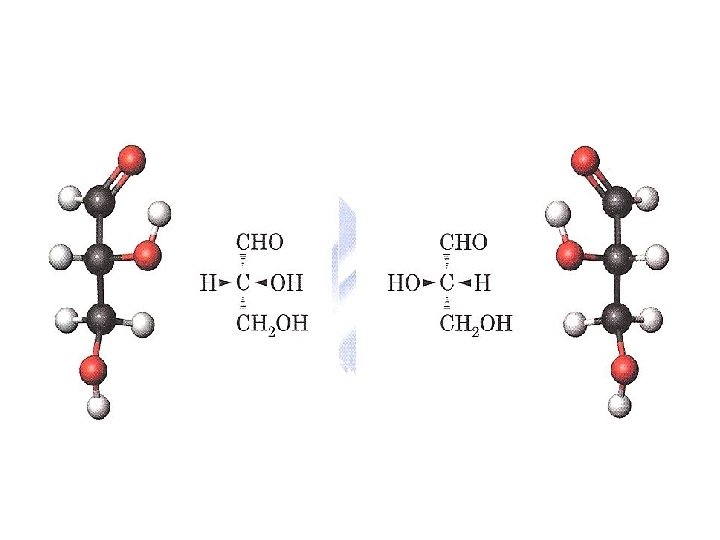

D- and L-Monosaccharides • The configuration of carbohydrates is commonly designated using the D, L system proposed by Emil Fischer in 1891. • Enantiomer of glyceraldehyde has a specific rotation of + 13. 5°; the other has a specific rotation of -13. 5°. • Fischer proposed that these enantiomers be designated D and L, but he had no experimental way to determine which enantiomer has which specific rotation.


• D-glyceraldehyde and L-glyceraldehyde serve as reference points for the assignment of relatively configurations to all other aldoses. • D- dihydroxyacetone and L- dihydroxyacetone serve as reference points for the assignment of relatively configurations to all other ketoses. ●A D-monosaccharide has the same configuration at its penultimate carbon as D-glyceraldehyde (its -OH group is on the right) in a Fischer projection; ●an L-monosaccharide has the same configuration at its penultimate carbon as L-glyceraldehyde (its -OH group is on the left).

Monosaccharides are chiral • The number of chiral carbons present in a ketose is always one less than the number found in the same length aldose • Number of possible steroisomers = 2 n (n = the number of chiral carbons)
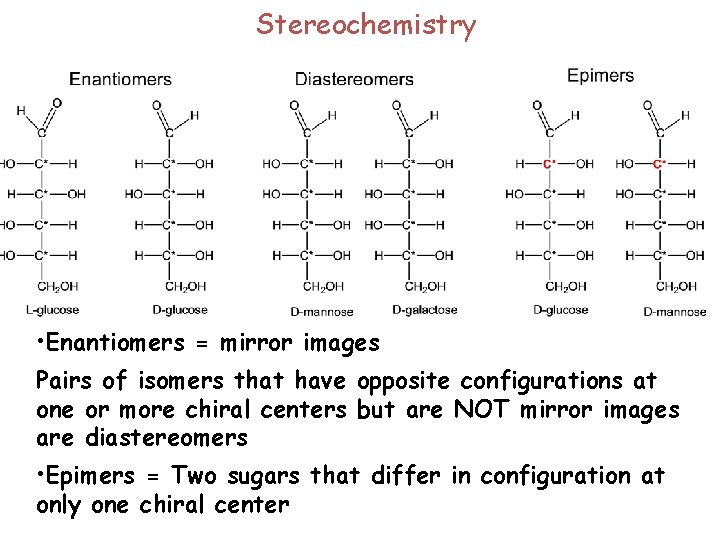
Stereochemistry • Enantiomers = mirror images Pairs of isomers that have opposite configurations at one or more chiral centers but are NOT mirror images are diastereomers • Epimers = Two sugars that differ in configuration at only one chiral center

D. Amino Sugars N Acetyl D glucosamine, (a derivat. Ive of D glucosamine), is a component of many polysaccharides, including connective tissue such as cartilage, chitin, the hard, shell like exoskeleton of lobsters, crabs, shrimp, and other shellfish. Several other amino sugars are components of naturally occurring antibiotics.


E. Physical Properties of Monosaccharides ●Monosaccharides are colorless, crystalline solids. ●all monosaccha rides are very soluble in water. Because hydrogen bonding is possible between their polar – OH groups and water, ● slightly soluble in ethanol and ● insoluble in nonpolar solvents such as diethyl ether, dichloromethane, and benzene.

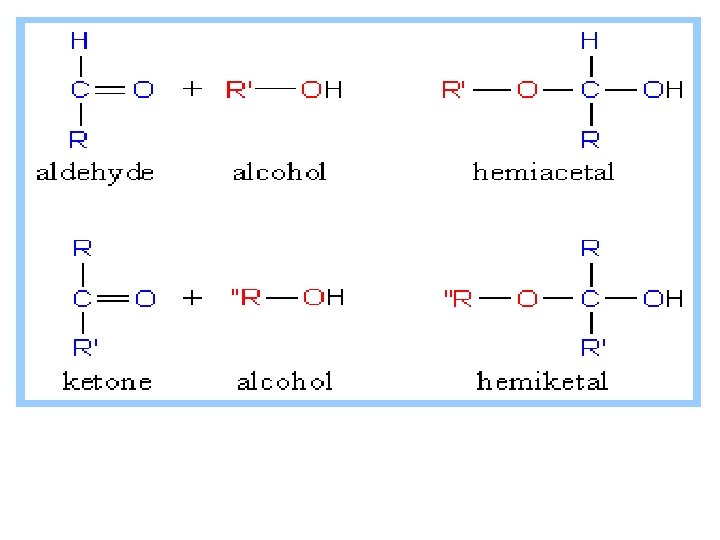
What Are the Cyclic Structures of Monosaccharides? we saw that aldehydes and ketones react with alcohols to form hemiacetals. We also saw that cyclic hemiacetals form very readily when hydroxyl and carbonyl groups are part of the same molecule and that their interaction produces a ring.
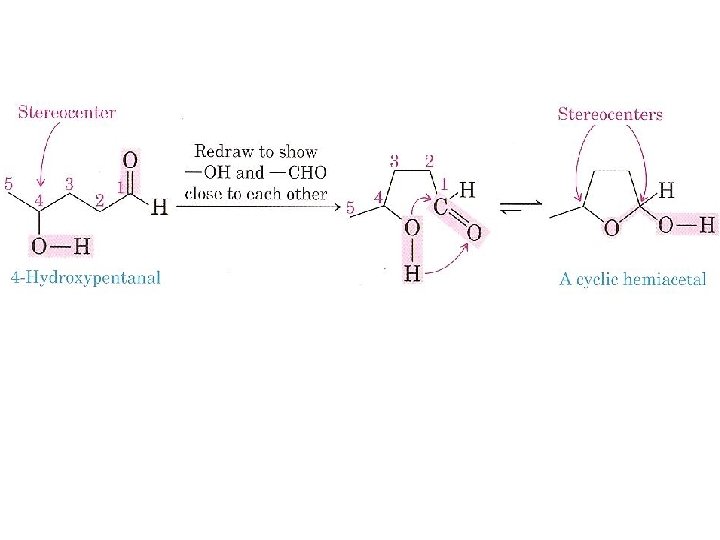

Monosaccharides have hydroxyl and carbonyl groups in the same molecule. As a result, they exist almost exclusively as five and six membered cyclic hemiacetals.

Cyclization of aldose and ketoses introduces additional chiral center • Aldose sugars (glucose) can cyclize to form a cyclic hemiacetal • Ketose sugars (fructose) can cyclize to form a cyclic hemiketal

A. Haworth Projections A common way of representing the cyclic structure of monosaccharides is the Haworth projection, named after the English chemist Sir Walter N. Haworth (Nobel Prize for chemistry, 1937).

• In a Haworth projection, a five or six memberedcyclic hemiacetal is represented as a planar pentagon or hexagon, • six membered hemiacetal ring is indicated by pyran , pyranose • five membered hemiacetal ring is indicated by furan-. furanose • Groups bonded to the carbons of the ring then lie either above or below the plane of the ring. • The new carbon stereocenter created in forming the cyclic structure is called an anomeric carbon. • Stereoisomers that differ in configuration only at the anomeric carbon are called anomers. • The anomeric carbon of an aldose is carbon 1; that of the most common ketoses is carbon 2.
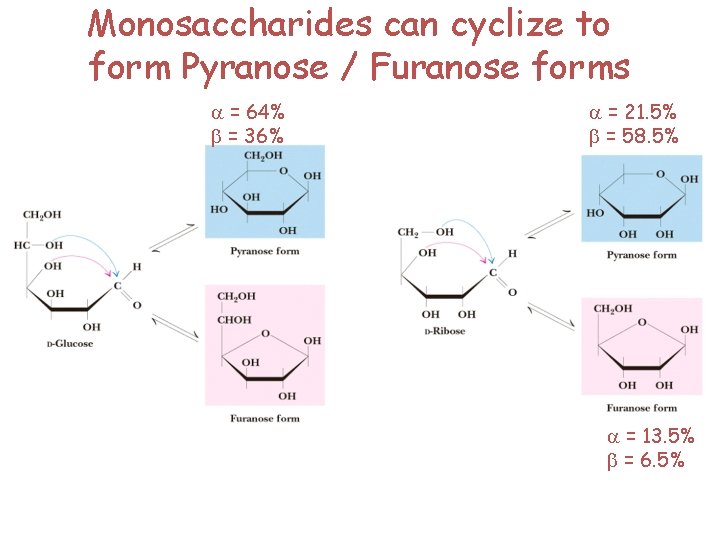
Monosaccharides can cyclize to form Pyranose / Furanose forms a = 64% b = 36% a = 21. 5% b = 58. 5% a = 13. 5% b = 6. 5%
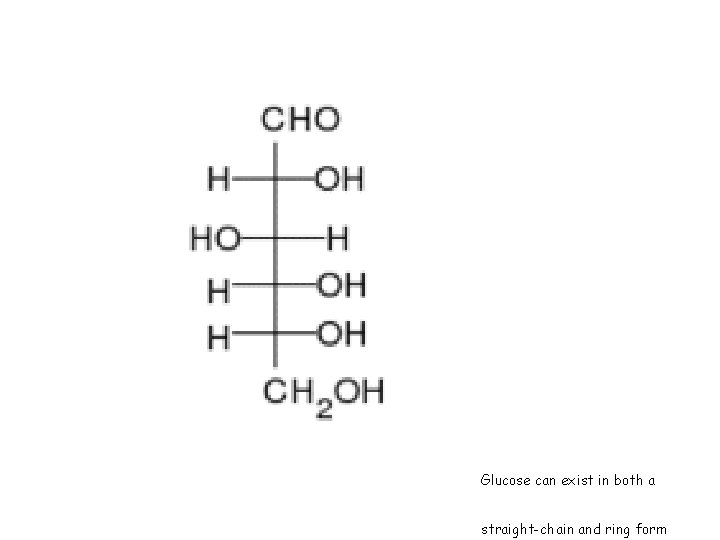
Glucose can exist in both a straight-chain and ring form

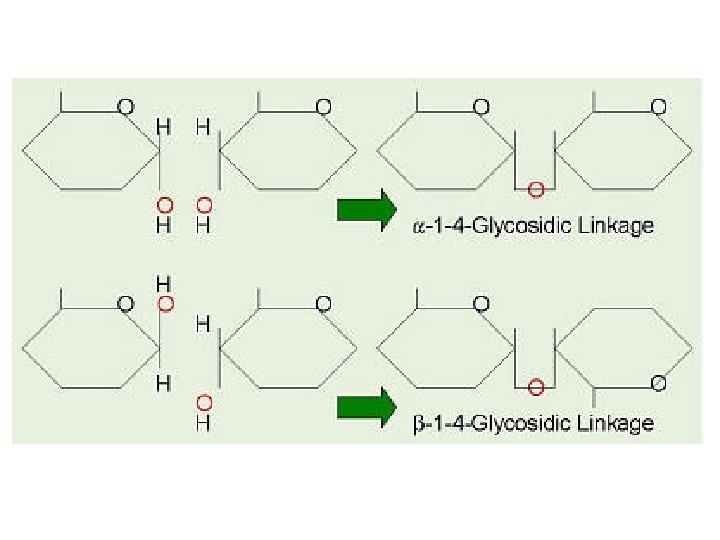
●the designation ß means that the OH on the anomeric carbon of the cyclic hemiacetal lies on the same side of the ring as the terminal –CH 2 OH. ●the designation α means that the OH on the anomeric carbon of the cyclic hemiacetal lies on the side of the ring opposite from the terminal CH 2 OH.


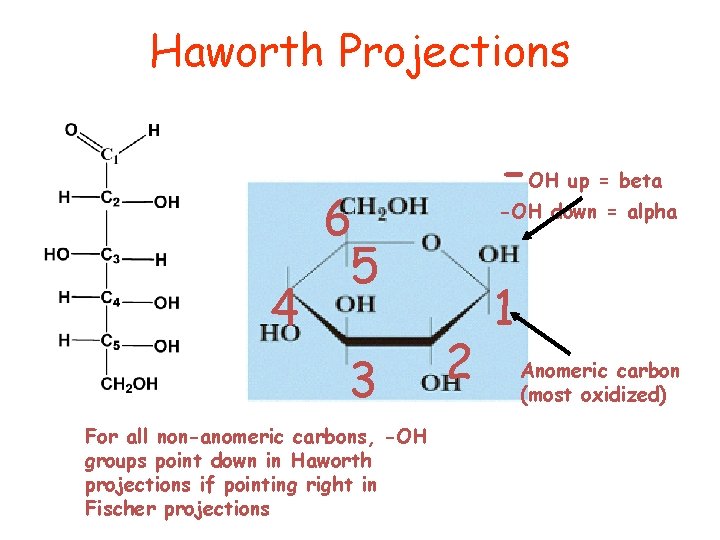
Haworth Projections 4 -OH up = beta 6 5 3 For all non-anomeric carbons, -OH groups point down in Haworth projections if pointing right in Fischer projections -OH down = alpha 2 1 Anomeric carbon (most oxidized)

C. Mutarotation The change in specific rotation that accompanies the equilibration of α and ß anomers in aqueous solution. A solution prepared by dissolving crystalline α D glucopyranose in water has a specific rotation of + 112°, which gradually decreases to an equilibrium value of +52. 7° as α D glucopyranose reaches equilibrium with ß D glucopyranose.
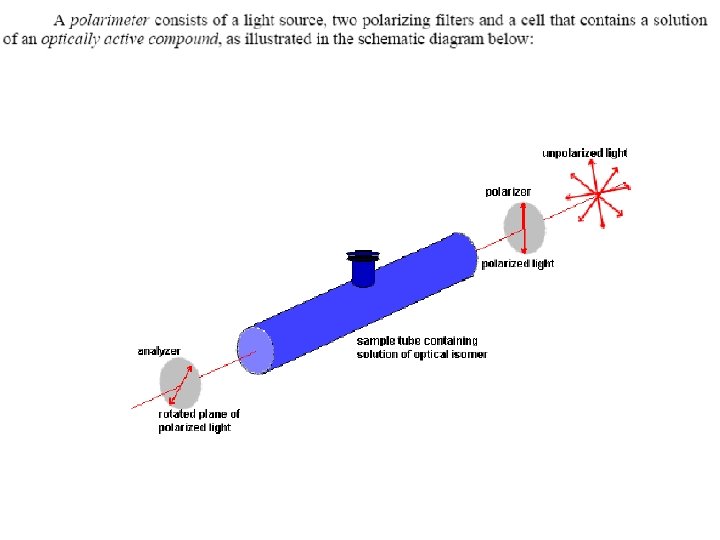

A solution of ß n glucopyranose also undergoes mutarotation, during which the specific rotation changes from + 18. 7° to the same equilibrium value of +52. 7°. The equilibrium mixture consists of 64% ß D glucopyranose and 36% α D glucopyranose, with only a trace (0. 003% ) of the open chain form.


What Are the Characteristic Reactions of Monosaccharides?

A. Formation of Glycosides (Acetals) Ø Treatment of an aldehyde or ketone with one molecule of alcohol yields a hemiacetal, Ø and treatment of the hemiacetal with a molecule of' alcohol yields an acetal. Treatment of a monosaccharide all forms of which exist almost exclusively as cyclic hemiacetals with an alcohol also yields an acetal,


Mutarotation is not possible in a glycoside because an acetal unlike a hemiacetal is no longer in equilibrium with the open chain carbonyl containing compound. Glycosides are stable in water and aqueous base; however, they are hydrolyzed in aqueous acid to an alcohol and a monosaccharide.

B. Reduction to Alditols The carbonyl group of a monosaccharide can be reduced to a hydroxyl group by a variety of reducing agents, including hydrogen in the presence of a transition metal catalyst and sodium borohydride. The reduction products are known as alditols.


• Sorbitol is found in the plant world in many berries and in cherries, plums, pears, apples, seaweed, and algae. • It is about 60% as sweet as sucrose (table sugar) and is used in the manufacture of candies and as a sugar substitute for diabetics. Other alditols commonly found in the biological world include erythritol, n mannitol, and xylitol. § Xylitol is used as a sweetening agent in "sugarless" gum, candy, and sweet cereals. •

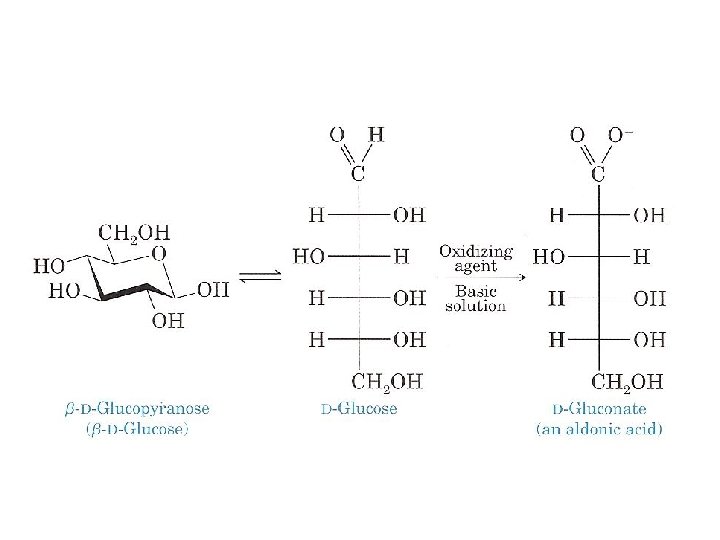
C. Oxidation to Aldonic Acids (Reducing Sugars) Aldehydes (RCHO) are oxidized to carboxylic acids (RCOOH) by several agents, including oxygen, O 2.

• Similarly, the aldehyde group of an aldose can be oxidized, under basic conditions, to a carboxylate group. • The cyclic form of the aldose is in equilibrium with the open chain form, which is then oxidized by the mild oxidizing agent. • D Glucose, for example, is oxidized to D gluconate (D gluconic acid).

Reducing and Non Reducing Sugar of Carbohydrates • Reducing sugar: In Reducing sugar, the two groups , aldehyde and ketone do not take part in the formation of glycosidic bond are present in a free or potentially free state and as such renders them powerful reducing agent. Reducing sugar include glucose, fructose, lactose which can reduce metal ions like Ca 2+ , Fe 3+ , in an alkaline medium. • Non Reducing sugar: In non reducing sugar , aldehyde and ketone group are involved in the formation of glycosidic bonds and thus are not free. Sucrose is a non reducing sugar.

• Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. • Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.

Reducing Sugars • When in the uncyclized form, monosaccharides act as reducing agents. • Free carbonyl group from aldoses or ketoses can reduce Cu 2+ and Ag+ ions to insoluble products

D. Oxidation to Uronic Acids Enzyme catalyzed oxidation of the primary alcohol at carbon 6 of a hexose yields a uronic acid. Enzyme catalyzed oxidation of D glucose, yields D glucuronic acids,


D Glucuronic acid is widely distributed in both the plant and animal worlds. In humans, it serves as an important component of the ■ acidic poly saccharides of connective tissues, ■ to detoxify foreign phenols and alcohols. ■ In the liver, these compounds are converted to glycosides of glucuronic acid (glucuronides) and excreted in the urine. ■The intravenous anesthetic propofol, for example, is converted to the following glucuronide and then excreted in urine


E. The Formation of Phosphoric Esters Mono and diphosphoric esters are important intermediates in the metabolism of monosaccharides. For example, the first step in glycolysis involves conversion of glucose to glucose 6 phosphate. Note that phosphoric acid is a strong enough acid so that at the p. H of cellular and intercellular fluids, both acidic protons of a phosphoric ester are ionized, giving the ester a charge of 2.

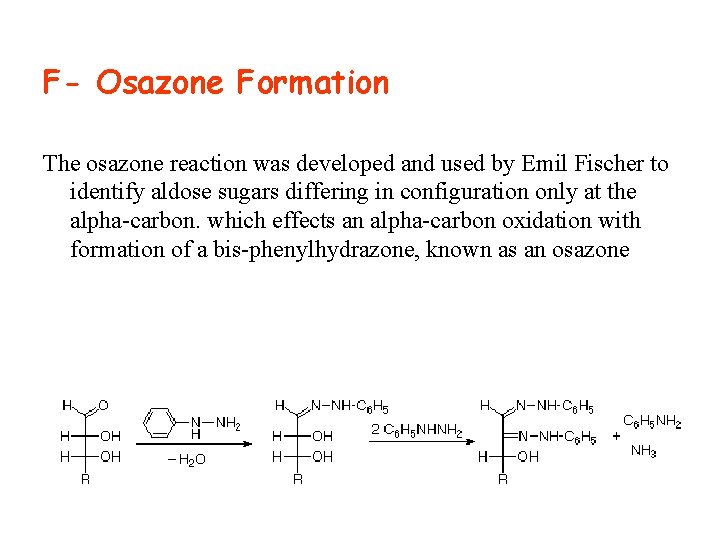
F- Osazone Formation The osazone reaction was developed and used by Emil Fischer to identify aldose sugars differing in configuration only at the alpha carbon. which effects an alpha carbon oxidation with formation of a bis phenylhydrazone, known as an osazone

• The technique was developed by Emil Fischer , , a. German chemist.

Application of the osazone reaction to D-glucose and Dmannose demonstrates that these compounds differ in configuration only at C-2.

Derivatives of Monosaccharides

Sugar Phosphates
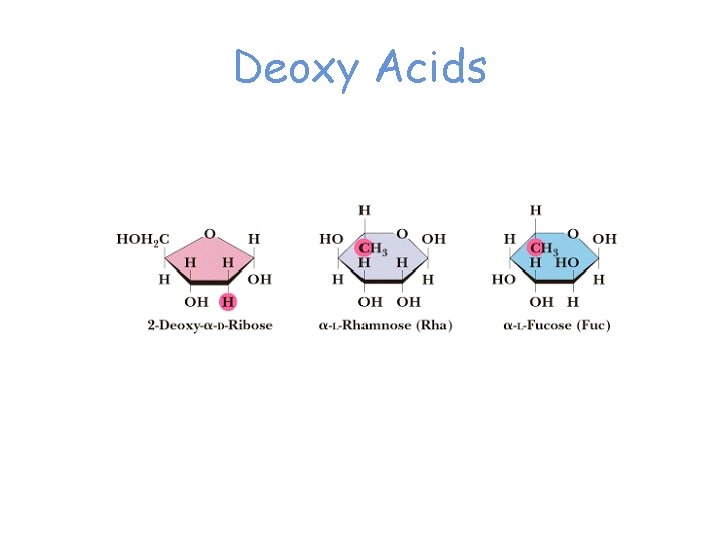
Deoxy Acids

Amino Sugars
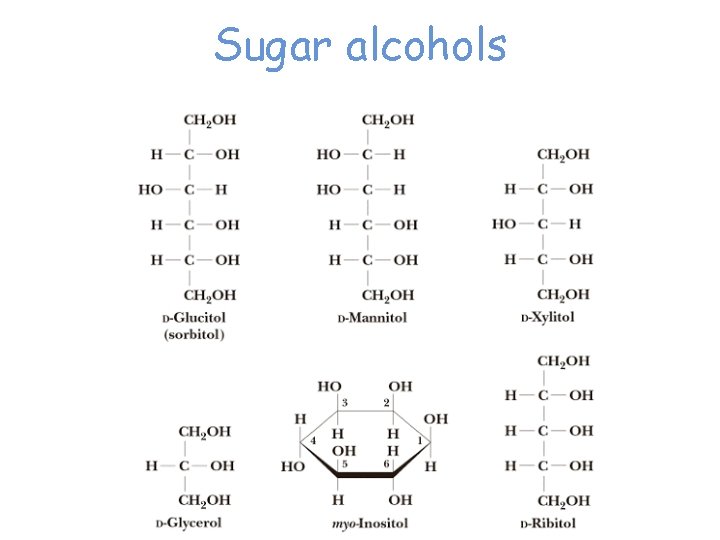
Sugar alcohols

What Are Disaccarides and Oligosaccharides? • Disaccharide a carbohydratc containing two monosaccharide units joined by a glycosidic boned • Most carbohydrates in nature contain more than one monosaccharide unit.

●disaccharides : those that contain two units ●trisaccharides: three units ●oligosaccharide: contain from six to ten monosaccharide units. ●polysaccharides: carbohydrates containing larger numbers of monosaccharide units.

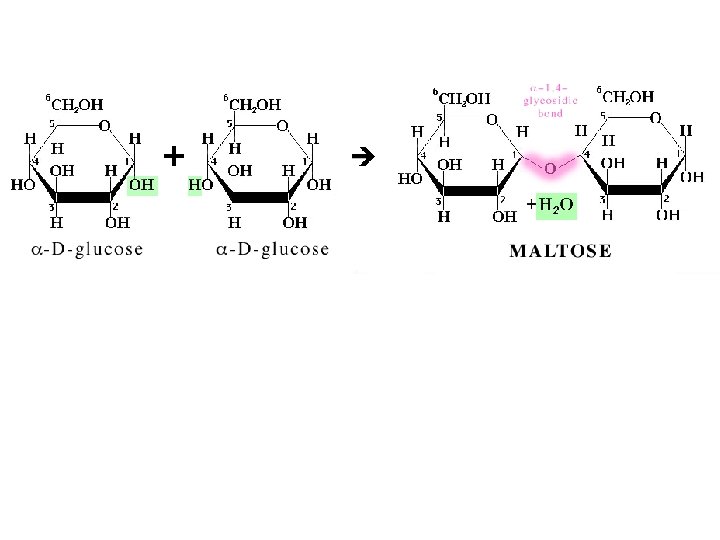
◊ In a disaccharide, two monosaccharide • units arc joined by a glycosidic bond between the anomeric carbon of one unit and an OH group of the other unit. ◊ Three important disaccharides are • sucrose, lactose, and maltose.

sugar 1 OH + HO sugar 2 sugar 1 + O sugar 2 + OH + H sugar 1 O sugar 2 + HOH Figure 3: Glycosidic Bond Formation

A. Sucrose (table sugar) is the most abundant disaccharide in the biological world. ●It is obtained principally from the juice of sugar cane and sugar beets ●carbon 1 of a D glucopyranose bonds to carbon 2 of D fructofuranose by an a l, 2 glycosidic bond.
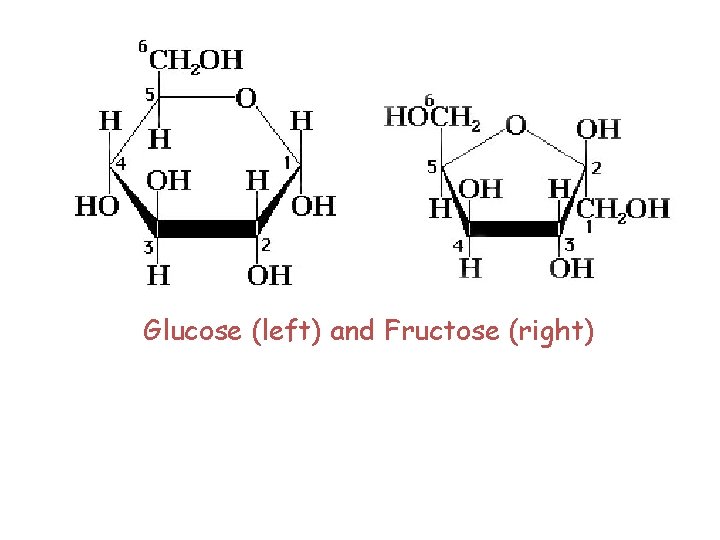
Glucose (left) and Fructose (right)
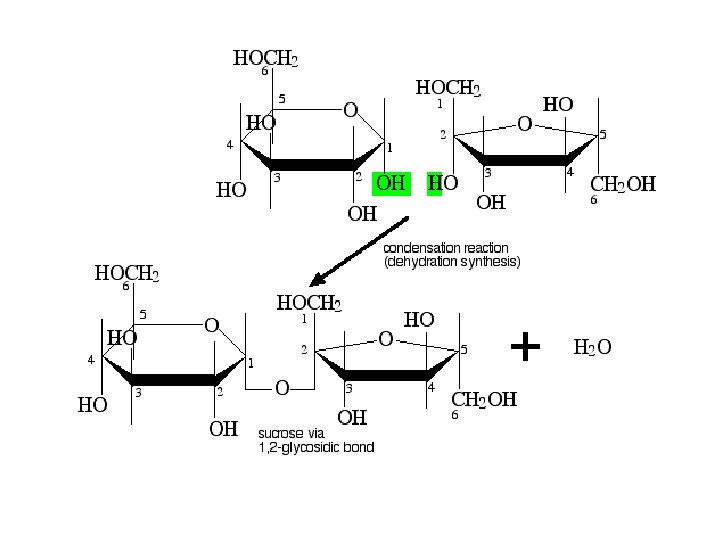
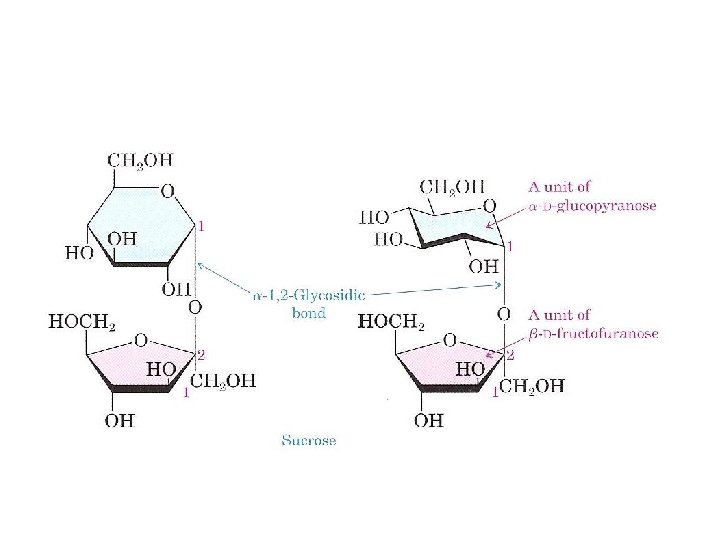


● non reducing sugar. Because the anomeric carbons of both the glucopyranose and fructofuranose units are involved in formation of the glycosidic bond, neither monosaccharide unit is in equilibrium with its open chain form. ● Oligosaccharide A carbohydrate containing from six to ten monosacchariede units, sauch joincd to thc next by a glycosidic boned • In the production of sucrose, sugar cane or sugar beet is boiled with water, and the resulting solution is cooled. Sucrose crystals separate and are collected. Subsequent boiling to concentrate the solution followed by cooling yields a dark, thick syrup known as molasses.

B. Lactose ◊ Lactose is the principal suger present in milk. ◊ It accounts for 5 to 8% of human milk and 4 to 6% of cows milk. ◊ Consists of D galactopyranose bonded by a β 1, 4 glycosidic bond to carbo 4 D glucopyranose. ◊ Lactose is a reducing suger, because the cyclic hemiacetal of the D glucopyranose unit is in equilibrium with its open chain form and can be oxidized to a carboxyl group.
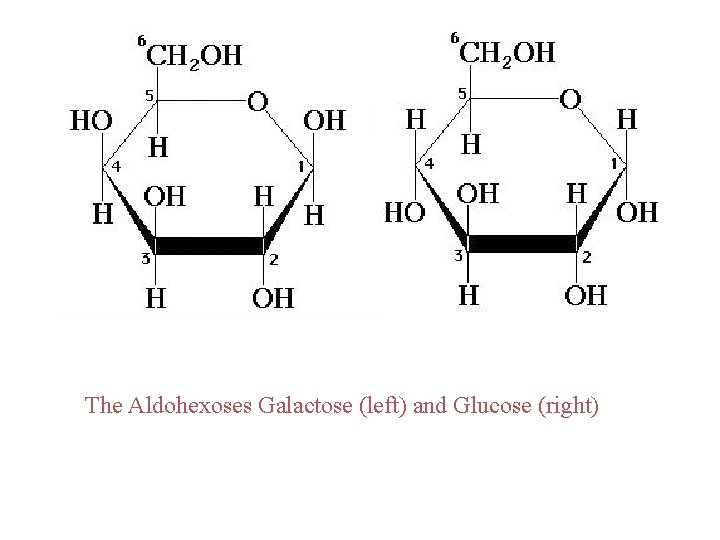
The Aldohexoses Galactose (left) and Glucose (right)

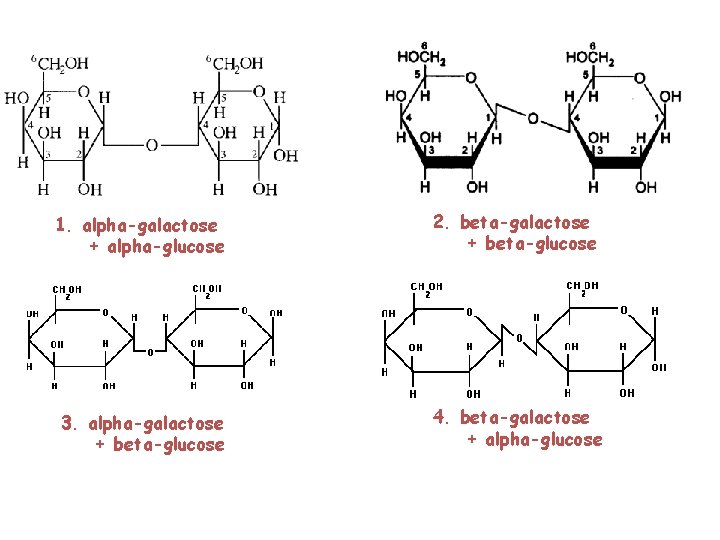
1. alpha-galactose + alpha-glucose 2. beta-galactose + beta-glucose 3. alpha-galactose + beta-glucose 4. beta-galactose + alpha-glucose



C. Maltose ●Maltose derives its name from its presence in malt, the juice from sprouted barley and other cereal grains. ●It consists of two units of D glucopyranose joined by glycosidic bond between carbon 1 (the unomeric r: arbon) of one unit and carbon 4 of the other unit.


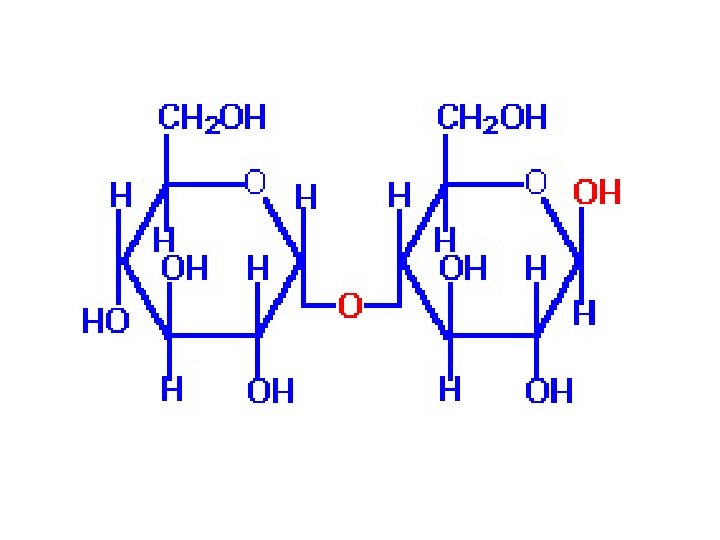

●Because the oxygen atom on the anomeric carbon of' the first glucopyranose unit is alpha, the bond joining the two units is an α l, 4 glycosidic bond. Following are a Haworth projection and a chair conformation for ß maltose, so named because the OH groups on the anomeric carbon of the glucose unit on the right are beta. ●Maltose is a reducing sugar; the hemiacetal group on the right unit of 11 glucopyranose is in equilibrium with the free aldehyde and can be oxidized to a carboxylic acid.

D. Relative Sweetness Among the disaccharide sweetening agents, n fructose tastes the sweetest even sweeter than sucrose. § The sweet taste of honey is due largely to 11 fructose and D glucose. § Lactose has almost no sweetness and is sometimes added to foods as a filler.
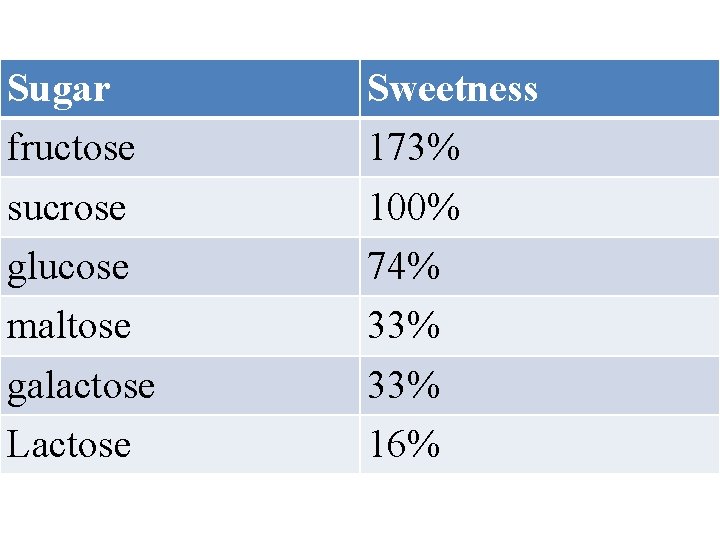
Sugar fructose sucrose glucose maltose galactose Lactose Sweetness 173% 100% 74% 33% 16%

What Are Polysaccharides? Polysaccharides consist of large numbers of monosaccharide units bonded together by glycosidic bonds. Three important polysaccharides, all made up of glucose units, are starch, glycogen, and cellulose.

A. Starch: Amylose and Amylopectin ●Starch is used for energy storage in plants. ●It is found in all plant seeds and tubers and is the form in which glucose is stored for later use. ●Starch can be separated into two principal polysaccharides: amylose and amylopectin, most starches contain 20 to 25% amylose and 75 to 80% mylopectin.


Ø Complete hydrolysis of both amylose and amylopectin yields only D glucose. q. Amylose is composed of continuous, unbranched chains of as many as 4000 n glucose units joined by α l, 4 g 1 ycosidic bonds. q. Amylopectin contains chains of as many as 10, 000 D glucose units also joined by α l, 4 g 1 ycosidic bonds. In addition, considerable branching from this linear network occurs. New chains of 24 to 30 units are started at branch points by α l, 6 g 1 ycosidic bonds.


B. Glycogen Ø Glycogen acts as the energy reserve carbohydrate for animals. Ø it is a branched polysaccharide(Like amy lopectin), containing approximately. l. O 6 glucose units joined by α l, 4 and α l, 6 glycosidic bonds. Ø The total amount of glycogen in the body of a well nourished adult human is about 350 g, divided almost equally between liver and muscle.

C. Cellulose Ø Cellulose, the most widely distributed plant skeletal polysaccharide, con stitutes almost half of the cell wall material of wood. Cotton is almost pure cellulose. Ø Cellulose is a linear polysaccharide of D glucose units joined by ß 1, 4 gly cosidic bonds. Ø It has an average molecular weight of 400, 000 g/mol, corresponding to approximately 2200 glucose units per molecule.


Why cellulose is insoluble in water? Cellulose molecules act much like stiff rods, a characteristic that enables them to align themselves side by side into well organized, water insoluble fibers in which the OH groups form numerous intermolecular hydrogen bonds. This arrangement of parallel chains in bundles gives cellulose fibers their high mechanical strength.

When a piece of cellulose-containing material is placed in water, there are not enough -OH groups on the surface of the fiber to pull individual cellulose molecules away from the strongly hydrogen-bonded fiber.

q. Humans and other animals cannot use cellulose as food because our digestive systems do not contain ß g 1 ucosidases, enzymes that catalyze the hydrolysis of ß glucosidic bonds. • Instead, we have only α glucosidases; hence, we use the polysaccharides starch and glycogen as sources of glucose. • In contrast, many bacteria and microorganisms do contain ß g 1 ucosidases and so can digest cellulose. ◊Termites, Ruminants

What Are Acidic Polysaccharides? • Acidic polysaccharides are a group of polysaccharides that contain carboxyl groups and/or sulfuric ester groups. • Acidic polysaccharides play important roles in the structure and function of connective tissues.

A. Hyaluronic Acid ●is the simplest acidic polysaccharide present in connective tissue. ●It has a molecular weight of between 105 and 107 glmol and contains from 300 to 100, 000 repeating units, depending on the organ in which it occurs. ●It is most abundant in embryonic tissues and in specialized connective tissues such as synovial fluid, the lubricant of joints in the body, and the vitreous of the eye, where it provides a clear, elastic gel that holds the retina in its proper position.


●Hyaluronic acid is also a common ingredient in lotions, moisturizers, and cosmetics. ● Hyaluronic acid is composed of n glucuronic acid joined by a β 1, 3 glycosidic bond to N acetyli n glucosamine, which is in turn linked to' n glucuronic acid by a β 1, 4 glycosidic bond.

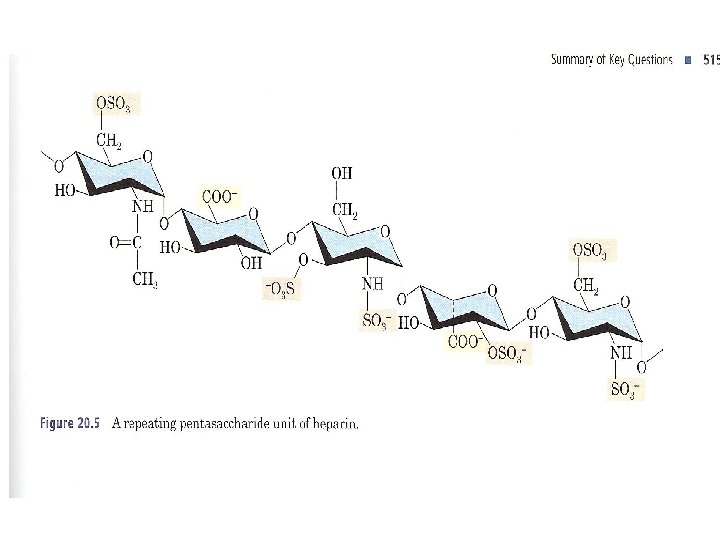
B. Heparin ●Heparin is a heterogeneous mixture of variably sulfonated polysaccharide chains, ●ranging in molecular weight from 6000 to 30, 000 g/mol. ● This acidic polysaccharide is synthesized and stored in mast cells of various tissues particularly the liver, lungs, and gut.

●Heparin has many biological functions, is anticoagulant. It binds strongly to antithrombin III, a plasma protein involved in terminating the clotting process. ●A heparin preparation with good anticoagulant activity contains a minimum of eight repeating units. The larger the molecule, the greater its anticoagulant activity. ●Because of this anticoagulant activity, it is widely used in medicine.

- Slides: 133