Introduction Water Resources Groundwater Surface water Rain water




























- Slides: 36


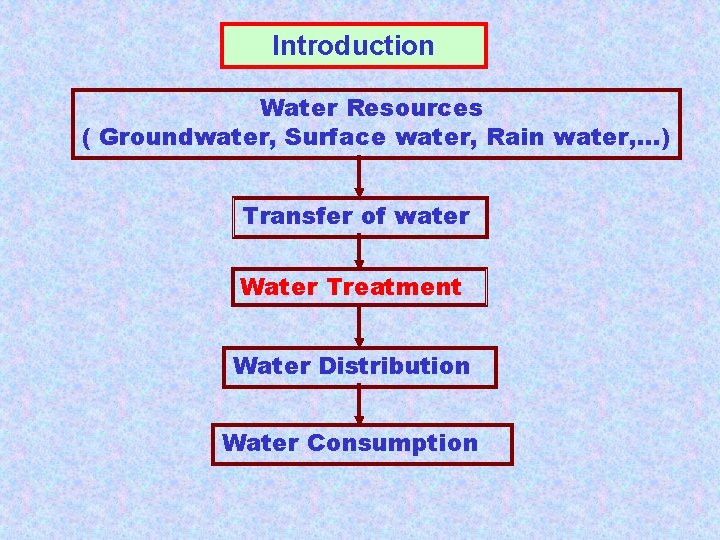
Introduction Water Resources ( Groundwater, Surface water, Rain water, …) Transfer of water Water Treatment Water Distribution Water Consumption

Water Treatment Objectives Removal of : q Suspended solids q Turbidity q Color q Taste & odor q Pathogens q Other specific pollutants (nitrate, heavy metals, organics, …) In order to Meet (Local, National, regional, and International) Standards

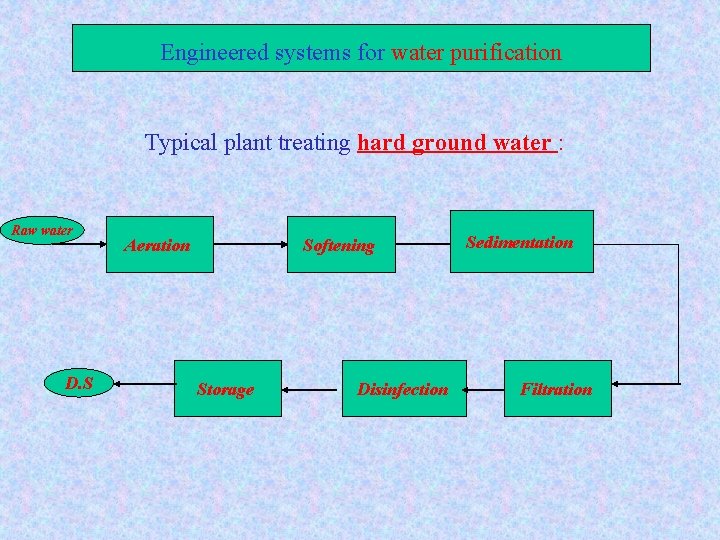
Engineered systems for water purification Typical plant treating hard ground water : Raw water D. S Aeration Softening Storage Disinfection Sedimentation Filtration

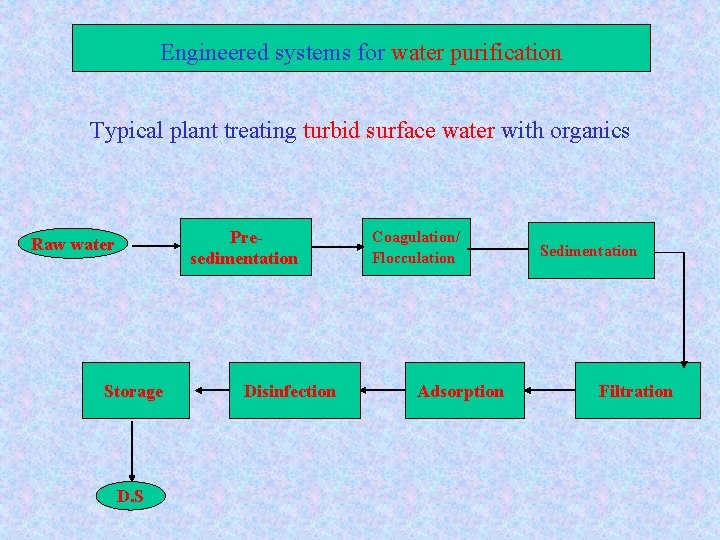
Engineered systems for water purification Typical plant treating turbid surface water with organics Presedimentation Raw water Storage D. S Disinfection Coagulation/ Flocculation Adsorption Sedimentation Filtration

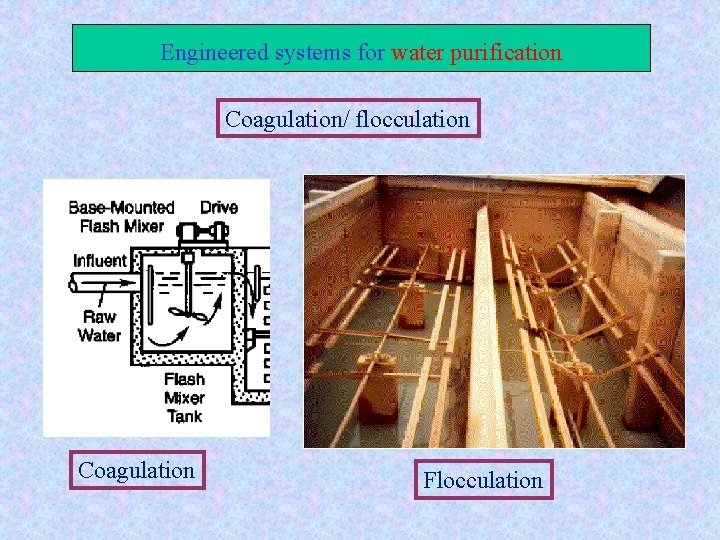
Engineered systems for water purification Coagulation/ flocculation Aim: To remove colloids from water Coagulation: chemical conditioning of colloids : flocculation : physical conditioning of colloids Main Coagulants: Aluminum sulfate : AL 2(SO 4)3 Iron salts : Fe. SO 4 PAC: Poly aluminum Chloride Floc

Engineered systems for water purification Coagulation/ flocculation Coagulation Flocculation

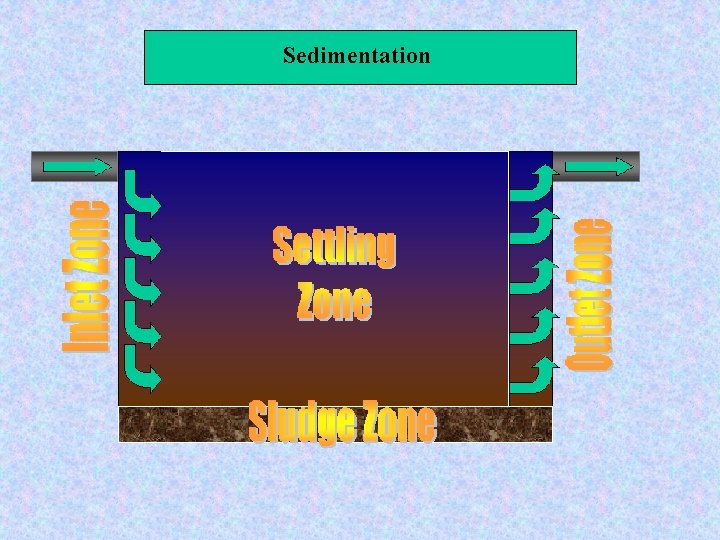
Sedimentation Aim: Solid- liquid separation Applications: - Plain settling of particles - Settling of flocculated particles - Settling of lime /soda softening particles - Settling of treated water from Fe/Mn removal

Engineered systems for water purification Sedimentation Types of sedimentation systems : - Circular: 4. 5 – 91 m Diameter, 3 – 4. 5 depth - Square: 10 – 60 m Diameter , 1. 6 - 6 m depth - Rectangular

Sedimentation

Sedimentation

Sedimentation Rectangular Clarifier (Outlet End)

Sedimentation Circular Clarifier (Outlet End)

Sedimentation

Sedimentation Settling types of solids : - Type 1: Low Conc. , Discrete particle - Type 2: - Type 3 : - Type 4 : High Conc. , Very flocculated

Sedimentation Type 1 settling: Discrete sedimentation fd Fb Stock’s Law Vp= g (ρp – ρw) d² /18μ Vp= Settling Velocity ρp = Solids density μ=Liquid Viscosity ρw = Liquid density d= Particle diameter 4 ﻭ 1 ﺍﻃﻼﻋﺎﺕ ﺑﻴﺸﺘﺮ ﻭ چﻨﺪ ﻣﺴﺌﻠﻪ ﺣﻞ ﺷﺪﻩ ﺩﺭ ﻣﺮﺍﺟﻊ ﺷﻤﺎﺭﻩ Fw fd

Sedimentation Design criteria: Over flow rate = Q/A = 20 -40 m³/m². d Detention time= 2 – 8 hr (4 -6 hr)

Filtration Filter types 1) Based on Driving Force 2) Based on Filtration Rate 3) Based on Flow direction 4) Based on media

Filtration 1) Based on Driving Force q Gravity filter q Pressure filter Gravity filter

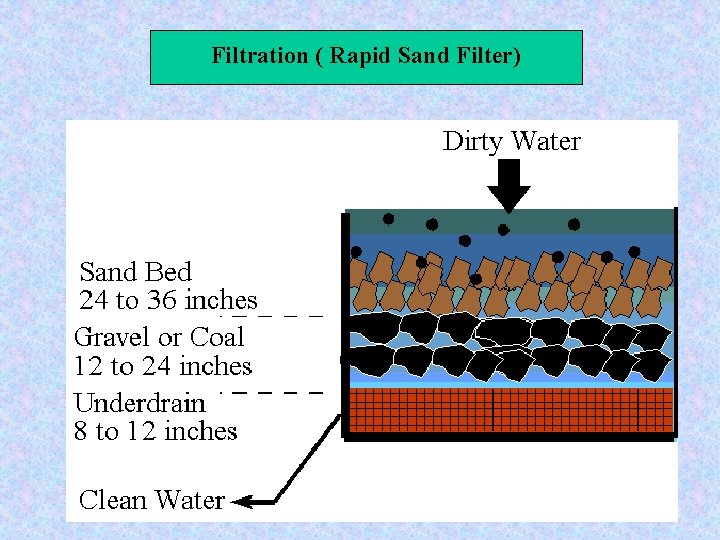
Filter Types Based on Filtration rate: - Slow sand filter (rate < 10 m³/m². d ) - Rapid filter : 120 m³/m². d - High rate : 240 m³/m². d

Filter Types Based on Flow direction : - Down flow - Up flow Based on media types: - Single medium filter - Dual medium filter - Multi media filter

Filtration ( Rapid Sand Filter)

Filtration ( Rapid Sand Filter)

Filtration ( Rapid Sand Filter)

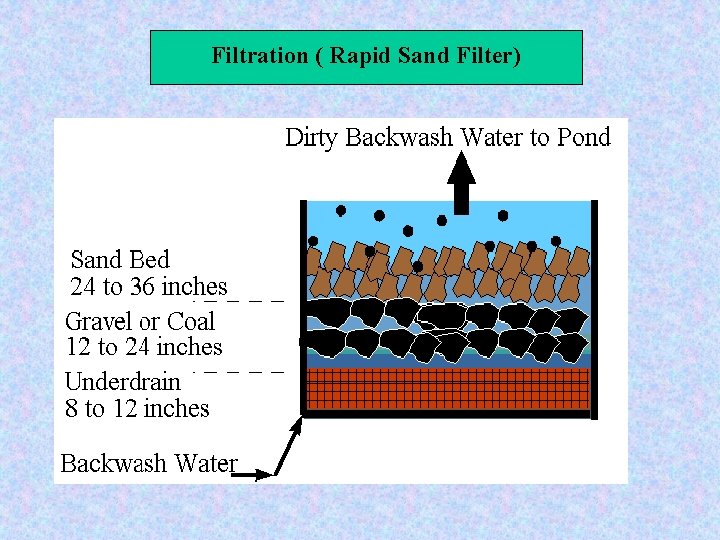
Filtration ( Rapid Sand Filter) Backwash Phase

Filtration ( Rapid Sand Filter) Filter Media Size and Uniformity of filter media: Effective size=ES= d 10 = the sieve size in mm which permit 10 percent of the medium to pass Uniformity coefficient = UC= d 60/ d 10 For Sand: ES= 0. 45 – 0. 55 mm US = 1. 2 -1. 7

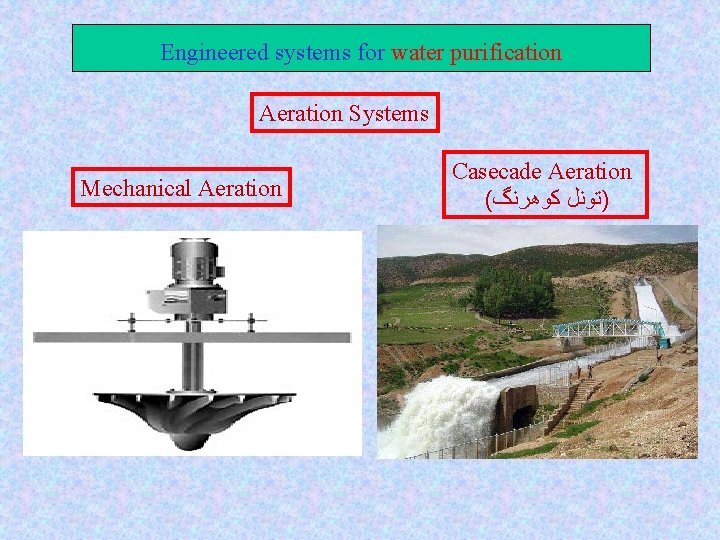
Engineered systems for water purification Aeration & gas transfer Aims: 1 - To remove undesirable dissolved gases in Water 2 - To remove dissolved inorganic substance such as Fe/Mn Aeration systems: q Compressed air --- Diffuser q Mechanical aeration q Cascade aeration q Spray aerator

Engineered systems for water purification Aeration Systems- Compressed air

Engineered systems for water purification Aeration Systems Mechanical Aeration Casecade Aeration ( )ﺗﻮﻧﻞ ﻛﻮﻫﺮﻧگ

Engineered systems for water purification Softening Aim: hardness reduction to desirable value (50 -150 mg/l) Important Softening methods: q Chemical Softening q Ion Exchange

Engineered systems for water purification Softening Chemical precipitation : - Ca hardness: Calcium Carbonate ↓ - Mg hardness: Magnesium Hydroxide ↓ Ion exchange : {Ca +² / Mg +²} + 2 Na-R → {Ca / Mg }-R+ 2 Na + {Ca / mg }-R+ 2 Na Cl → { Ca / Mg} Cl 2 + 2 Na- R

Disinfection Aim: Destruction of pathogens from water Main Disinfectants used in water treatment: q Chlorine q Ozone q UV

Disinfection q Chlorine is most common disinfectant in water and wastewater treatment Disadvantages: q Chlorine is a dangerous chemical q Can form disinfection byproducts (DBPs), Trihalomethanes (THMs) which are possible carcinogens

Disinfection Technology Environmentally Byproducts Effect Investment Operational costs + + ++ - + ++ ++ + -/+ ++ -- -- - + ++ friendly Ozone UV Chlorine gas Ref: http: //www. lenntech. com/processes/disinfection/

Ozonation system in Water Treatment Plants

Disinfection By UV radiation