INTRODUCTION v Upper gastrointestinal UGI bleeding arising proximal



INTRODUCTION v Upper gastrointestinal (UGI) bleeding (arising proximal to the ligament of Treitz in the distal duodenum) commonly presents with hematemesis (vomiting of red blood or coffee ground-like material) and/or melena (black, tarry stools). v Hematochezia (bright red or maroon-colored blood or fresh clots per rectum) is usually a sign of a lower gastrointestinal (LGI) source (defined as distal to the ligament of Treitz).

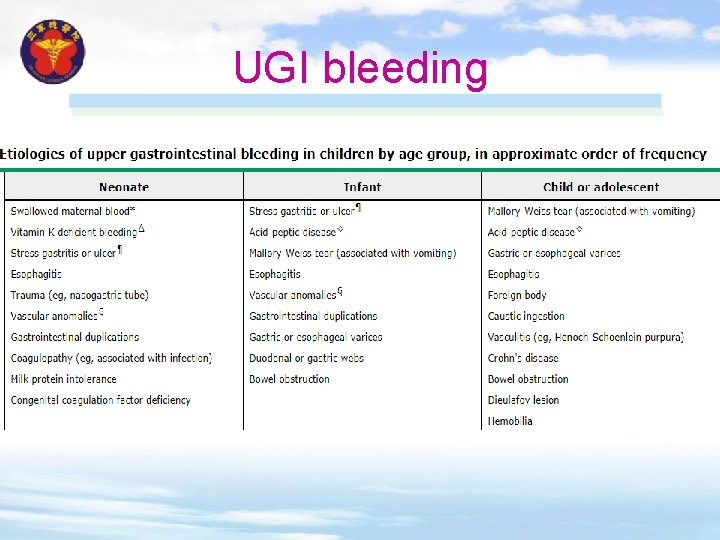
ETIOLOGY v The most common causes of UGI bleeding in children vary depending upon age and the geographic setting. v In Western countries, the most common causes are gastric and duodenal ulcers, esophagitis, gastritis, and varices v The most common causes of rectal bleeding in infants are anal fissure or cow's milk or soy protein-induced colitis. v In children 12 months and older, the most common causes of rectal bleeding are infectious gastroenteritis and anal fissures.
UGI bleeding

Neonates — UGI bleeding v True UGI bleeding in a neonate must be distinguished from swallowed maternal blood v Vitamin K deficient bleeding (hemorrhagic disease of the newborn) should be considered in neonates who were not given vitamin K prophylaxis at birth v Stress gastritis or ulcers are associated with critical illness but also may occur spontaneously even in the first few days of life v Congenital anomalies including intestinal duplications or vascular anomalies may present with gastrointestinal (GI) hemorrhage v Coagulopathy in a neonate may also be caused by infection, liver failure, or a congenital coagulation factor deficiency. Several types of coagulopathies can present during the newborn period v Milk protein intolerance may present with UGI bleeding, although lower gastrointestinal (LGI) bleeding is much more common.

Infants, children and adolescents — UGI bleeding v Mallory-Weiss syndrome – Mallory-Weiss syndrome is characterized by longitudinal mucosal lacerations in the distal esophagus, usually developing after forceful retching. The bleeding is usually small and self-limited, but occasionally is severe v Esophageal or GI foreign body – A foreign body can cause GI bleeding if it is sharp, caustic, and/or lodged in the esophagus. Clinical clues to this possibility include a history of a choking episode, even if it was transient or occurred days or even weeks before the bleeding episode. Rarely, ingestion of a button battery has led to severe UGI hemorrhage due to aortoesophageal fistula, which can be fatal. v Esophagitis – Esophagitis in this age group usually is caused by gastroesophageal reflux disease or eosinophilic esophagitis, and occasionally by caustic ingestion. Peptic esophagitis also may develop after recurrent vomiting from other causes

v Peptic ulcers and gastritis – Gastritis and ulcers occasionally occur in all age groups, typically in the setting of critical illness or use of nonsteroidal antiinflammatory drugs (NSAIDs). Young children are particularly susceptible to developing UGI bleeding after NSAID use. Binge drinking of alcohol is an important cause of gastritis in adolescents. Gastritis or peptic ulcers also may be related to Helicobacter pylori infection or occasionally to a viral infection including cytomegalovirus v Bleeding from esophageal varices – Variceal bleeding is the most common cause of severe acute UGI bleeding in children. Esophageal varices are caused by portal vein hypertension. Clues to portal hypertension include splenomegaly and/or a history of thrombocytopenia, even in a patient without a history of liver disease.

CLINICAL ASSESSMENT v The initial evaluation of the patient with UGI bleeding should always start with an assessment of hemodynamic stability and resuscitation, if indicated. v Endoscopy usually is indicated if the bleeding is brisk or unexplained after a thorough history and physical examination, or if there associated signs of shock. Brisk bleeding is suggested by repeated episodes of grossly bloody emesis or large volumes of blood aspirated through a nasogastric tube, or a persistent drop in hemoglobin. v In some cases, the source of the bleeding can be treated through the endoscopic procedure. v Endoscopic evaluation and treatment should generally be performed after the patient is stabilized, and within 24 to 48 hours of presentation of the gastrointestinal (GI) bleed.
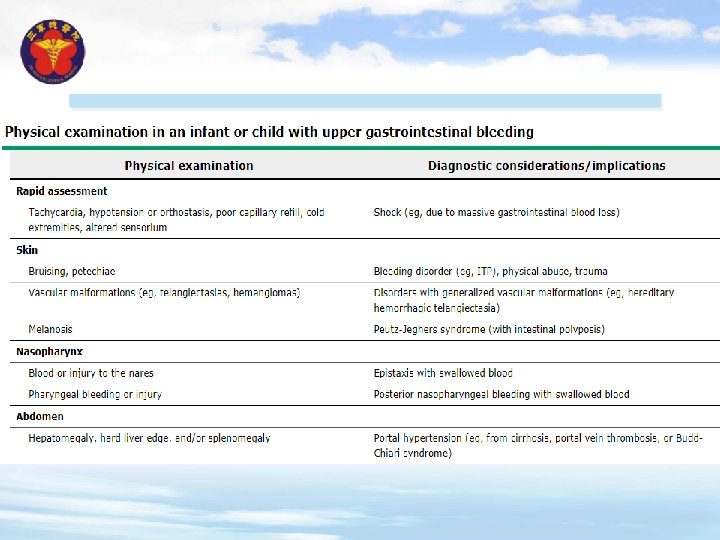

Laboratory evaluation v In most cases, it should include a complete blood count, coagulation studies, tests evaluating liver function, blood urea nitrogen (BUN), and serum creatinine. v For patients with epigastric abdominal pain, pancreatitis also should be ruled out with screening amylase and lipase; pancreatitis occasionally is associated with gastritis, duodenitis, and peptic ulcer disease. v The BUN result can be helpful for confirming the source of bleeding. An increase in BUN in the absence of renal disease is consistent with an UGI (rather than a lower gastrointestinal [LGI]) source of blood loss because blood in the proximal GI tract has relatively more time to be absorbed, leading to an increase in the BUN.

Other diagnostic tests v Angiography may be useful in patients with rapid bleeding in whom endoscopy is unsuccessful in finding a source. For diagnostic purposes, magnetic resonance angiography (MRA) or computed tomographic angiography (CTA) may be used. v For therapeutic purposes, standard angiography may be useful in treating some patients with vascular anomalies, hemobilia, or some ulcers that are not amenable to other types of treatment. v Radionucleotide imaging (a tagged red blood cell scan) also can be used to detect an obscure bleeding source for patients with very brisk bleeding. However, this is rarely helpful in patients with UGI bleeding because endoscopy is far more sensitive for detecting bleeding above the ligament of Treitz.

MANAGEMENT v Nasogastric tube — In patients presenting with unexplained gastrointestinal (GI) bleeding that is clinically significant (eg, more than a teaspoon estimated blood loss), nasogastric or orogastric tube lavage is sometimes used to confirm the diagnosis and to determine if the bleeding is ongoing. v Pharmacologic options — Acid suppression usually is appropriate for children with clinically significant UGI bleeding, to treat or prevent any peptic component of the underlying disorder. Vasoactive agents may be helpful for selected cases of vascular bleeding

v Endoscopy — Guidelines recommend that endoscopy be performed within 24 to 48 hours for infants and children presenting with UGI bleeding that is acute and severe, particularly if transfusions are required. Earlier endoscopy may be needed if bleeding cannot be controlled. Hemodynamically unstable patients should be stabilized prior to endoscopy, including transfusion and correction of coagulopathy if present. Endoscopy is also appropriate in children with low-grade bleeding that is unexplained and persistent or recurrent. v Upper endoscopy permits identification of the bleeding source, allows for risk stratification regarding the likelihood of continued bleeding, and permits therapeutic intervention

v Surgery or angiography is reserved for the uncommon patients in whom endoscopy fails to control bleeding or in whom an anatomic abnormality exists that requires surgery, and if the patient cannot be fully stabilized despite resuscitative measures.
LGI bleeding

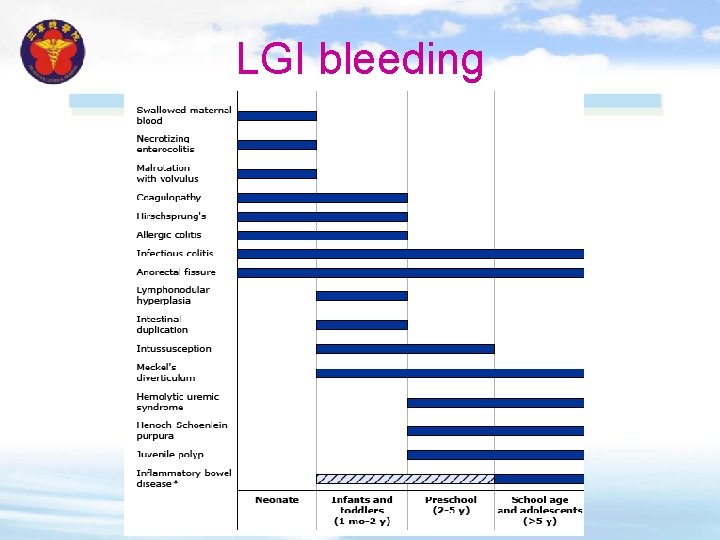
Neonatal period v Swallowed maternal blood — In a newborn infant with rectal bleeding, the rectal blood should be tested to determine whether it comes from the infant or whether it represents maternal blood, which may have been swallowed during delivery or ingested during breast feeding from cracked nipples. This is accomplished using the Apt test (hemoglobin alkaline denaturation test), which detects fetal hemoglobin. v Anal fissures — Anal fissures are the most common cause of rectal bleeding in patients younger than one year and are also common in older children and adults. They are diagnosed easily by spreading the perineal skin to evert the anal canal. In an infant, the history often suggests painful defecation with straining, grunting, and leg stiffening or back arching consistent withholding behavior and streaks of bright red blood on the surface of the stools.

v Necrotizing enterocolitis — Necrotizing enterocolitis (NEC) is an acute illness of unclear etiology associated with intestinal necrosis. NEC should be suspected in a newborn with nonspecific systemic signs such as apnea, respiratory failure, lethargy, poor feeding, or temperature instability, and abdominal signs including distention, gastric retention (residual milk in the stomach before a feeding), tenderness, vomiting, diarrhea, and gross or occult LGIB. Although most infants who develop NEC were born prematurely, approximately 13 percent of cases occur in term. Pneumatosis intestinalis, the hallmark of NEC, appears as bubbles of gas in the bowel wall or in the portal system.

v Malrotation with midgut volvulus — Newborns who have malrotation with midgut volvulus typically present with abdominal distension, emesis which may or may not be bilious, and melena or hematochezia (in 10 to 20 percent of cases). Bilious emesis in the neonatal period should be assumed to represent a surgical emergency due to obstruction until proven otherwise. A limited upper gastrointestinal (UGI) contrast series is the best examination to visualize the position of the duodenum. UGI contrast series should be performed whenever possible, under fluoroscopy and by an experienced pediatric radiologist. Barium contrast studies may reveal a corkscrew appearance of the twisted small bowel, or a "bird's peak" if complete obstruction is present.

v Hirschsprung disease — Newborns with Hirschsprung disease frequently have delayed passage of meconium (>48 hours after birth). Some present with acute obstruction manifested by vomiting (which may be bilious or feculent) and abdominal distension. Other infants may present at several weeks of age with progressive constipation or diarrhea associated with abdominal distension. Only one-quarter of the patients have blood in the stool. Significant blood in the stool with abdominal distension in infants with known or suspected Hirschsprung disease may be indicative of Hirschsprungassociated enterocolitis (toxic megacolon) and should be considered a medical emergency. The gold standard for diagnosis is the demonstration of complete absence of ganglion cells in the Meissner and the Auerbach plexus on a biopsy specimen of intestinal mucosa and submucosa. Treatment generally is surgical resection of the aganglionic segment.

v Coagulopathy — Several types of coagulopathies can present during the newborn period. Most present with other bleeding symptoms, such as a large cephalohematoma after vaginal delivery, oozing from the umbilical stump, prolonged bleeding after circumcision or blood sampling, or intracranial hemorrhage in a term infant. Occasionally, these coagulopathies come to medical attention because of LGIB, although this rarely occurs during the neonatal period.

Infants and toddlers v Anal fissures v Milk- or soy-induced colitis — Milk- or soy protein-induced colitis is an inflammatory reaction caused by ingestion of cow's milk or soy proteins, and is a common cause of bloody stools in infants. It occurs almost exclusively in infants and usually resolves within 6 to 18 months of age. Up to 25 percent of patients with cow's milk protein intolerance will have a cross-reaction to soy protein, and a few infants are sensitive to other food proteins. Treatment involves meticulous elimination of the causative protein from the mother's diet if the infant is breastfed, or the use of a casein-hydrolysate formula.

v Intussusception — Intussusception is the most common cause of intestinal obstruction in infants between 6 and 36 months of age. In younger group, intussusception usually is idiopathic and occurs in the ileocecal region, in contrast to older children in whom a polyp or Meckel's diverticulum or other lesions often serve as a lead point. Patients may awaken from sleep with severe abdominal pain. They vomit and may pass a stool and improve temporarily before the cycle repeats. A sausage-shaped mass in the distribution of the colon, typically in the area of the transverse colon, may occasionally be palpable on abdominal examination. The stool contains gross or occult blood in most but not all cases, and sometimes has the appearance of "currant jelly. “ Ultrasonography is the method of choice to detect intussusception in most institutions. The diagnosis can also be established with an air or water-soluble contrast enema, which also can treat ("reduce") the intussusception in 75 to 90 percent of children in whom a lead point is not present.

v Meckel's diverticulum — Meckel's diverticulum results from incomplete obliteration of the omphalomesenteric duct. It is usually asymptomatic, but may cause painless rectal bleeding, which may be chronic and insidious, or acute and massive. The bleeding is often caused by mucosal ulceration of adjacent small bowel tissue due to production of acid by ectopic gastric tissue within the diverticulum. Other complications associated with a Meckel's diverticulum are obstruction, perforation, diverticulitis, and intussusception. The diagnosis is made by a Meckel scan. The scan consists of the intravenous administration of 99 m technetium pertechnetate, which has an affinity for gastric mucosa, followed by scintigraphy to identify areas of ectopic gastric mucosa. Any Meckel's diverticulum that is symptomatic should be resected. An asymptomatic Meckel's diverticulum discovered incidentally at laparotomy usually also is resected in children.

v Lymphonodular hyperplasia — Lymphonodular hyperplasia is a common finding in infants and young children who undergo endoscopy or radiographic studies of the intestinal tract. The etiology is unknown. It occurs frequently in children with food protein induced colitis, in whom it may be an abnormal finding at colonoscopy. Because lymphonodular hyperplasia disrupts the normal mucosa, it may potentially lead to mucosal thinning and predisposes to ulceration, which may cause hematochezia. Lymphonodular hyperplasia resolves spontaneously over time and is an unlikely source of bleeding in older children.

v Gastrointestinal duplication cyst — Gastrointestinal duplication cysts can be found at any level of the GI tract and frequently do not communicate with the bowel lumen. Gastric mucosa (present in approximately 50 percent of duplication cysts) can ulcerate, perforate, and form fistulas. Formation of a gastric duplicationcolonic fistula is unusual but can result in a lower gastrointestinal bleed. In addition, a duplication cyst that communicates with the intestine can result in bleeding into the GI tract. Gastrointestinal duplication cysts tend to present in infancy if they are symptomatic, but they may present in any age group, and often remain asymptomatic. v Infantile and very early onset inflammatory bowel disease — Rarely, inflammatory bowel disease (IBD) presents before six years of age.

Preschool period v. Anal fissures v. Intussusception v. Meckel's diverticulum

v Infectious colitis — A number of pathogens can cause LGIB in preschool children: Salmonella, Shigella, Campylobacter, E. coli 0157: H 7, and Clostridioides (formerly Clostridium) difficile are the most common. The diagnosis of an infectious etiology is made by isolating the organism from the stool or blood. Other ancillary tests such as occult blood, fecal leukocytes, fecal calprotectin, or fecal lactoferrin are nonspecific. v Hemolytic-uremic syndrome — HUS is characterized by the simultaneous occurrence of microangiopathic hemolytic anemia, thrombocytopenia, and acute renal injury. The highest rates are in children under the age of five years. Most cases are associated with a prodromal infection with an enteropathogen producing a shiga-like toxin, such as E. coli 0157: H 7, in which case diarrhea is a prominent feature and is frequently bloody.

v Ig. A vasculitis (Henoch-Schönlein purpura) — Immunoglobulin A vasculitis (Ig. AV; Henoch-Schönlein purpura [HSP]) is a systemic vasculitis of unclear etiology characterized by palpable cutaneous purpura, abdominal pain, and arthralgias. It is primarily a childhood disease that occurs between the ages of 3 and 15 years. Up to 50 percent of patients develop gross or occult gastrointestinal bleeding, and up to 50 percent develop renal disease. v Juvenile polyps — Juvenile polyps are benign hamartomas, which typically occur between the ages of 2 and 10 years, with a peak at 3 to 4 years. Patients usually present with painless rectal bleeding, with or without mucus; a few may have lower abdominal pain from traction on the polyp. Colonoscopy is the best way to diagnose polyps and permits their immediate, painless removal. If possible and safe, all polyps should be removed and evaluated by microscopy to confirm that they are juvenile hamartomas rather than adenomas.

v Solitary rectal ulcer syndrome — Solitary rectal ulcer syndrome (SRUS) is a benign but potentially chronic ulcerative disease of the rectum that is infrequent in childhood. It tends to present with bleeding, passage of mucus, straining during defecation, and a sense of incomplete evacuation. Treatment of associated constipation and strategies to avoid dysfunctional stooling or excessive straining may help alleviate this condition.

School-aged children and adolescents v. Anal fissures v. Ig. AV (HSP) v. Meckel's diverticulum v. Infectious colitis v. Juvenile v. Hemorrhoids (primarily in older adolescents) v. Solitary rectal ulcer syndrome (SRUS)

v Inflammatory bowel disease — Inflammatory bowel disease may present in preschool children and even in infancy but is more common in school-aged children and adolescents. IBD is comprised of two major disorders: ulcerative colitis (UC), which affects only the colon, and Crohn disease (CD), which can involve any portion of the gastrointestinal tract. The most common presenting symptoms of IBD are abdominal pain, fever, and diarrhea (with or without blood), occurring in about 80 percent of patients with CD; about 20 percent of patients with CD and about 95 percent of patients with UC will have visible rectal bleeding. Some patients with IBD may have extraintestinal symptoms, including anorexia, arthralgia, and erythema nodosum. The diagnosis is confirmed with imaging of the small bowel, upper endoscopy, colonoscopy, and biopsy.

DIAGNOSTIC APPROACH

v. History v. Physical examination v. Laboratory studies and imaging
- Slides: 34