Introduction to the Periodic Table Chapter 4 The
Introduction to the Periodic Table

Chapter 4 The Periodic Table

Who organized the Periodic table? • Why does it have the funky shape that it does? http: //www. privatehand. com/flash/elements. html http: //www. youtube. com/watch? v=LFsdb. LFHg. Y 8
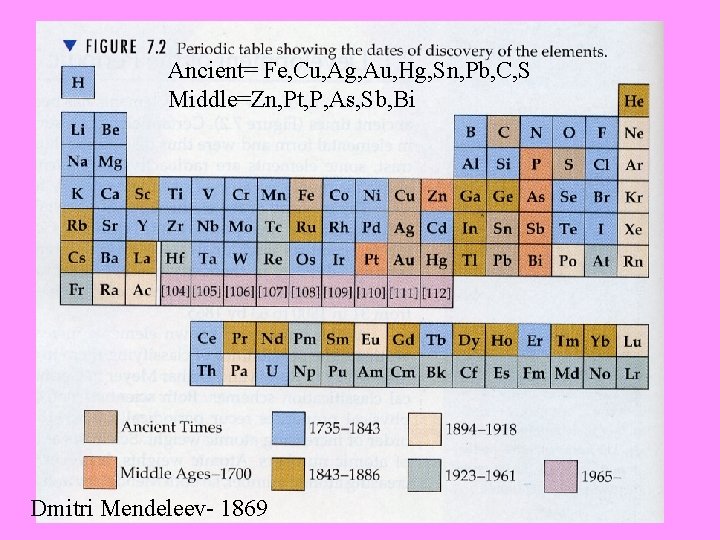
Ancient= Fe, Cu, Ag, Au, Hg, Sn, Pb, C, S Middle=Zn, Pt, P, As, Sb, Bi Dmitri Mendeleev- 1869

1834 -1907(published his table in 1869) Dmitri Mendeleev- http: //videos. howstuffworks. com/sciencechannel/27862 -100 -greatest-discoveries-the -periodic-table-video. htm

• Dmitri Mendeleev arranged the known elements into columns • These elements were arranged by similar properties in each row (I vs. Te) • However, the elements were ordered by increasing atomic mass because the atomic number was not known
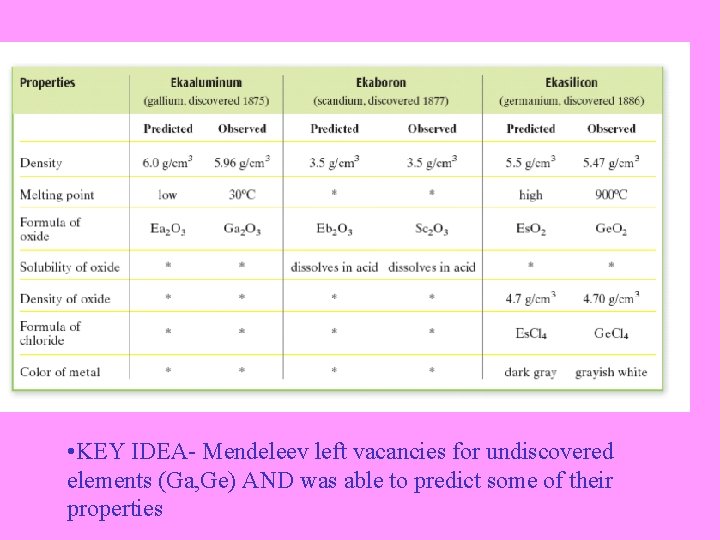
• KEY IDEA- Mendeleev left vacancies for undiscovered elements (Ga, Ge) AND was able to predict some of their properties

• Hence, Mendeleev is called the “Father of the Periodic Table” “I began to look about and write down the elements with their atomic weights and typical properties, analogous elements and like atomic weights on separate cards, and this soon convinced me that the properties of elements are in periodic dependence upon their atomic weights. ” --Mendeleev, Principles of Chemistry, 1905, Vol. II Periodic= occurring repeatedly at regular intervals, cyclic

Henry Moseley -British physicist (1887 -1915) There are 4 places on the table where the atomic number increases But the atomic mass decreases, find them. • Defined the atomic number • Arranged the table in increasing atomic number • Periodic law= properties of the elements are a function of their atomic numbers

The Modern Periodic Table and Glenn Seaborg (1912 -1999) Starting with his synthesis of plutonium(#94) in 1940, he created all of the transuranic elements from #94 to 102. He reconfigured the periodic table by placing the actinide series below the lanthanide series. Seaborg won the Nobel Prize in 1951. Element 106 has been named seaborgium (Sg) in his honor- the only person to have an element named for him/her while still living.

Before Seaborg After Seaborg

groups= columns rows = period
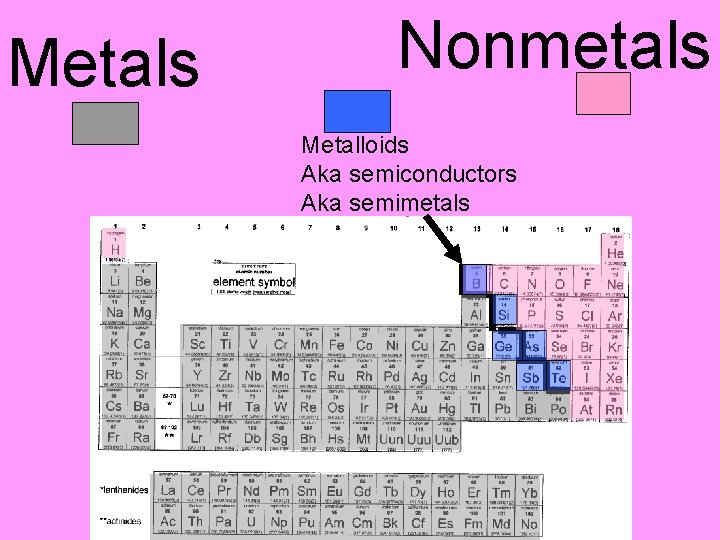
Metals Nonmetals Metalloids Aka semiconductors Aka semimetals

• Valence electrons: e- in the outermost shell of an atom and that determines the atom’s chemical properties. – Elements in each column (group) of the PT have the same number of e- in their outer energy level. – Elements with the same number of valence electrons tend to react in similar ways. – Because s and p electrons fill sequentially, the number of valence electrons in s- and pblock elements are predictable.

PROPERTIES OF: • METALS –Conduct heat and electricity –Low specific heat capacity! • NON-METALS –poor conductors of heat and electricity

PROPERTIES OF: • NON-METALS • METALS – Noble gases and N 2, O 2, F 2, Cl 2 are gases at – Can be malleable room temp and (rolled into pressure sheets) or ductile (squeezed into a – Br 2 is a liquid wire) – All other nonmetals – All are solids at are solids room temp and pressure except for Hg – Mixtures of metals=alloys

• Elements up to atomic number 92 are natural EXCEPT- At #85, and Fr#87 are natural but uncommon. Tc#43 and Pm#61 are not found in nature. • All atoms beyond atomic number 83 are unstable and radioactive.

Chemistry around town. Which allotrope of carbon is being served?


Most Elements Exist as Written on the Periodic Table EXCEPT the 7 DIATOMIC MOLECULES (two atoms)

• All halogens, no At • Nitrogen- N 2 • Oxygen-O 2 • Hydrogen - H 2

Some elements come in different forms • These forms are named allotropes. • Example- carbon exists as diamond, graphite, and C 60 is buckminsterfullerene (“soccer ball”). • See page 106

Buckeyballs • Co-discoverer of the buckeyball, Rice University professor and statesman for nanotechnology, Richard Smalley, died October 28, 2005 at the age of 62.
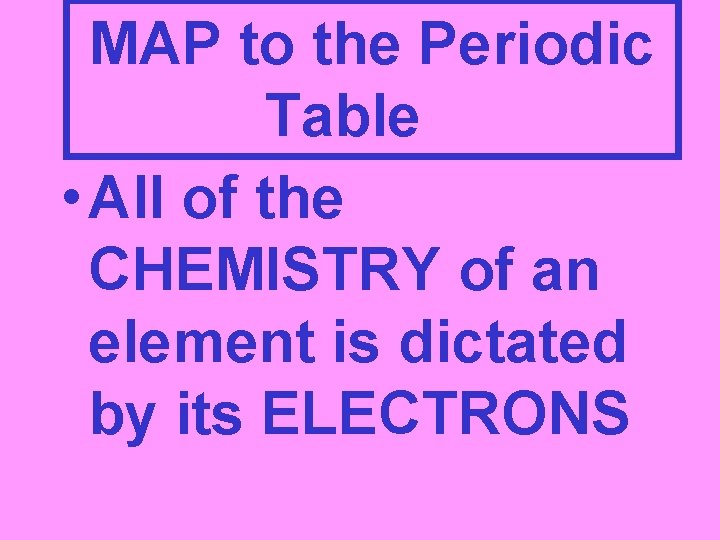
MAP to the Periodic Table • All of the CHEMISTRY of an element is dictated by its ELECTRONS

NOBLE GAS GAME • Elements want to lose or gain electrons to “resemble” the NOBLE GASES

A CHARGED Element is called an ION • Cation = positively charged ion • Anion = negatively charged ion
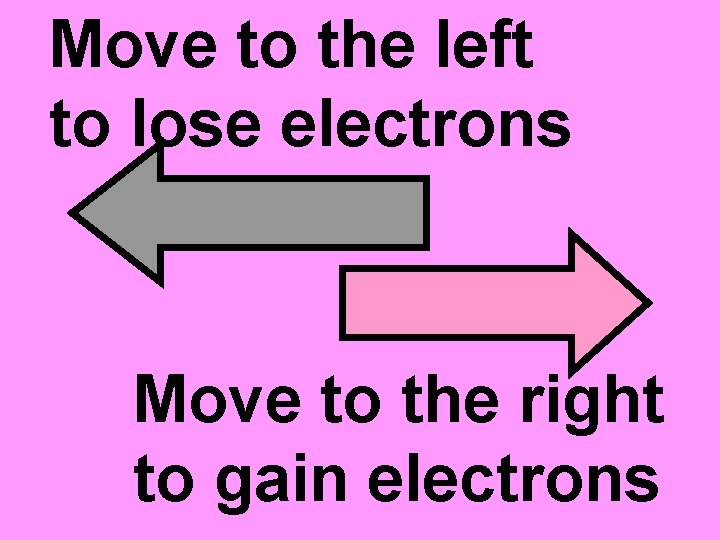
Move to the left to lose electrons Move to the right to gain electrons

Practice. What would the ionic charge be of: 1. Li 2. Mg 3. N 4. O 5. I 6. Ba
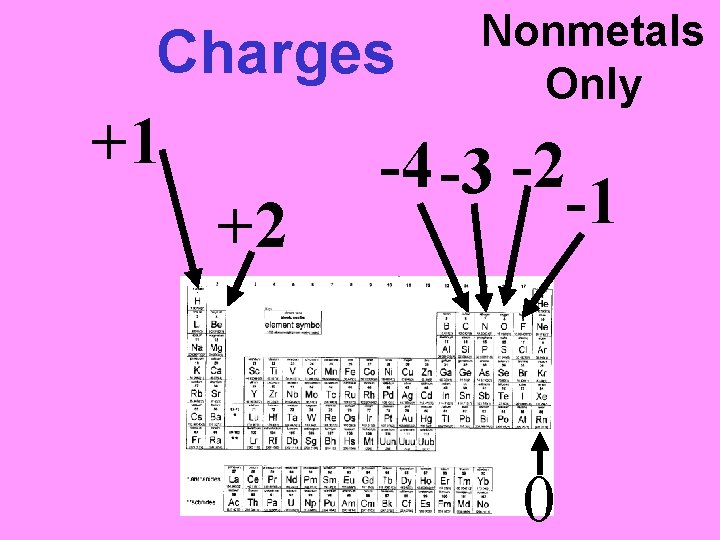
Charges +1 +2 Nonmetals Only -4 -3 -2 -1 0

METALS NONMETALS • most are gases or brittle solids • generally at room lustrous, temperature malleable, • Exception: solid Br(#35) is a • Exception: liquid Hg (#80) is a liquid

Families (or Groups) of the Periodic Table • • • First family- Alkali metals (NOT H) Second family - Alkali earth metals Last family - Noble gases Next to last family - Halogens Next to halogens- Chalcogens

Lanthanoid and Actinoid series • Named for the first element in each period • Color these ‘f’ block areas into your periodic table.
Representative vs. Transition Elements

Summary from Wednesday • Atomic size ______ down a family. • Atomic size _____ across a period. • Anions (negative ions) are always ______ than their parent atom. • Cations (positive ions) are always _____ than their parent atom.

• Elements - the simplest pure substance –Electrons - charges –Protons + charges –Neutrons no charge

What are atomic particles made of? • Sub-atomic particles named QUARKS –Quarks are held together with GLUONS

How are atoms organized? • Protons and neutrons are in the nucleus • Electrons outside • All elements are neutral
- Slides: 38