Introduction to the Periodic Table Atomic Number Symbol

Introduction to the Periodic Table Atomic Number ● Symbol ● Atomic Mass

I am Dmitri Mendeleev! I made the PERIODIC TABLE !

Mendeleev u Late 1800’s u Arranged table by atomic mass u Since chemical properties repeated periodically, he called the table the Periodic Table.

Dmitri Mendeleev: Father of the Table HOW HIS WORKED… u Put elements in rows by increasing atomic mass. u Put elements in columns by the way they reacted. SOME PROBLEMS… u left blank spaces for undiscovered elements. (Turned out he was right!) u broke the pattern of increasing atomic weight to keep similar reacting elements together.

Weird!!! u Mendeleev left blank spaces in his table to keep the elements lined up according to their chemical properties. u He predicted properties and mass numbers of elements yet to be discovered. u And later, THEY WERE!!!! Creepy!


The Current Periodic Table u Mendeleev wasn’t too far off. But there was room for improvement! u Mass doesn’t always increase from left to right in Mendeleev's table. u Now the elements are put in rows by increasing atomic numbers.

Henry Moseley u 1913 u British u Corrected Mendeleev’s table by arranging elements by atomic number.

Glenn T. Seaborg co-discovered 10 new elements u in 1944 he moved 14 elements out of the main body of the periodic table to their current location below the Lanthanide series and Actinide series. u

Glenn T. Seaborg He is the only person to have an element named after him while still alive.


What is the PERIODIC TABLE? o Shows all known elements in the universe. o Organizes the elements by chemical properties.
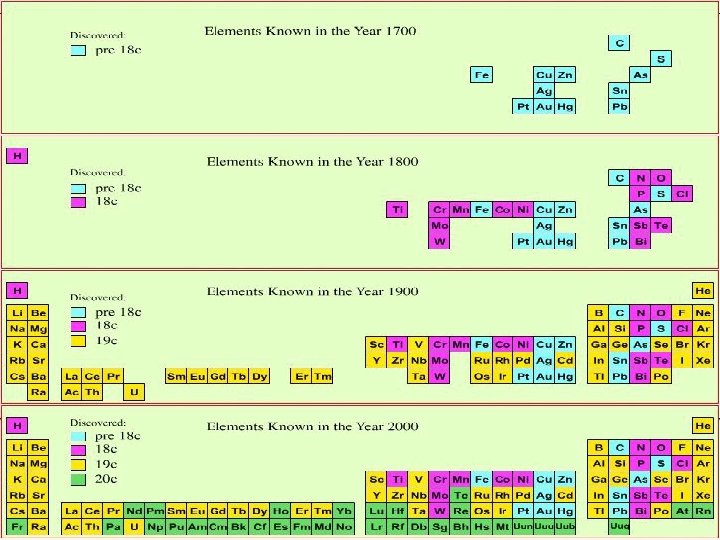
Elements by Discovery Year

How do you PERIODIC read the TABLE?
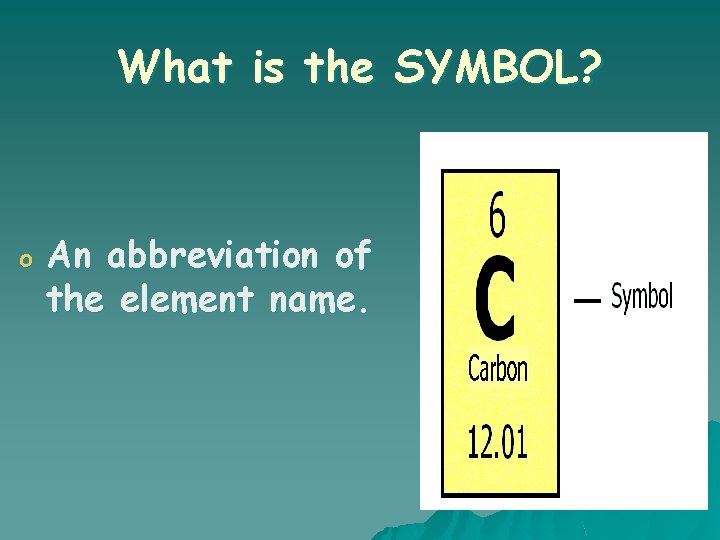
What is the SYMBOL? o An abbreviation of the element name.

What is the ATOMIC WEIGHT? o The number of protons and neutrons in the nucleus of an atom.

What is the ATOMIC NUMBER? o o The number of protons found in the nucleus of an atom Or The number of electrons surrounding the nucleus of an atom.

Again, how do I find the number of protons, electrons, and neutrons in an element using the periodic table? o # of PROTONS = ATOMIC NUMBER o # of ELECTRONS = ATOMIC NUMBER o # of NEUTRONS = ATOMIC _ ATOMIC WEIGHT NUMBER

Atoms, Electrons & Energy Levels Electrons are the bonds that hold the world together!

Periodic Law: When elements are arranged in order of increasing atomic number, there is a periodic pattern in their physical and chemical properties.
Periodic Table Navigation

The Current Periodic Table u The modern periodic table is organized into periods and families. u The horizontal rows are called periods and are labeled from 1 to 7. u The vertical columns are called families or groups and are labeled from 1 to 18.

Periods u The horizontal rows on the Periodic Table are called periods. u Each row is its own period. u Elements in the same period increase by atomic number. u There are seven periods on the Periodic Table.
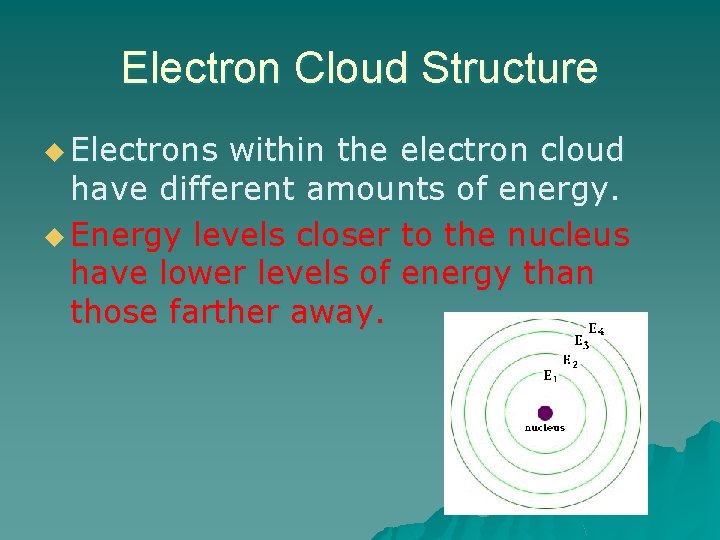
Electron Cloud Structure u Electrons within the electron cloud have different amounts of energy. u Energy levels closer to the nucleus have lower levels of energy than those farther away.

These energy levels are named using numbers 1 – 7. u The period the element is in determines the number of energy levels the element will have. u A complete and stable energy level will contain a maximum of eight electrons. More on this later…don’t freak! u Levels
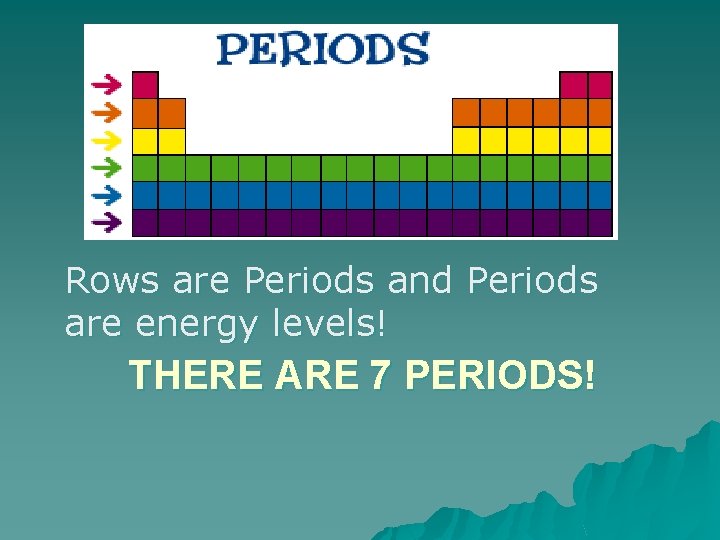
Rows are Periods and Periods are energy levels! THERE ARE 7 PERIODS!
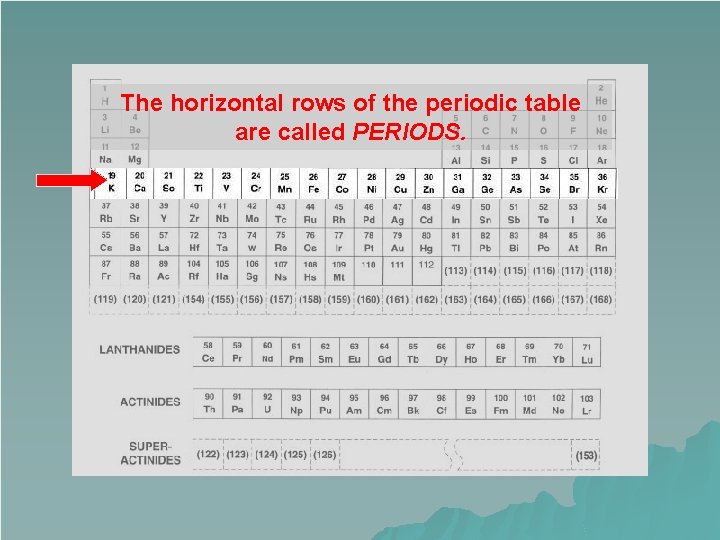
The horizontal rows of the periodic table are called PERIODS.
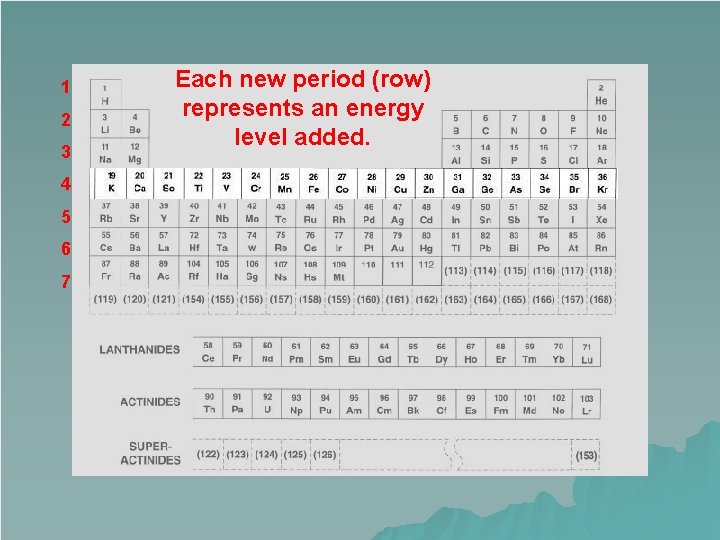
1 2 3 4 5 6 7 Each new period (row) represents an energy level added.

Periods u Do you see that the elements increase by one electron and one proton as you move across the table, left to right?

Groups u Vertical columns on the Periodic Table are known as groups or families. u There are 18 groups on the Periodic Table

u Members of the same group have many similarities. – Physical and chemical properties – Valence electrons (hang on…I’m getting there!) – Oxidation numbers (I’m getting here too!) – Trends

Valence and Oxidation Number u OK, since you could not wait… Valence is the number of electrons that an element has in its outermost energy level. NO questions now! u Oxidation number is the number of electrons that an element gains or loses when it forms a bond. Guess what? NO questions now either!
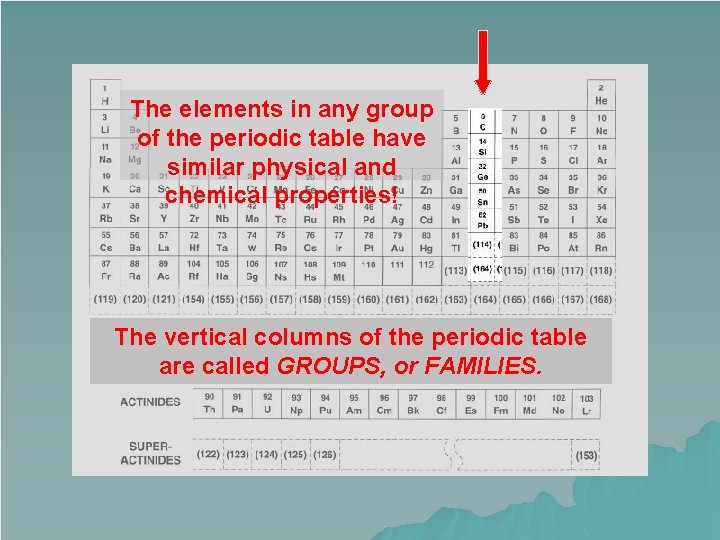
The elements in any group of the periodic table have similar physical and chemical properties! The vertical columns of the periodic table are called GROUPS, or FAMILIES.

1 2 13 14 15 16 17 18 The GROUP provides information about the number of valence electrons.

Periodic Table Trends

Metal trivia u Au was the first metal used, 6000 years ago. Ag and Cu followed a few thousand years later. Al not until the 1800’s.

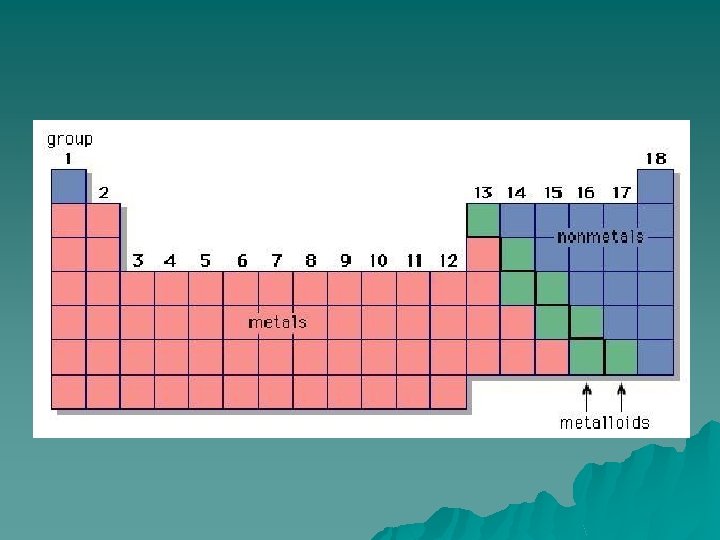
Metals u Metals are found to the left of the stair-step line. u Tend to give up (lose) electrons. u Like to combine with non-metals to form ionic bonds. (wait…more later)

Metals • Usually have 1 – 3 valence electrons • Good conductors of heat and electricity • Malleable – can be flattened into sheets • Ductile – can be pulled into a wire • Shiny • Solid at room temperature (except Mercury)
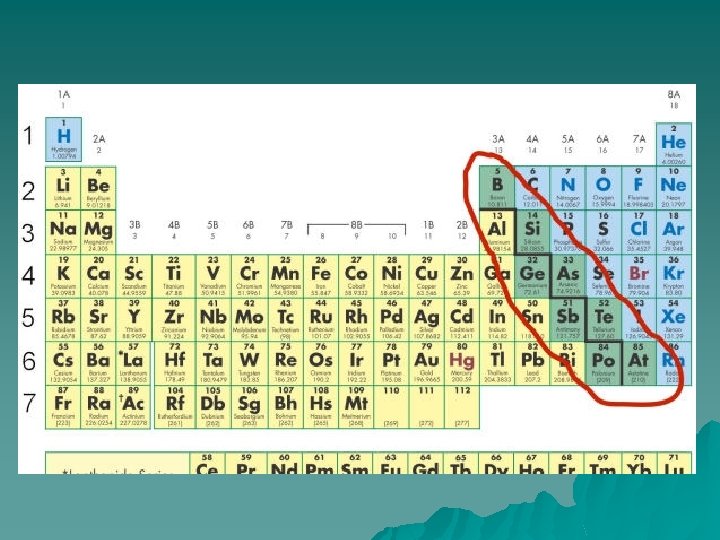

Nonmetals u Found to the right of the stair-step line. u Only the noble gases are all nonmetals.
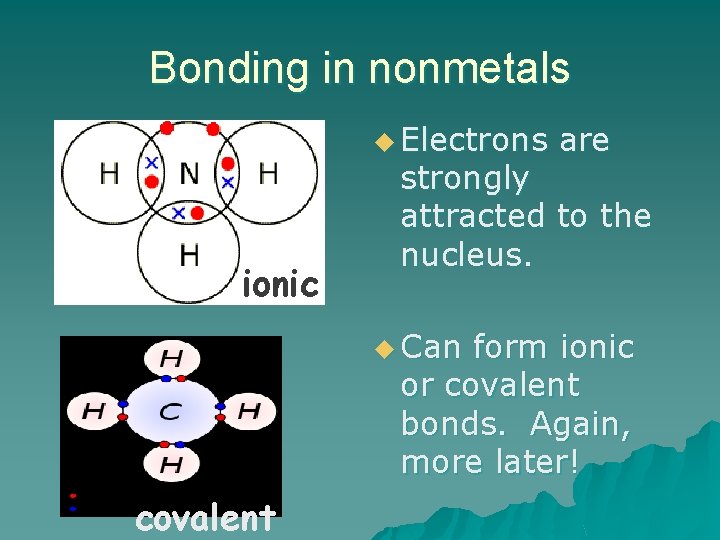
Bonding in nonmetals u Electrons ionic are strongly attracted to the nucleus. u Can form ionic or covalent bonds. Again, more later! covalent

Nonmetals u Usually have 4 – 8 valence electrons u Can be a solid, liquid, or gas at room temperature u Brittle (if a solid), so not ductile or malleable u Dull, not shiny u Poor conductors of heat and electricity
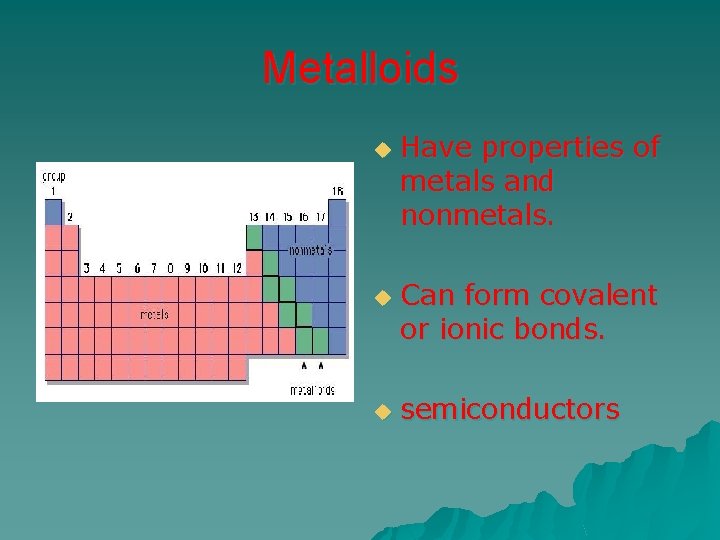
Metalloids u u u Have properties of metals and nonmetals. Can form covalent or ionic bonds. semiconductors

Metalloids Exhibit characteristics of metals and nonmetals u Semi-conductors – conduct electricity under special conditions u u The metalloids are: – Boron – Silicon – Germanium – Arsenic – Antimony – Tellurium – Polonium – Astatine
Families

Hydrogen u Makes up 99% of the universe. u Most H on Earth is found in water. u The word hydrogen means “water forming”. u Can be diatomic H 2

Hydrogen – the weird kid u Commonly has an oxidation number of +1 and one valence electron, so it is grouped with the alkali metals. u But its natural state is a gas, so it really isn’t an alkali metal. u Sometimes has an oxidation number of -1, so it is occasionally grouped with group 17 – the halogens. u Happy now?
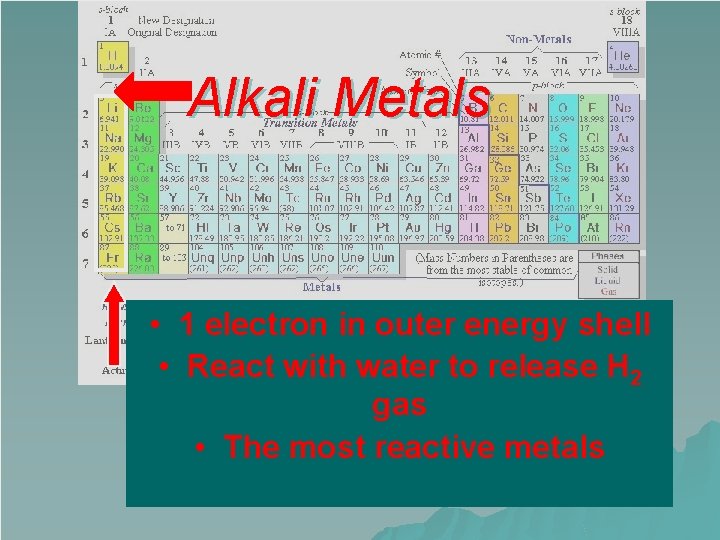
Alkali Metals • 1 electron in outer energy shell • React with water to release H 2 gas • The most reactive metals

Group 1 – Alkali Metals Very reactive elements u Rarely found on their own in nature u 1 valence electron u +1 oxidation number u u The alkali metals are: – Lithium – Sodium – Potassium – Rubidium – Cesium – Francium
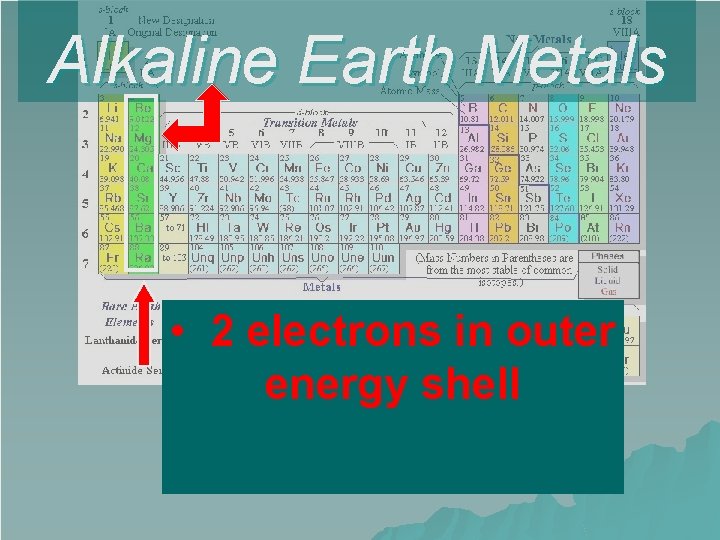
Alkaline Earth Metals • 2 electrons in outer energy shell

Alkaline Earth Metals u Group 2 u Lose electrons and become positive u Fireworks! Sr = red, Mg = white, Ba = green u Medical uses

Group 2 – Alkaline Earth Metals Very reactive (but not as reactive as the Alkali Metals) u Not found freely in nature u 2 valence electrons u + 2 oxidation number u u The Alkaline Earth Metals are: – Beryllium – Magnesium – Calcium – Strontium – Barium – Radium

Transition Metals • Generally 2 electrons in outer energy level • Commonly have more than one oxidation number • Form compounds that are brightly colored

Groups 3 -12 The Transition Metals u All metals, so exhibit the properties of metals u No set number of valence electrons or oxidation numbers u Often found in compounds, but can be solitary

Transition cont. Iron, cobalt, and nickel – only known elements to produce a magnetic field … called the iron triad u Used to make alloys u

Transition cont. u Copper, silver and gold called the coinage metals. Too expensive to use now. Zinc and cadmium are used to make batteries and plate things. u Mercury is toxic but used in thermometers. u

Lanthanide/ Actinide Series • 2 electrons in outer energy level • Primary oxidation number is +3 • Lanthanide series: soft, malleable metals with high luster and conductivity • Actinide series: All radioactive; most humanmade
Rare Earth Metals Also called the inner transition metals u They were moved below the table simply for space. u Lanthanides and Actinides Series u Fit within periods 6 &7 u

Lanthanides and Actinides Series Lanthanides u Used in the film industry and to make TV screen colors. u Elements 58 -71. Actinides u Elements 90 – 103 u All are radioactive and unstable u Can be used in nuclear reactors and weapons

Halogens • 7 electrons in their outer energy level • Easily combine with metals to form salts • Most reactive of all nonmetals

Group 17 – The Halogens u “Halogen” means “salt-former” – compounds containing halogens are known as “salts” u 7 valence electrons u - 1 oxidation number u The halogens exist in all states of matter: – Solid- Iodine, Astatine – Liquid- Bromine – Gas- Fluorine, Chlorine

Noble Gases • Full outer energy level • Almost completely inactive (“inert”) • All are colorless gases • Argon is the most abundant, making up almost 1% of air

Group 18 – The Noble Gases Stable elements u Do not form compounds in nature (Inert) u Full outermost orbital u – Helium has 2 – All the others have 8 u 0 oxidation number u The Noble Gases are – Helium – Neon – Argon – Krypton – Xenon – Radon

The Kinda Special Groups u. Group Family 13 – The Boron § 3 valence electrons, + 3 oxidation number

u. Group Family 14 – The Carbon § 4 valence electrons, ± 4 oxidation number

u. Group 15 – The Nitrogen Group § 5 valence electrons, - 3 oxidation number

u. Group Family 16 – The Oxygen § 6 valence electrons, - 2 oxidation number
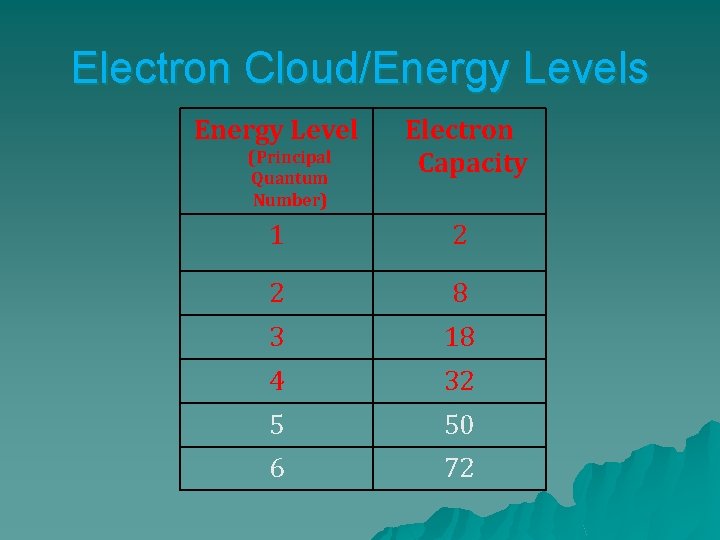
Electron Cloud/Energy Levels Energy Level (Principal Quantum Number) Electron Capacity 1 2 2 8 3 18 4 32 5 50 6 72
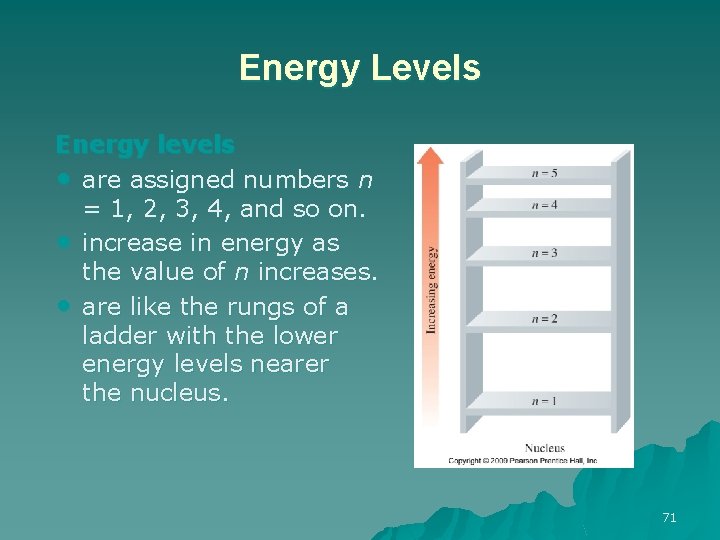
Energy Levels Energy levels • are assigned numbers n = 1, 2, 3, 4, and so on. • increase in energy as the value of n increases. • are like the rungs of a ladder with the lower energy levels nearer the nucleus. 71

Energy Levels Energy levels have a maximum number of electrons equal to 2 n 2. Energy level electrons n=1 n=2 n=3 Maximum number of 2(1)2 2(2)2 2(3)2 = = = 2(1) 2(4) 2(9) = 2 = 8 = 18 72

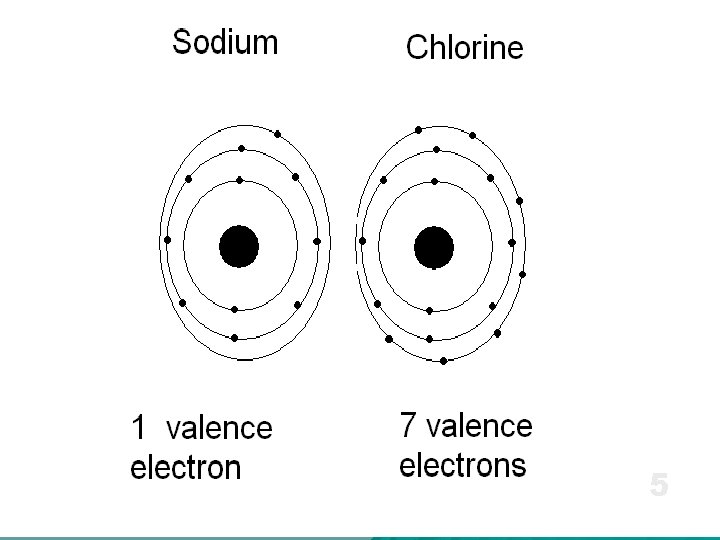
Valence Electrons u When an atom undergoes a chemical reaction, only the outermost electrons are involved.

u Valence electrons are the electrons in the outer energy level of an atom. u These are the electrons that are transferred or shared when atoms bond together. Valence Electrons

Lost/Gained Electrons that are gained or lost are from an elements’ outermost energy level. These electrons are called valence electrons. To determine the number of valence electrons an element has, you must look on the periodic table!

Predicting Valence Electrons u The Roman numeral in the American convention indicates the number of valence electrons. – Group IA elements have 1 valence electron – Group VA elements have 5 valence electrons

u When using the IUPAC designations for group numbers, the last digit indicates the number of valence electrons. – Group 14 elements have 4 valence electrons – Group 2 elements have 2 valence electrons

The big point… VALENCE ELECTRONS ARE WHERE THE ACTION IS AT SINCE THEY ARE THE ONLY ONES THAT ARE INVOLVED IN CHEMICAL REACTIONS!
5
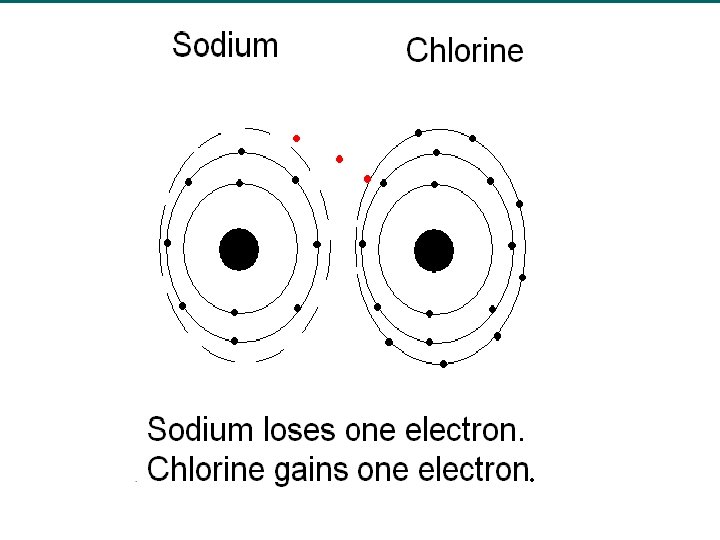
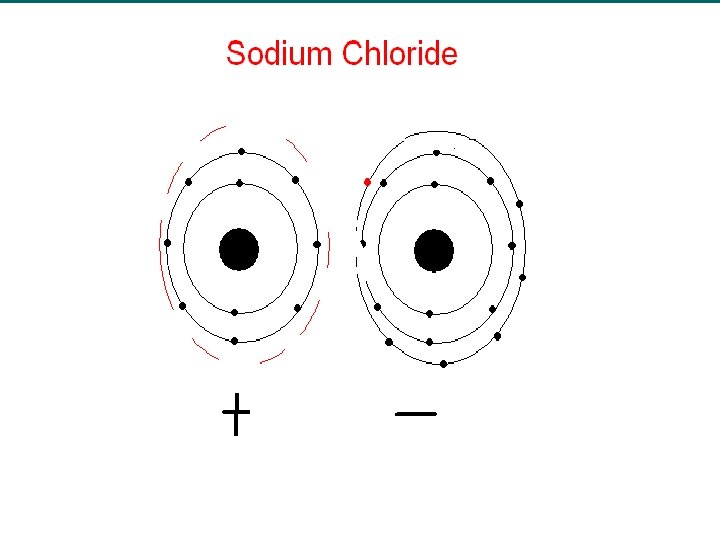

Ionic Charge u Recall, that atoms lose or gain electrons to form ions. u The charge of an ion is related to the number of valence electrons on the atom. u Group IA/1 metals lose their one valence electron to form 1+ ions. – Na → Na+ + e- u Metals lose their valence electrons to form ions.

Who loses?

Who gains?

So what about all of the other electrons?

u No one cares because only the electrons in the outer most level are involved with forming a compound.
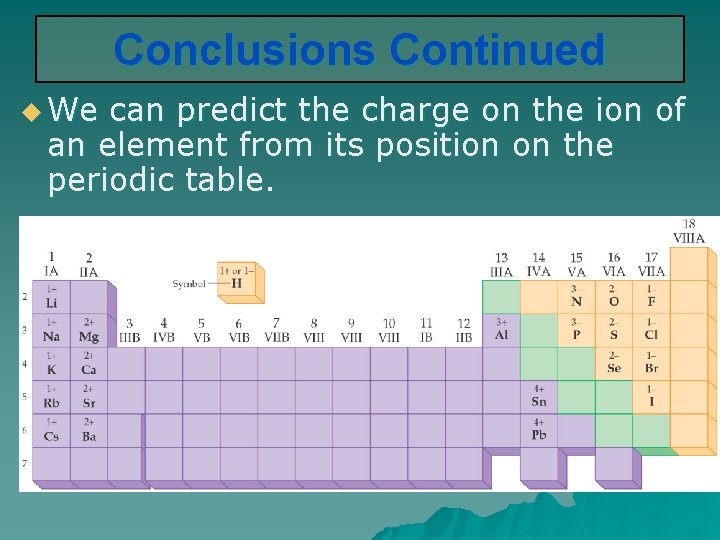
Conclusions Continued u We can predict the charge on the ion of an element from its position on the periodic table.


Electrons Remember, the only thing that can move to change the charge on an atom is an electron! Cathode ray tube – Electrons moving! Sweet!

Ions An ion is an element or compound with a charge.

u. Anion: ion that gains electrons to have a negative charge u. Cation: it has lost electrons to have a positive charge

What motivates an atom to become an ion? Ø Ø The ultimate goal of each element is to have the same number of electrons as the NEAREST noble gas an element can either gain an electron(s) or lose an electron(s) to reach this goal

Ions • Change in electrons which gives an atom a charge (+ or -) • You can only add or subtract electrons! (protons don’t change) – Ex. Count the number of electrons below… Carbon ion (-1 charge) 7 electrons (-) 6 protons (+) Neutral Carbon 6 electrons (-) 6 protons (+) Carbon ion (+1 charge) 5 electrons (-) 6 protons (+)
- Slides: 93