Introduction to the Periodic Table Atomic Number Symbol

Introduction to the Periodic Table Atomic Number ● Symbol ● Atomic Weight Element ● Compound ● Mixture

1/2/19 Objective: You will identify patterns in the Periodic Table Vocabulary: Atomic Number ● Symbol ● Atomic Weight Element ● period ● group Do NOW: 1. Copy HW- Finish periodic table SNB entry 2. Glue Introducing the Periodic table onto your next clean INPUT page- make sure you have a blank OUTPUT page facing it. 3. Update your TOC & add the vocabulary to your index using your OUTPUT page #

Elements u u Science has come along way since Aristotle’s theory of Air, Water, Fire, and Earth. Scientists have identified 90 naturally occurring elements, and created about 28 others.

Elements u The elements, alone or in combinations, make up our bodies, our world, our sun, and in fact, the entire universe.

I am Dmitri Mendeleev! I developed the PERIODIC TABLE !

What is the PERIODIC TABLE? o Shows all known elements in the universe. o Organizes the elements by chemical properties.
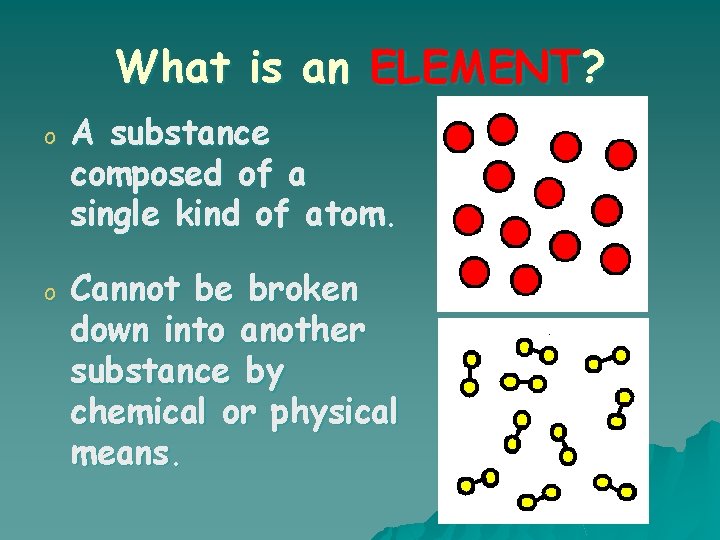
What is an ELEMENT? o A substance composed of a single kind of atom. o Cannot be broken down into another substance by chemical or physical means.
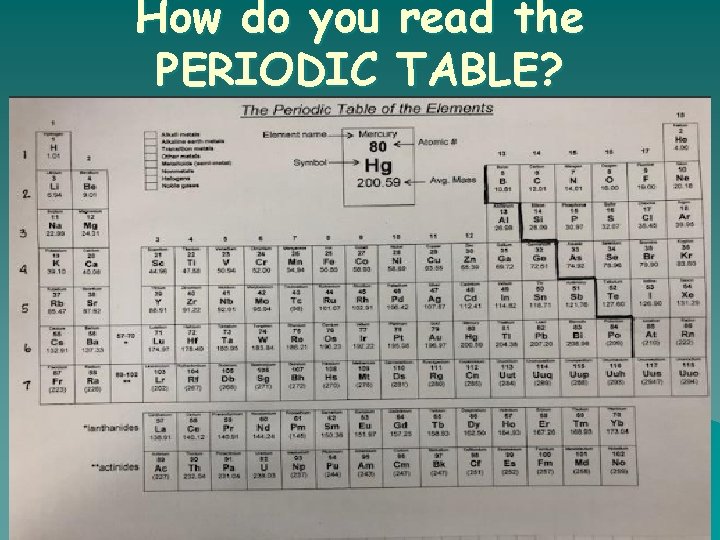
How do you read the PERIODIC TABLE?

Look inside the box

How do you PERIODIC read the TABLE?

What is the ATOMIC NUMBER? o o The number of protons found in the nucleus of an atom Or The number of electrons surrounding the nucleus of an atom.
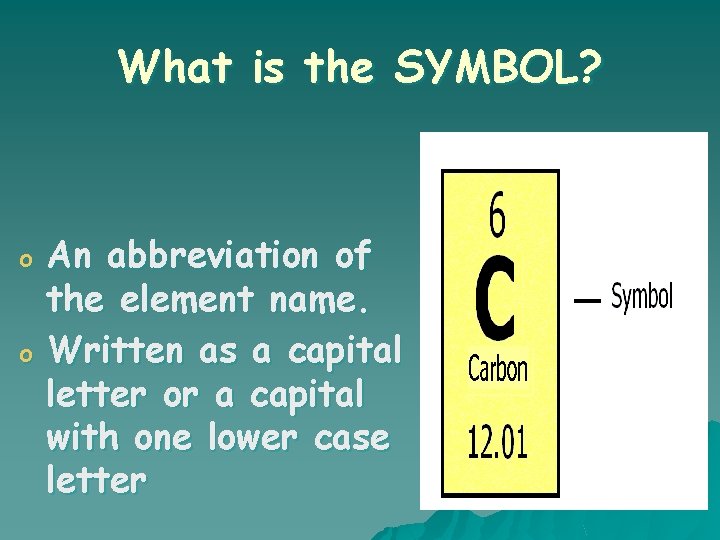
What is the SYMBOL? o o An abbreviation of the element name. Written as a capital letter or a capital with one lower case letter

What is the ATOMIC WEIGHT? o The number of protons and neutrons in the nucleus of an atom.

Patterns in the PERIODIC TABLE
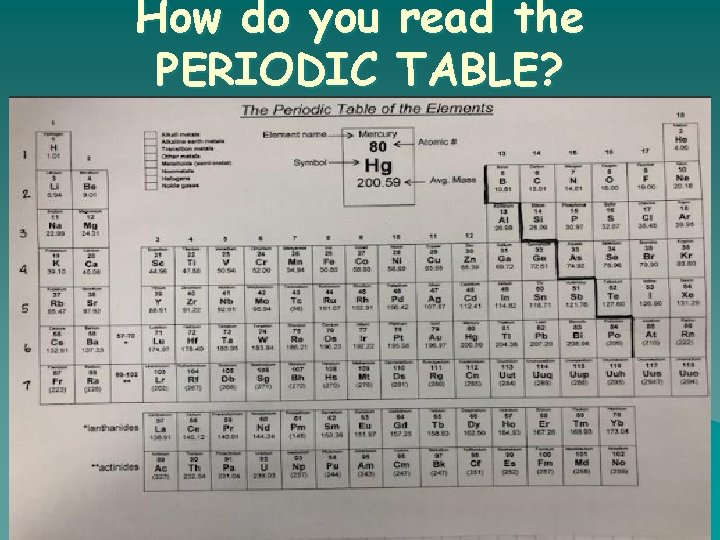
How do you read the PERIODIC TABLE?




How do I find the number of protons, electrons, and neutrons in an element using the periodic table? o # of PROTONS = ATOMIC NUMBER o # of ELECTRONS = ATOMIC NUMBER o # of NEUTRONS = ATOMIC _ ATOMIC WEIGHT NUMBER

1/4/19 Objective: You will identify the difference between an element, compound, and a mixture Vocabulary: Element ● Compound ● Mixture

Elements, Compounds, and Mixtures

What is a COMPOUND? o A substance in which two or more different elements are CHEMICALLY bonded together.

What is a MIXTURE? o Two or more substances that are mixed together but are NOT chemically bonded.

Element, Compound or Mixture?
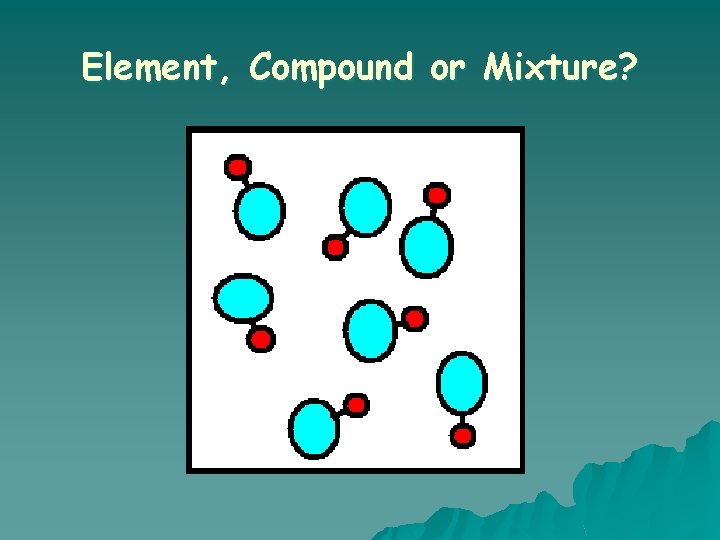
Element, Compound or Mixture?

Element, Compound or Mixture?

Element, Compound or Mixture?
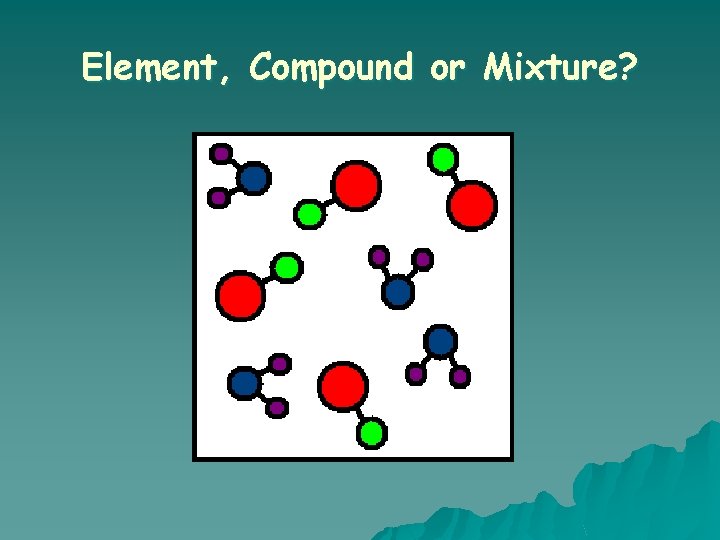
Element, Compound or Mixture?

Element, Compound or Mixture?
- Slides: 29