Introduction to the Mole Background When you buy



Introduction to the Mole

Background • When you buy eggs you usually ask for a _______ eggs. • You know that one dozen of any item is ______.

Paper • Paper is packaged by a ream. • A ream of paper has 500 sheets.

• Why is it useful to use units like a dozen or a ream? • What determines how many items should make up a particular unit?

One MOLE of anything has 602, 200, 000, 000 items.

AVOGADRO’S NUMBER • One MOLE of anything has 6. 02 x 23 10 items. –Items can be particles, atoms, molecules, etc.

Converting Moles and Particles • • Write down what you are given. Set up a conversion factor to cancel units. 1 mole = 6. 02 x 1023 ______ Reminder: Units must be diagonal from each other in order to cancel!
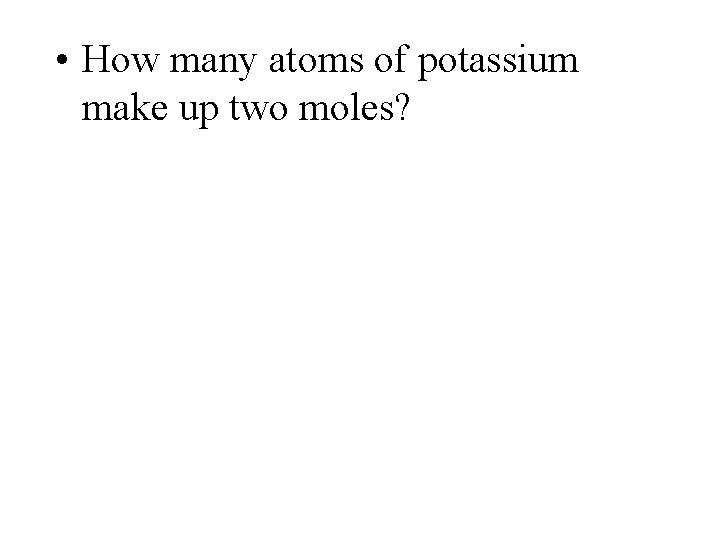
• How many atoms of potassium make up two moles?
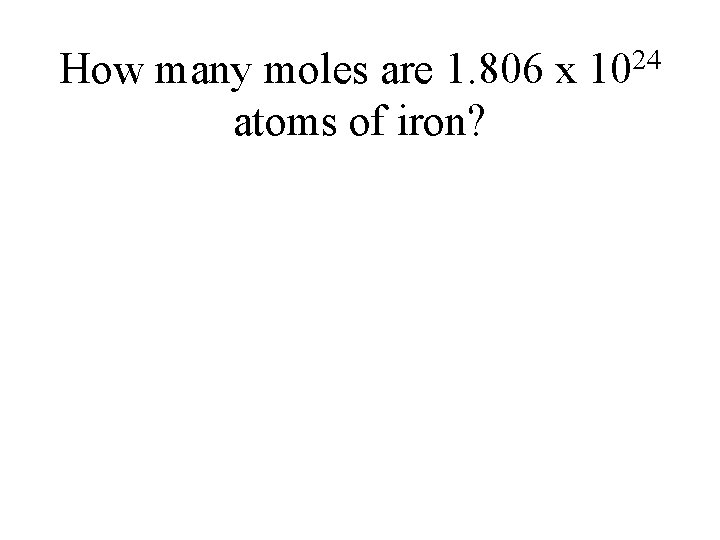
How many moles are 1. 806 x 1024 atoms of iron?

• How many hydrogen atoms are in 2. 5 moles of water?

• On the periodic table, the atomic mass given is the mass (in grams) of one mole of that atom. – Sodium (Na) Molar Mass = 22. 99 g – Magnesium (Mg) = ____ g

Gram Formula Mass • The mass of one mole of a substance • Sodium • Potassium carbonate • Barium hydroxide

• How many years would it take you to count to Avogadro’s Number if you counted one number per minute?
- Slides: 15