Introduction to the Human Body Tenth Edition Gerard

Introduction to the Human Body Tenth Edition Gerard J. Tortora • Bryan H. Derrickson Chapter 2 Introductory Chemistry Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Introductory Chemistry n Introduction to chemistry q q n Chemical elements and atoms Ions, molecules, and compounds Chemical bonds Chemical reactions Chemical compounds and life processes q q Inorganic compounds Organic compounds Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

INTRODUCTORY CHEMISTRY Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Elements and Atoms n n Chemistry is the science of the structure and interactions of matter, which is anything that occupies space and has mass. All forms of matter are made up of a limited number of building blocks called chemical elements, substances that cannot be broken down into a simpler form. Each element is made up of atoms, the smallest units of matter that retain the properties and characteristics of the element. Examples are hydrogen, carbon, oxygen. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

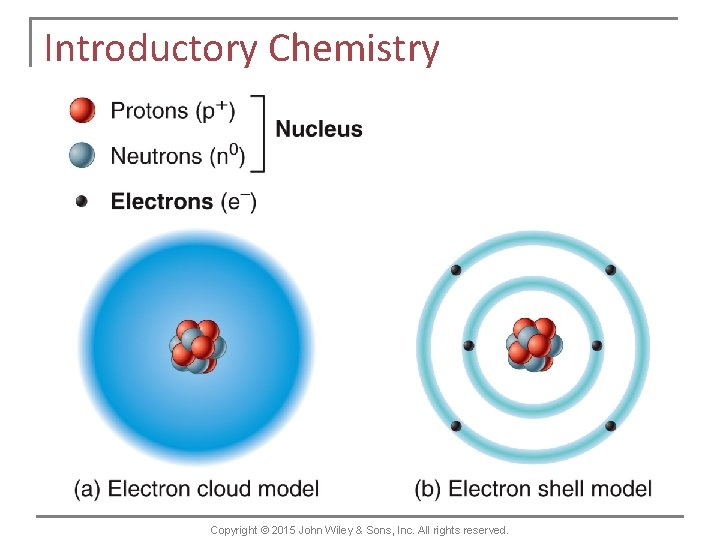
Chemical Elements and Atoms n n n An atom consists of two basic parts: a nucleus and one or more electrons. The centrally located nucleus contains positively charged protons and uncharged (neutral) neutrons. Because each proton has one positive charge, the nucleus is positively charged. The electrons are tiny, negatively charged particles that move about in a large space surrounding the nucleus. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Elements and Atoms Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Elements and Atoms n The number of protons in the nucleus of an atom is called the atom’s atomic number. n A hydrogen atom has 1 proton, a carbon atom has 6 protons, a sodium atom has 11 protons, etc. n Electrons move about within certain regions around the nucleus. These regions are called electron shells. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Elements and Atoms n The electron shell nearest the nucleus (the first electron shell) can hold a maximum of 2 electrons. n The second electron shell can hold a maximum of 8 electrons. n The third can hold up to 18 electrons. n Higher electron shells (there as many as seven) can contain many more electrons. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.
Introductory Chemistry Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Introductory Chemistry Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Ions, Molecules, and Compounds n n n The atoms of each element have a characteristic way of losing, gaining, or sharing their electrons when interacting with other atoms. If an atom either gives up or gains electrons, it becomes an ion, which is an atom that has a positive or negative charge due to unequal numbers of protons and electrons. When two or more atoms share electrons, the result is called a molecule. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Ions, Molecules, and Compounds n n n A molecular formula indicates the number and type of atoms that make up a molecule. For example, the molecular formula for a molecule of oxygen is O 2. A compound is a substance containing atoms of two or more different elements. A water molecule, H 2 O, one atom of oxygen shares electrons with two atoms of hydrogen. A free radical is an ion or molecule that has an unpaired electron in its outermost shell. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Introductory Chemistry Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Bonds n n n The forces that bind the atoms of molecules and compounds together, resisting their separation, are chemical bonds. This depends on the number of electrons in its outermost shell, also called the valence shell. Atoms with an outer shell holding 8 electrons is chemically stable, which means it is unlikely to form chemical bonds with other atoms (Octet rule). Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Bonds n n Ionic Bonds – the force of attraction between ions of opposite charges. Positively charged ions and negatively charged ions are attracted to one another. The sodium atom becomes a cation (Na+), a positively charged ion, when it loses its one valence shell electron to chlorine. The chlorine atom becomes an anion (Cl–), a negatively charged ion, when it gains an electron from sodium. The result is Na. Cl, an ionic compound. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Introductory Chemistry Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Bonds n n n Covalent bonds – the atoms form a molecule by sharing one, two, or three pairs of their outer shell electrons. Covalent bonds are the most common chemical bonds in the body, and the compounds that result from them form most of the body’s structures. Unlike ionic bonds, most covalent bonds do not break apart when the molecule is dissolved in water. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Introductory Chemistry Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Bonds n Hydrogen Bonds – the polar covalent bonds that form between hydrogen atoms and other atoms give rise to this third type of bond. n Hydrogen bonds result from attraction of oppositely charged parts of molecules rather than from sharing of electrons as in covalent bonds. These bonds are weak when compared to ionic and covalent bonds. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Animation • Chemical Bonding You must be connected to the internet to run this animation. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Animation • Polarity and Solubility of Molecules You must be connected to the internet to run this animation. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Reactions n n n A chemical reaction occurs when new bonds form and/or old bonds break between atoms. Through chemical reactions, body structures are built and body functions are carried out, processes that involve transfers of energy. Energy is the capacity to do work. The two main forms of energy are potential energy, energy stored by matter due to its position, and kinetic energy, the energy of matter in motion. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Reactions n n n When two or more atoms, ions, or molecules combine to form new and larger molecules, the process is a synthesis reaction (“to put together”). All of the synthesis reactions that occur in your body are collectively referred to as anabolism. Combining simple molecules like amino acids to form large molecules such as proteins is an example of anabolism. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Reactions n n n In a decomposition reaction, a molecule is split apart. The word decompose means to break down into smaller parts. Large molecules are split into smaller molecules, ions, or atoms. This is called catabolism. Breakdown of large starch molecules into many small glucose molecules during digestion is an example of catabolism. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Chemical Reactions n Reversible reactions can go in either direction. n Some chemical reactions proceed in only one direction, as discussed in synthesis and decomposition reactions. These chemical reactions may be reversible. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Animation • Types of Reactions and Equilibrium You must be connected to the internet to run this animation. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

CHEMICAL COMPOUNDS AND LIFE PROCESSES Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds n n n Chemicals in the body can be divided into two main classes of compounds: inorganic and organic. Inorganic compounds usually lack carbon, are structurally simple, and are held together by ionic or covalent bonds. They include water, many salts, acids, and bases. Organic compounds, by contrast, always contain carbon, usually contain hydrogen, and always have covalent bonds. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds: Water n Water is the most important and most abundant inorganic compound in all living systems, making up 55 to 60% of body mass in lean adults. q q q Water is an excellent solvent Water participates in chemical reactions Water absorbs and releases heat very slowly Water requires a large amount of heat to change from a liquid to a gas Water serves as a lubricant Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds: Acids, Bases, and Salts n Acids are a substance that break apart or dissociate into one or more hydrogen ions (H+) when it dissolves in water. n Bases usually dissociate into one or more hydroxide ions (OH–) when it dissolves in water. n Salts, when dissolved in water, dissociate into cations and anions, neither of which is H+ or OH–. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds: Acid-Base Balance: p. H n n n To ensure homeostasis, body fluids must contain almost balanced quantities of acids and bases. The chemical reactions that take place in the body are very sensitive to even small changes in the acidity or alkalinity of the body fluids in which they occur. Any departure from the narrow limits of normal H+ or OH– concentrations greatly disrupts body functions. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds: Buffer Systems n Homeostatic mechanisms maintain the p. H of blood between 7. 35 and 7. 45, so that it is slightly more basic than pure water. n Buffers are chemical compounds that act quickly to temporarily bind H+, removing the highly reactive, excess H+ from solution but not from the body. n Buffers prevent rapid, drastic changes in the p. H of a body fluid by converting strong acids and bases into weak acids and bases. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Inorganic Compounds Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Animation • Acids and Bases You must be connected to the internet to run this animation. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Carbohydrates n n Carbohydrates are organic compounds that contain carbon, hydrogen, and oxygen and include sugars, glycogen, starches, and cellulose. They are divided into three major groups: q q q Monosaccharides - simple sugars (glucose) Disaccharides - simple sugars (two monosaccharides linked together) Polysaccharides - large, complex carbohydrates that contain tens or hundreds of monosaccharides joined through dehydration synthesis reactions. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Sucrose Molecule Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Glycogen Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Lipids n n Lipids are hydrophobic, that is, they are insoluble in water. They contain carbon, hydrogen, and oxygen. The diverse lipid family includes: q q Triglycerides (fats and oils) Phospholipids (lipids that contain phosphorus) Steroids Fat-soluble vitamins (vitamins A, D, E, and K) Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Triglycerides Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Phospholipids Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Steroids Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Proteins n Proteins are large molecules that contain carbon, hydrogen, oxygen, and nitrogen; some proteins also contain sulfur. n Much more complex in structure than carbohydrates or lipids, proteins have many roles in the body and are largely responsible for the structure of body cells. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Proteins n n Amino acids are the building blocks of proteins. All amino acids have an amino group (— NH 2) at one end a carboxyl group (— COOH) at the other end. Each of the 20 different amino acids has a different side chain (R group). The covalent bonds that join amino acids together to form more complex molecules are called peptide bonds. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Amino Acids Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Enzymes Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Anatomy Overview • Common Biomolecules You must be connected to the internet to run this animation. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: Nucleic Acids n DNA (deoxyribonucleic acid) – provides the blueprint of life and contains: q n RNA (ribonucleic acid) – intermediary between DNA and protein that contains: q n Nucleotide bases: A=Adenine, G=Guanine, C=Cytosine, T=Thymine Nucleotide bases: A=Adenine, G=Guanine, C=Cytosine, U=Uracil Adenosine triphosphate (ATP) q q Chemical energy used by all cells Energy released when phosphate bond is broken Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: DNA Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Organic Compounds: ATP and ADP Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

Animation • Enzyme Functions and ATP You must be connected to the internet to run this animation. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.

End of Chapter 2 Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Reproduction or translation of this work beyond that permitted in section 117 of the 1976 United States Copyright Act without express permission of the copyright owner is unlawful. Request for further information should be addressed to the Permission Department, John Wiley & Sons, Inc. The purchaser may make back-up copies for his/her own use only and not for distribution or resale. The Publishers assumes no responsibility for errors, omissions, or damages caused by the use of these programs or from the use of the information herein. Copyright © 2015 John Wiley & Sons, Inc. All rights reserved.
- Slides: 53