Introduction to the Gas Laws About Gases Gases

Introduction to the Gas Laws

About Gases • Gases are the most understood form of matter. • Even though different gases have different chemical properties, they tend to exhibit similar physical properties • This situation arises because gas molecules expand to fill a given space, and are relatively far apart from one another. • A volume of gas consists mostly of empty space. Thus, each gas atom/molecule behaves as if the others are not there (I. E. they are unaffected by intermolecular attractions)

Pressure • The most readily measured properties of a gas are its temperature, volume, and pressure • Pressure describes the force that a gas exerts on an area, A. P = F/A • The image below shows gas molecules inside of a cubic container. The gas molecules strike against the walls of the container. These collisions are the source of the internal pressure.

Atmospheric Pressure • You and I are currently experiencing an attractive force that pulls us toward the center of the earth (gravity). • Gas molecules in the atmosphere also experience gravity. • Because of their small masses and thermal energies, gas molecules can somewhat counteract gravity, which is why gases don’t just sit on the surface • Nonetheless, gravity causes the gases in the atmosphere to “press down” on the surface. This is atmospheric pressure.

Atmospheric Pressure • The mass of a 1 m 2 column of air extending through the entire atmosphere would be approximately 104 kg. • The pressure of this column is approximately 105 (kgm 2/s)/m 2 = 105 Pa • SI unit of pressure is the Pascal (Pa). Related units are bar, mm. Hg, and atmospheres.
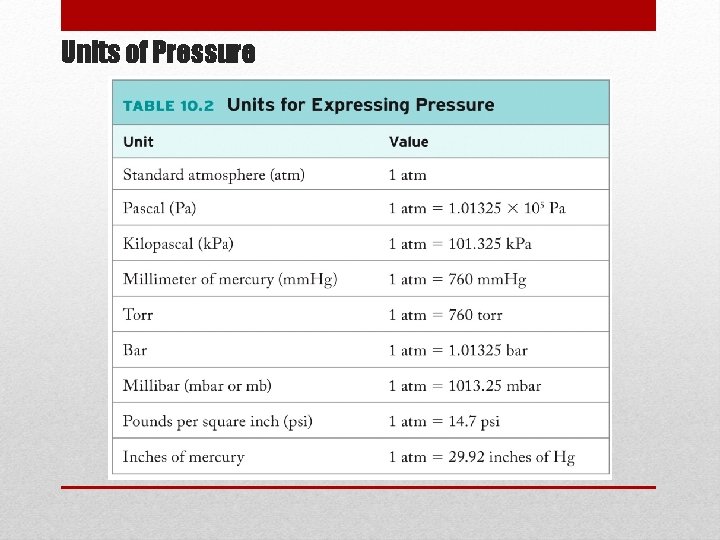
Units of Pressure

How Atmospheric Pressure Is Measure • A barometer is shown. It is composed of a glass tube that is open on one end and closed on the other. • The tube is inverted into a dish that contains mercury. Some mercury flows into the tube because the atmospheric pressure forces it upward. • Flow stops when the pressure from the weight of Hg matches the pressure of the atmosphere. 1 atm = 760 mm Hg

Kinetic Theory of Gases •

Gas Constant •

Effusion • No container is perfect. Every surface has microscopic holes through which trapped gases may escape. The rate of escape is called effusion. • It is known that the effusion rate is inversely proportional to the square root of the molar mass • This means that lighter gases escape faster than heavier ones. Why? • At equal T, both gases have the same kinetic energy, but very different speeds due to differences in mass

Effusion • Relative rates of effusion can be compared (e. g. using a known standard) to find the predicted effusion rate for a given compound: • Ex. It is found that 2. 3 m. L of carbon monoxide leaks from a sealed vessel over a 24 hr period. How much oxygen gas would you expect to leak from this vessel in 4 hours?

Gas Laws: Boyle’s Law •
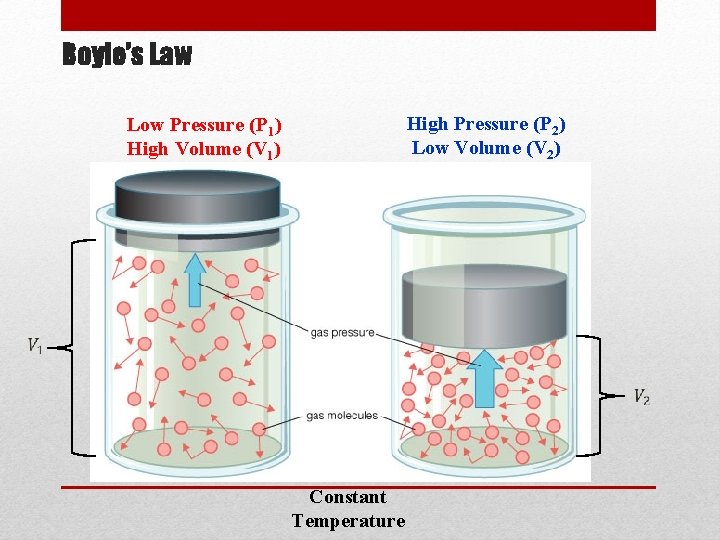
Boyle’s Law High Pressure (P 2) Low Volume (V 2) Low Pressure (P 1) High Volume (V 1) Constant Temperature

Example •
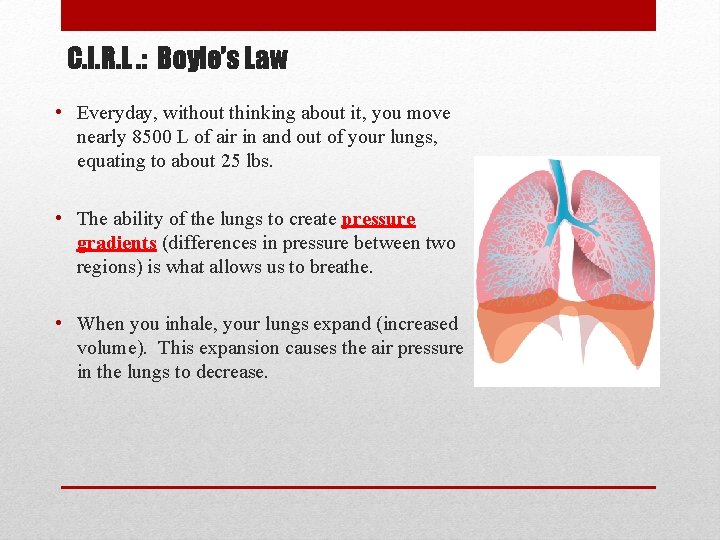
C. I. R. L. : Boyle’s Law • Everyday, without thinking about it, you move nearly 8500 L of air in and out of your lungs, equating to about 25 lbs. • The ability of the lungs to create pressure gradients (differences in pressure between two regions) is what allows us to breathe. • When you inhale, your lungs expand (increased volume). This expansion causes the air pressure in the lungs to decrease.

C. I. R. L. : Boyle’s Law • The pressure outside of the lung remains high (HP). Now, there is a pressure gradient. • Immediately, air flows from the HP region (atmosphere) to the LP region (lungs) until the pressures are equal. HP HP HP L P
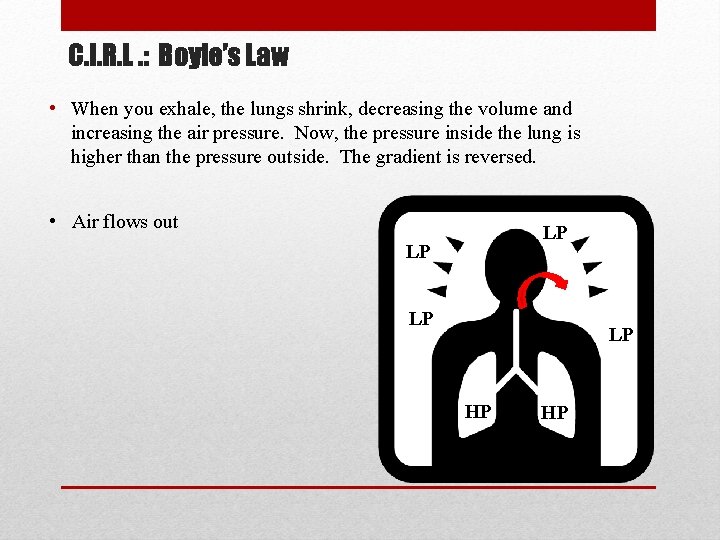
C. I. R. L. : Boyle’s Law • When you exhale, the lungs shrink, decreasing the volume and increasing the air pressure. Now, the pressure inside the lung is higher than the pressure outside. The gradient is reversed. • Air flows out LP LP HP HP

Charles’s Law • So how can we predict the change in volume with temperature? • Charles’s Law tells us that volume is directly proportional to temperature at constant pressure. When temperature goes up, volume goes up! • We can express this as: Constant Pressure
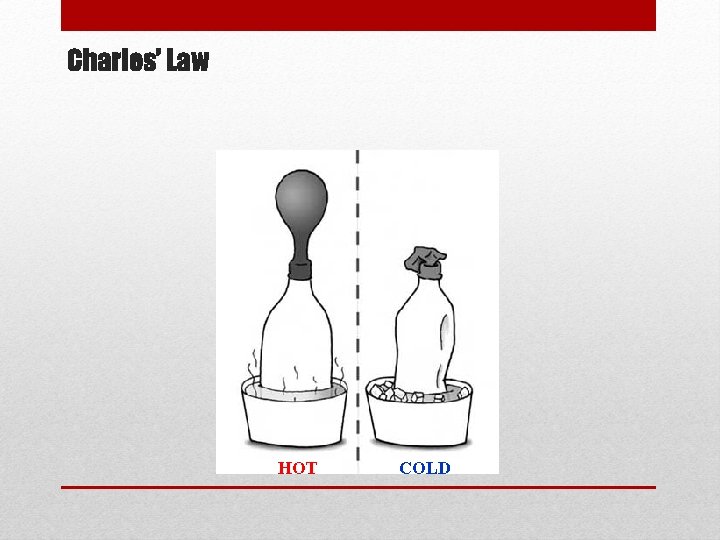
Charles’ Law HOT COLD

Example • At 25 o. C, the volume of the air in a balloon is 1. 3 L. Then, the balloon is cooled to -73 o. C using N 2 (L). What is the new volume of air?
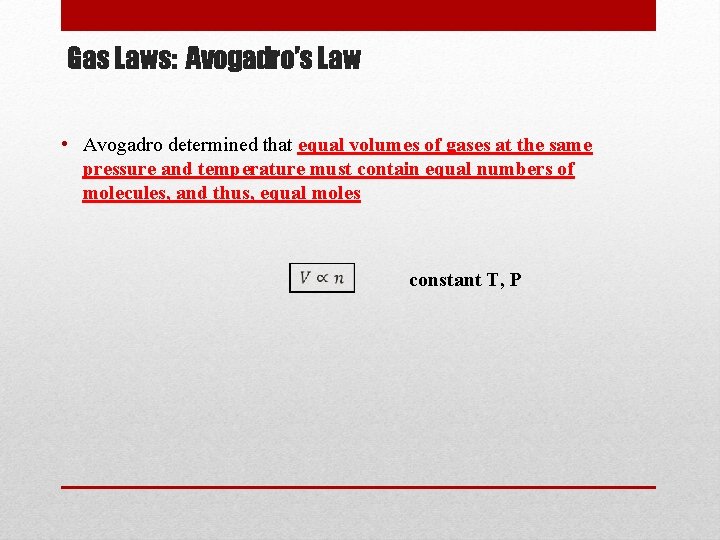
Gas Laws: Avogadro’s Law • Avogadro determined that equal volumes of gases at the same pressure and temperature must contain equal numbers of molecules, and thus, equal moles constant T, P

The Combined (Ideal) Gas Law • PV = n. RT

Ideal Gases • Any gas that follows the ideal gas law is considered an ideal (perfect) gas. • One mole of an ideal gas at 0 o. C and 1 atm of pressure occupies 22. 4 L of space. The value of R is based on these values of n, T, P, and V. • The conditions listed above (0 o. C, 1 atm) are referred to as standard temperature and pressure (STP) • Note: The Ideal gas law is a theoretical approximation, and no gas follows this law exactly, but most gases are within a few percent of this approximation, so it is very useful.
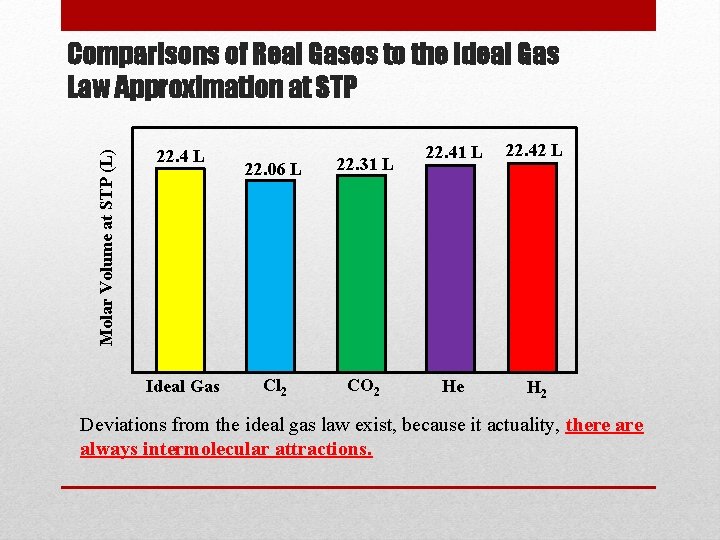
Molar Volume at STP (L) Comparisons of Real Gases to the Ideal Gas Law Approximation at STP 22. 4 L Ideal Gas 22. 06 L 22. 31 L Cl 2 CO 2 22. 41 L 22. 42 L He H 2 Deviations from the ideal gas law exist, because it actuality, there always intermolecular attractions.

Example The pressure in a 10. 0 L gas cylinder containing N 2(g) is 4. 15 atmospheres at 20. 0 o. C. How many moles of N 2(g) are there in the cylinder? • = 1. 72 moles N 2(g)

Example 2 KCl. O 3(s) 2 KCl(s) + 3 O 2(g) • The reaction above is carried out at 2. 1 atm and 70 o. C. Assuming perfect yield, how many grams of potassium chlorate are needed to make 250 m. L of oxygen? • GAS LAWS ONLY APPLY TO GASES!!! Find moles of oxygen! Use stoichiometry to find mass of potassium chlorate.

Example for you to work on at home. . • 18. 7 g of solid manganese (IV) oxide is added to 600 m. L of an aqueous solution of 1. 33 M hydrochloric acid in a sealed container at standard temperature and pressure. The reaction produces aqueous manganese (II) chloride, water, and chlorine gas. Given that the reaction generates 0. 85 L of chlorine gas, calculate the % yield.
- Slides: 27