Introduction to the Bohr Atom Model 1 Introduction

Introduction to the Bohr Atom Model 1

Introduction to the Bohr Atom Model Bohr’s “Planetary” Model (1913) Electrons posses specific amounts of energy and they must stay fixed distances (orbits) from the nucleus. Electrons that leave one orbit of an atom must move to another orbit. Electrons can only change orbits if they receive specific amounts of extra energy (quanta of energy) from the outside world. 2

Introduction to the Bohr Atom Model Bohr’s 1913” Planetary” Model (continued) If an electron receives enough energy from the outside world, it can not only leave the orbit it is in but it can leave the atom it is in. If an electron returns to the orbit it used to reside in, it will give up the extra energy it had when it moved in the first place. The energy that electrons give up when they move back into an original orbit often shows up as a specific color of light. 3

Introduction to the Bohr Atom Model Bohr’s 1913” Planetary” Model Stable atoms are arrangements of electrons around a nucleus that represent the lowest possible use of energy. This atomic arrangement of electrons about a nucleus with the lowest possible energy is known as the “ground” state for an atom. 4

Introduction to the Bohr Atom Model Bohr’s 1913” Planetary” Model This atom is in it’s “ground” state. What element does it represent? 3 p+ Which atomic arrangement of electrons represents an atom that has acquired more energy? This atom is in an “excited” state. What element does it represent? Why? ? 5 3 p+

Introduction to the Bohr Atom Model Exploring the components of the atom as envisioned by Bohr. • The electron Millikan’s Oil Drop Experiment (1909) • The nucleus Chadwick’s Beryllium Radiation Experiments (1932) 6
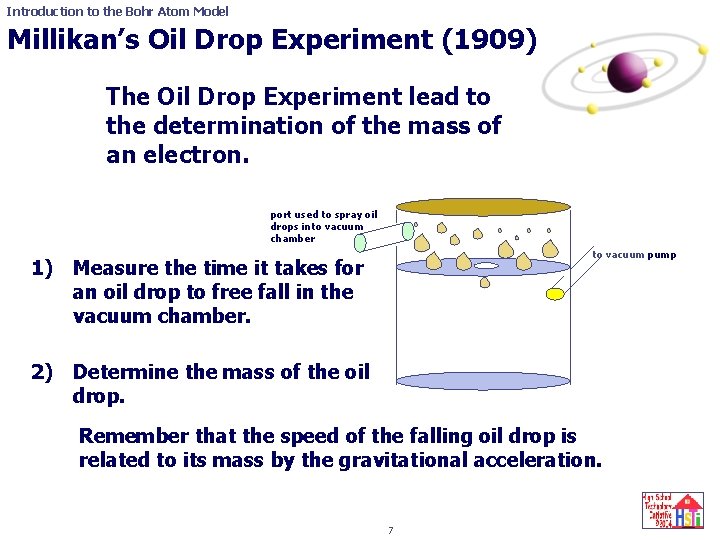
Introduction to the Bohr Atom Model Millikan’s Oil Drop Experiment (1909) The Oil Drop Experiment lead to the determination of the mass of an electron. port used to spray oil drops into vacuum chamber 1) Measure the time it takes for an oil drop to free fall in the vacuum chamber. 2) Determine the mass of the oil drop. to vacuum pump Remember that the speed of the falling oil drop is related to its mass by the gravitational acceleration. 7
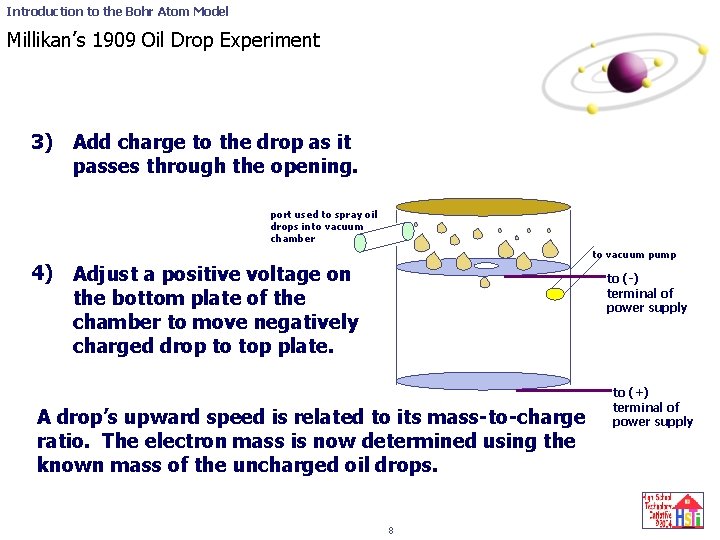
Introduction to the Bohr Atom Model Millikan’s 1909 Oil Drop Experiment 3) Add charge to the drop as it passes through the opening. port used to spray oil drops into vacuum chamber 4) to vacuum pump Adjust a positive voltage on the bottom plate of the chamber to move negatively charged drop to top plate. to (-) terminal of power supply A drop’s upward speed is related to its mass-to-charge ratio. The electron mass is now determined using the known mass of the uncharged oil drops. 8 to (+) terminal of power supply

Introduction to the Bohr Atom Model Chadwick’s Beryllium Radiation Experiment (1932) The experiment Beryllium was bombarded with alpha particles. The observation A radiation beam was given off but this stream of high energy particles was not deflected by electric or magnetic fields. The conclusion Radiation beam emitted from the beryllium consisted of particles that were not positive or negative. The impact on technology These particles were named neutrons, have about the same mass as protons, and lead to the development of uncontrolled and controlled nuclear reactors. 9

Introduction to the Bohr Atom Model View of the Atom by early 1930’s An atom had 1) An electron orbital structure as outlined in Bohr’s model. 2) Electrons whose mass is very small compared to the mass of protons. 3) A center dense core called the nucleus that had a number of protons exactly equal to the number of electrons in the orbits outside the nucleus. had a number of neutrons with each neutron weighting about the same as a proton. 10

Introduction to the Bohr Atom Model View of the Atom by the early 1930 s An atom is the smallest particle of an element that consists of a specific number of protons. The only difference between a large amount of an element and a small amount of the same element is the number of these atoms. Elements get their names from the number of protons they have in the nucleus of their atoms. An atom has the same number of electrons outside its nucleus as it has protons inside its nucleus. The physical properties of an element are due to the electron arrangement (configuration) about the nucleus of the atoms in the element. 11
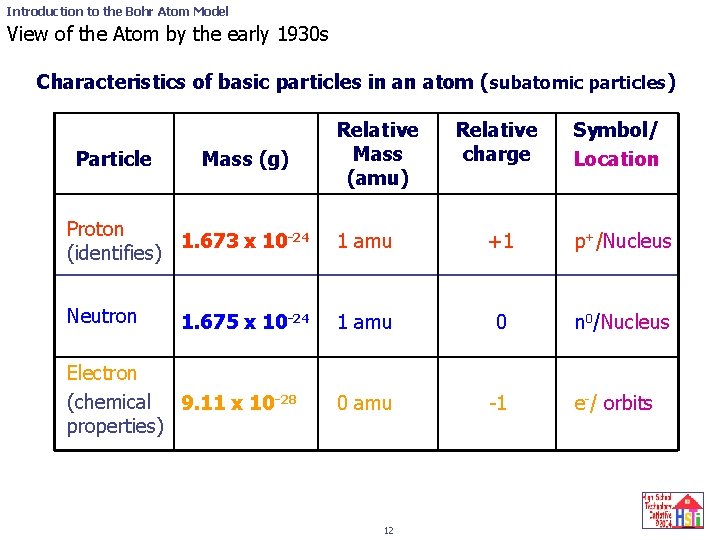
Introduction to the Bohr Atom Model View of the Atom by the early 1930 s Characteristics of basic particles in an atom (subatomic particles) Particle Mass (g) Relative Mass (amu) Relative charge Symbol/ Location Proton 1. 673 x 10 -24 (identifies) 1 amu +1 p+/Nucleus Neutron 1 amu 0 n 0/Nucleus 0 amu -1 1. 675 x 10 -24 Electron (chemical 9. 11 x 10 -28 properties) 12 e-/ orbits
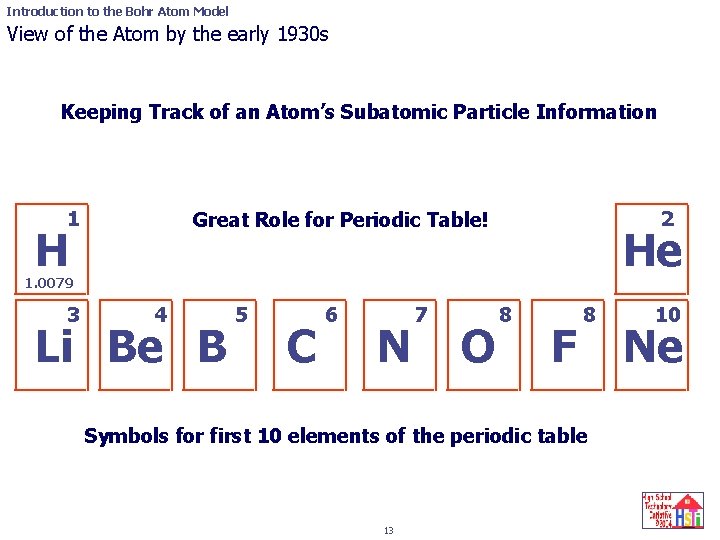
Introduction to the Bohr Atom Model View of the Atom by the early 1930 s Keeping Track of an Atom’s Subatomic Particle Information 1 2 Great Role for Periodic Table! H He 1. 0079 3 4 Li Be B 5 C 6 N 7 O 8 F 8 Symbols for first 10 elements of the periodic table 13 10 Ne

Introduction to the Bohr Atom Model Keeping Track of an Atoms Subatomic Particle Information 1 (the number of protons in the nucleus) Atomic Number H (the number of electrons in the atom) Element Symbol 1. 0079 (indicates the name of the element) Atomic Mass in atomic mass units (amu) (approximate mass of protons plus the mass of neutrons) Remember (1) The mass of a proton almost equals 1 amu (2) The mass of a proton equals the mass of a neutron (3) The number of neutrons equals the nearest whole number difference between the atomic mass and the atomic number (4) The mass of the electron is small compared to the mass of the protons and neutrons. 14
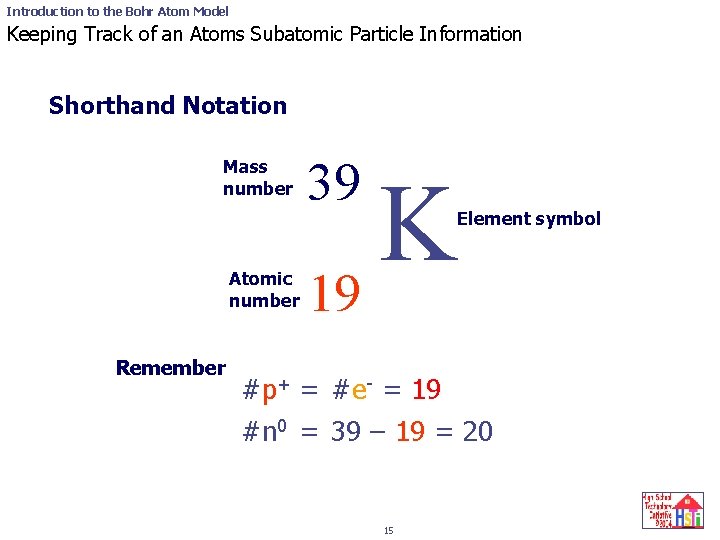
Introduction to the Bohr Atom Model Keeping Track of an Atoms Subatomic Particle Information Shorthand Notation Mass number Atomic number Remember 39 K 19 Element symbol #p+ = #e- = 19 #n 0 = 39 – 19 = 20 15
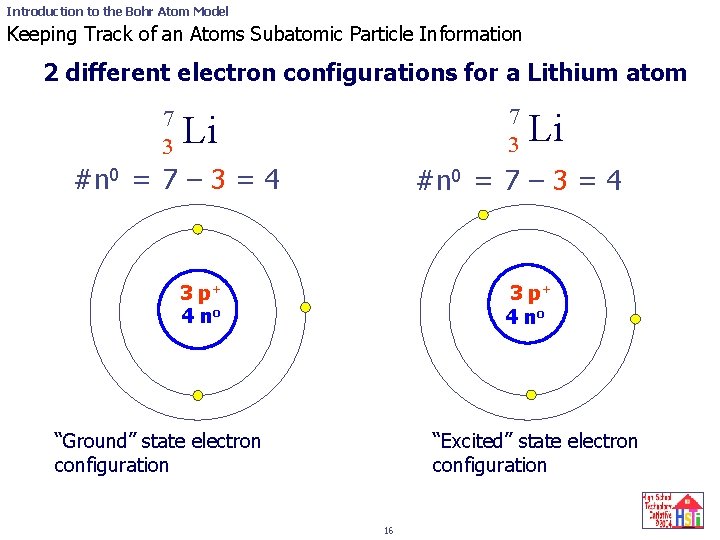
Introduction to the Bohr Atom Model Keeping Track of an Atoms Subatomic Particle Information 2 different electron configurations for a Lithium atom 7 3 Li #n 0 = 7 – 3 = 4 3 p+ 4 no “Ground” state electron configuration “Excited” state electron configuration 16
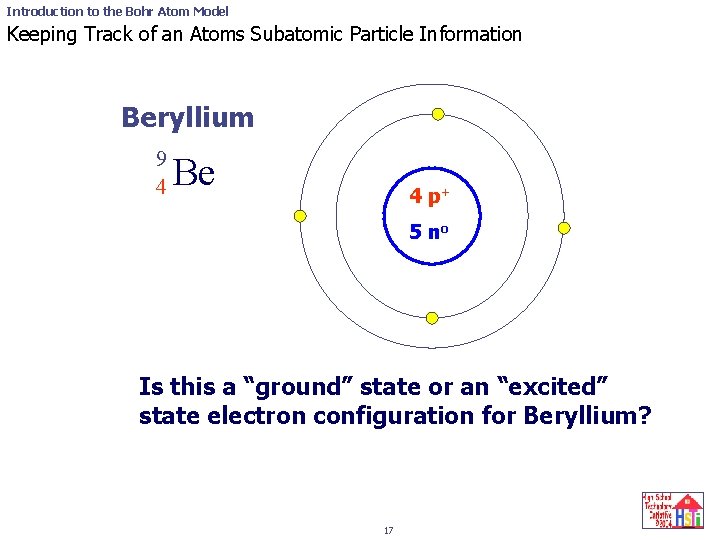
Introduction to the Bohr Atom Model Keeping Track of an Atoms Subatomic Particle Information Beryllium 9 4 Be 4 p+ 5 no Is this a “ground” state or an “excited” state electron configuration for Beryllium? 17

Introduction to the Bohr Atom Model Keeping Track of an Atoms Subatomic Particle Information Boron 11 5 B 5 p+ ? no Is this a “ground” state or an “excited” state electron configuration for Boron? 18
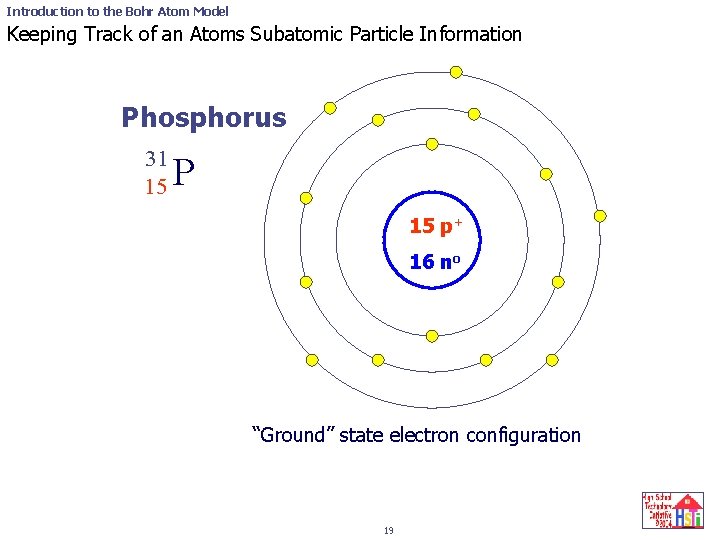
Introduction to the Bohr Atom Model Keeping Track of an Atoms Subatomic Particle Information Phosphorus 31 15 P 15 p+ 16 no “Ground” state electron configuration 19
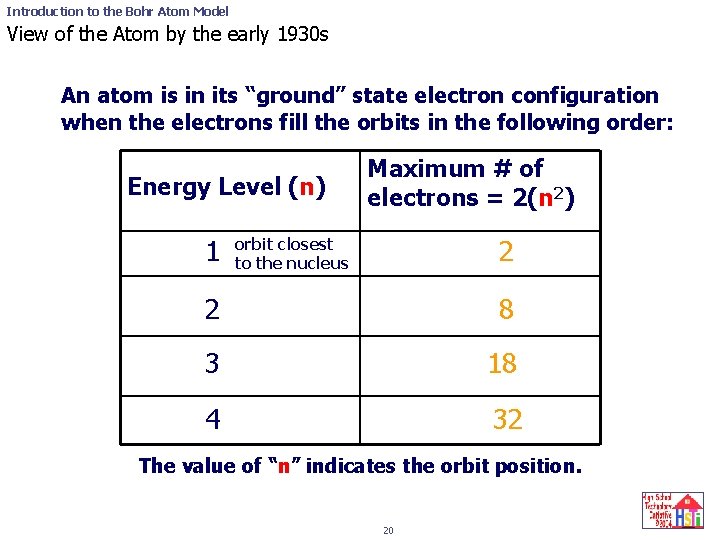
Introduction to the Bohr Atom Model View of the Atom by the early 1930 s An atom is in its “ground” state electron configuration when the electrons fill the orbits in the following order: Energy Level (n) 1 Maximum # of electrons = 2(n 2) 2 orbit closest to the nucleus 2 8 3 18 4 32 The value of “n” indicates the orbit position. 20

Introduction to the Bohr Atom Model Summary Bohr’s atom Electrons posses specific amounts of energy and they must stay fixed distances (orbits) from the nucleus. Electrons that leave one orbit of an atom must move to another orbit. Electrons can only change orbits if they receive specific amounts of extra energy (quanta of energy) from the outside world. 21

Introduction to the Bohr Atom Model Summary Bohr’s atom If an electron receives enough energy from the outside world, it can not only leave the orbit it is in but leave the atom it is in. If an electron returns to the orbit it used to reside in, it will give up the extra energy it had when it moved in the first place. The energy electrons give up when they move back into an original orbit often shows up as a specific color of light. 22

Introduction to the Bohr Atom Model Summary Bohr’s atom Stable atoms are arrangements of electrons around the nucleus that represent the lowest possible use of energy. This atomic arrangement of electrons about the nucleus is known as the “ground” state for an atom. 23

Introduction to the Bohr Atom Model 24
- Slides: 24