Introduction to Substance Use Disorder Neurobiology Melissa Weimer

Introduction to Substance Use Disorder: Neurobiology Melissa Weimer, DO, MCR OHSU Addiction Medicine Section Oregon Pain Guidance 6 th Annual Conference, May 4, 2017

Disclosures Dr. Weimer was a consultant to Indivior, for which she received honorarium. This was a one-time consultation to discuss how to stop taking medication they manufacture.

Learning Objectives 1. Provide a conceptual framework for understanding substance use disorders 2. Understand the alignment between the neurobiology of addiction and trauma 3. Understand addiction as a chronic disease of the brain that includes disordered behaviors

Case 1 Jessica is a 23 year old healthy female who presents to the hospital asking for detox from heroin. She states she has been using about 1 -1. 5 grams of IV heroin for the last 1 year. She also smokes cannabis on a near daily basis and will sometimes use methadone to help prevent opioid withdrawal if she can not afford heroin. She has a stable living situation with her supportive boyfriend who does not endorse a substance use disorder. He asks you to “please help her” so that “she does not die”. She states that she was introduced to heroin by a “friend”, and she can’t really say why she started using it.

Case 1, physical exam ■ On presentation, she is diaphoretic, tremulous, and anxious appearing. Her pupils are slightly pinpoint, she has no gooseflesh, and her abdomen is soft and nontender. She does not have bony pain in her spine, neurologic deficits, or a heart murmur. Her lungs are clear. She has track marks in her bilateral upper extremities. She has linear thoughts and intact judgement. ■ Her Clinical Opioid Withdrawal Score (COWS) score is 5

What happened to Jessica? • • Genetic Predisposition Environmental factors Complex Trauma Exposure to the Substance • All together, these lay the groundwork for the persistent neuobiological changes that effect learning, motivation, behavior, and decision making
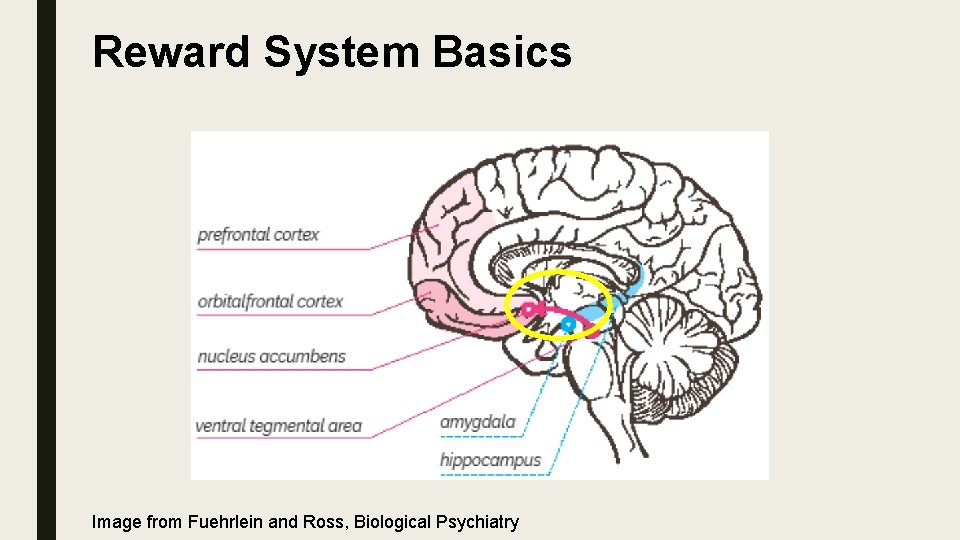
Reward System Basics Image from Fuehrlein and Ross, Biological Psychiatry
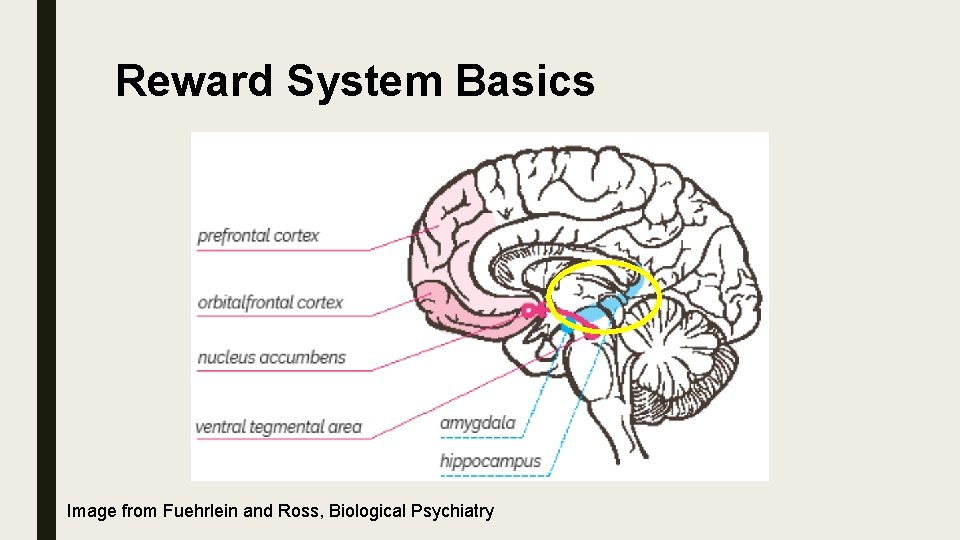
Reward System Basics Image from Fuehrlein and Ross, Biological Psychiatry
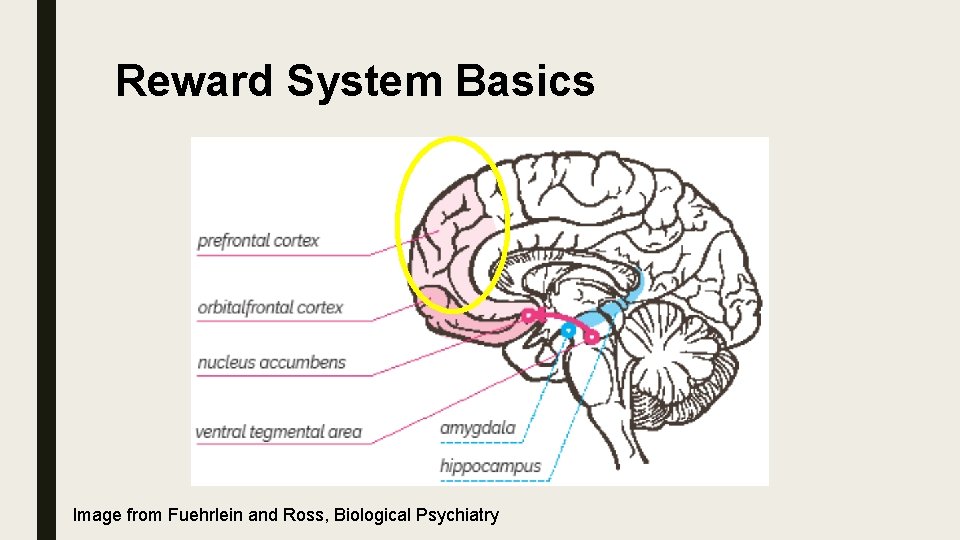
Reward System Basics Image from Fuehrlein and Ross, Biological Psychiatry

Reward System Basics ■ Biologically and evolutionary, the primary purpose of life is to survive and pass on genetics ■ The reward system is designed to reinforce eating, drinking water, sexual activity, and raising offspring ■ These are activities designed for survival and procreation, which are the most important things to the organism and the species ■ For lower level organisms, the reward system is critical to survival and drives daily activity

Basic Review ■ “GO” Dopaminergic reward system ■ “STOP” Cortex ■ Reward system – Reinforce primitive behaviors designed for survival of person and species ■ Cortex – Higher executive planning and long term goals

Key Points ■ The cortex becomes “hijacked” by the reward system and the behavior of the individual is under the control of the reward system ■ The most important thing to the individual with substance use disorder is the next use of the substance ■ All consequences must take a back seat to short term survival

SOUNDS SIMPLE, RIGHT? WAIT, THERE’S MORE….
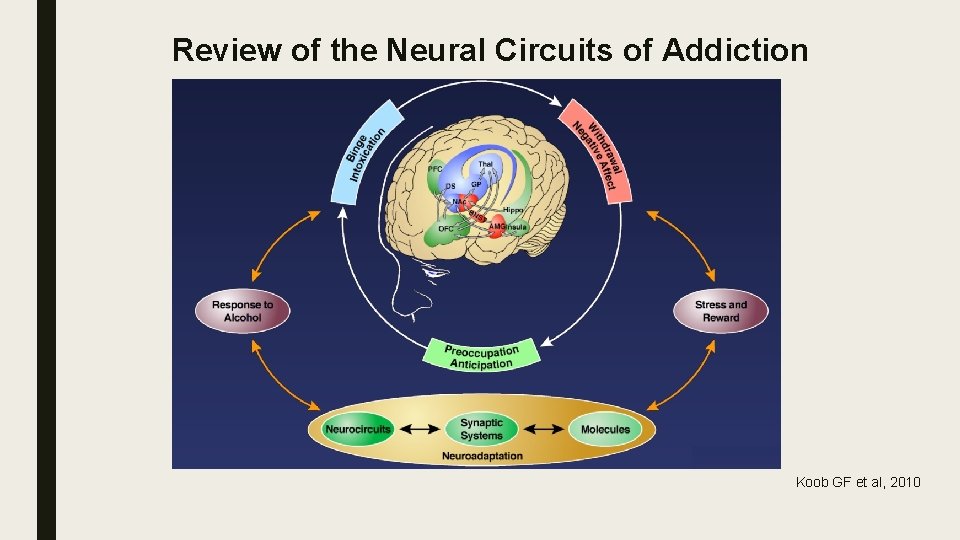
Review of the Neural Circuits of Addiction Koob GF et al, 2010
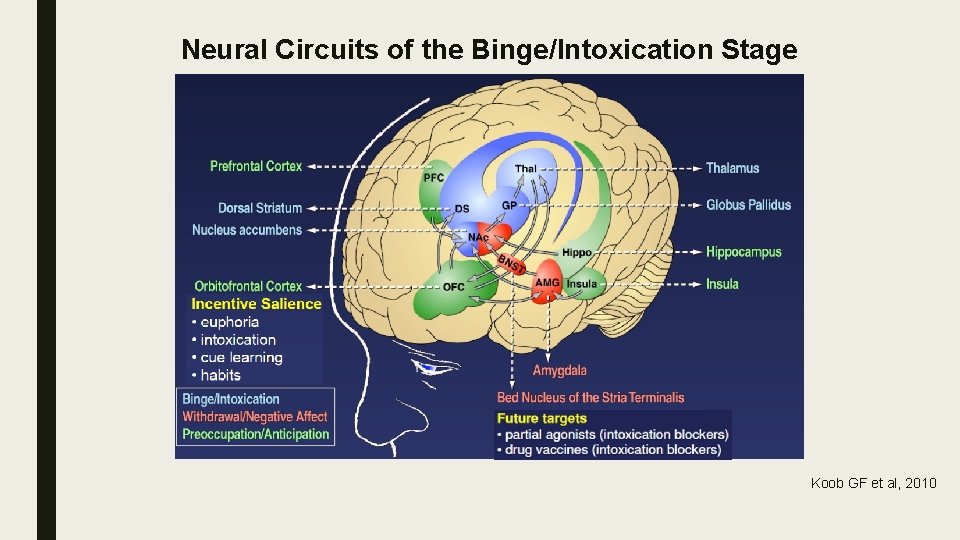
Neural Circuits of the Binge/Intoxication Stage Koob GF et al, 2010
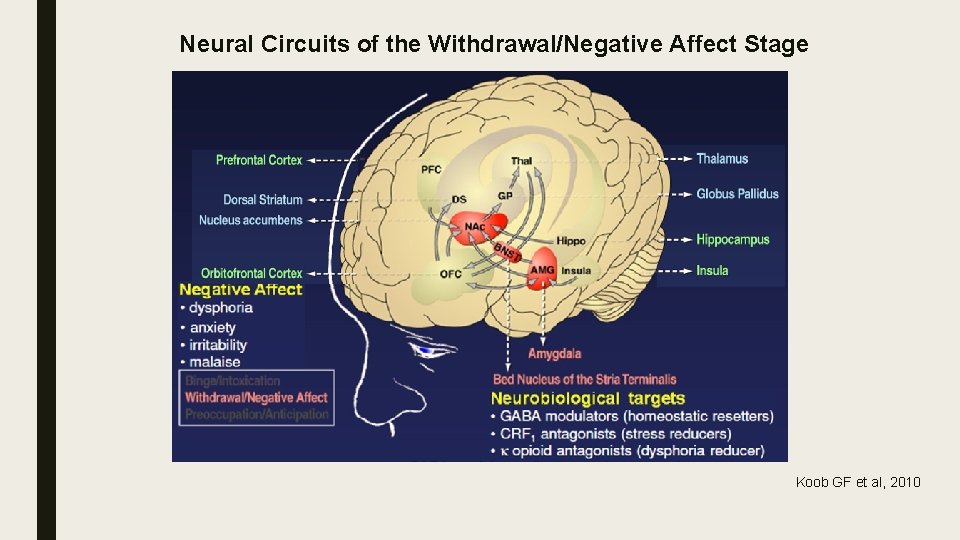
Neural Circuits of the Withdrawal/Negative Affect Stage Koob GF et al, 2010
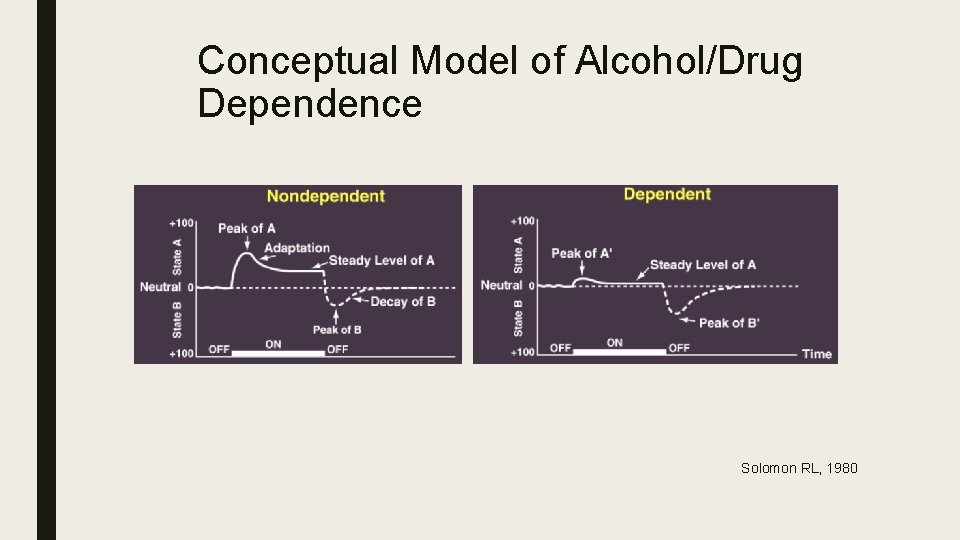
Conceptual Model of Alcohol/Drug Dependence Solomon RL, 1980

Reward Transmitters Implicated in the Motivational Effects of Substances Positive Hedonic Effects Negative Hedonic Effects of Withdrawal Dopamine – “dysphoria” Opioid Peptides – pain Serotonin – “dysphoria” GABA – anxiety, panic attacks

Anti-Reward Transmitters Implicated in the Motivation Effects of Substances Positive Hedonic Effects Dynorphin – “dysphoria” Corticotropin-Releasing Factor (CRF) – stress Norepinephrine – stress These are ACTIVATED in amygdala and ventral striatum during withdrawal
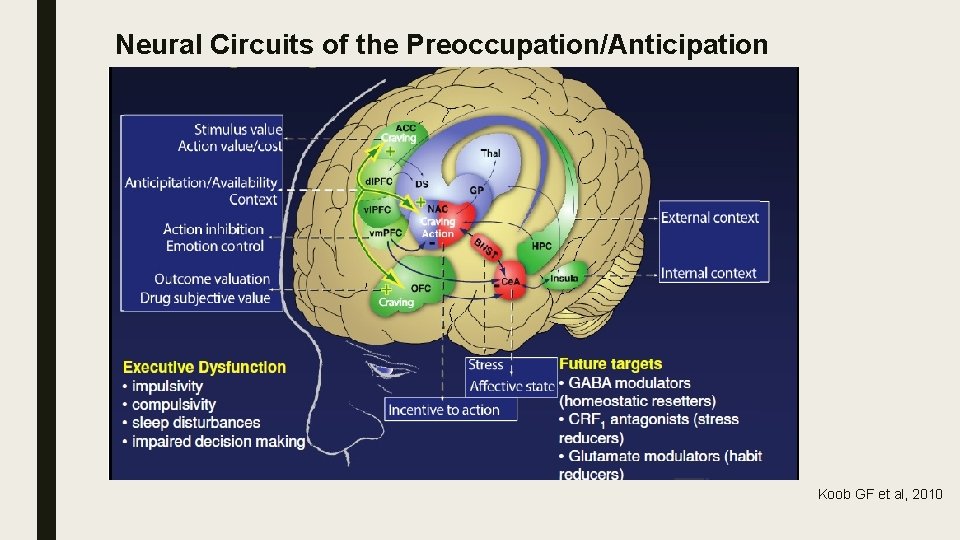
Neural Circuits of the Preoccupation/Anticipation “Craving” Stage Koob GF et al, 2010
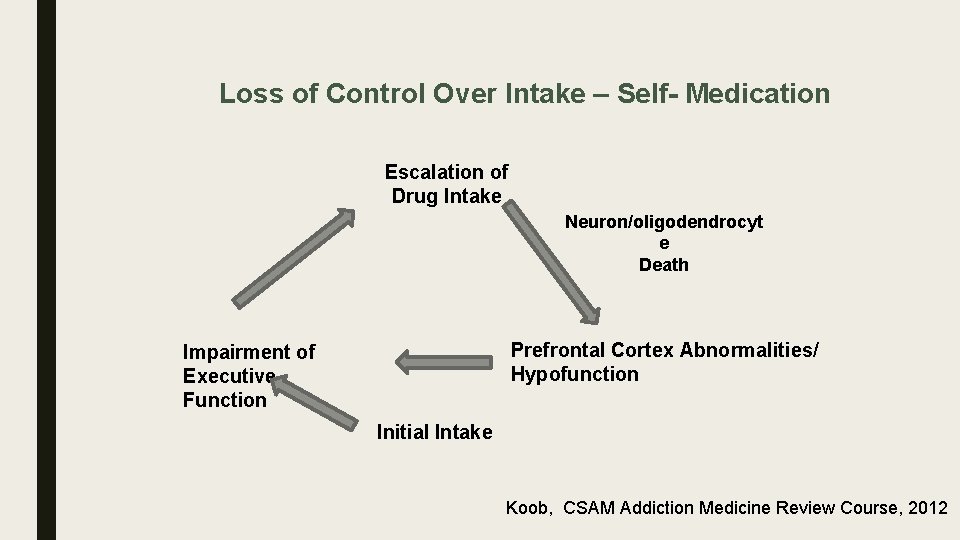
Loss of Control Over Intake – Self- Medication Escalation of Drug Intake Neuron/oligodendrocyt e Death Prefrontal Cortex Abnormalities/ Hypofunction Impairment of Executive Function Initial Intake Koob, CSAM Addiction Medicine Review Course, 2012
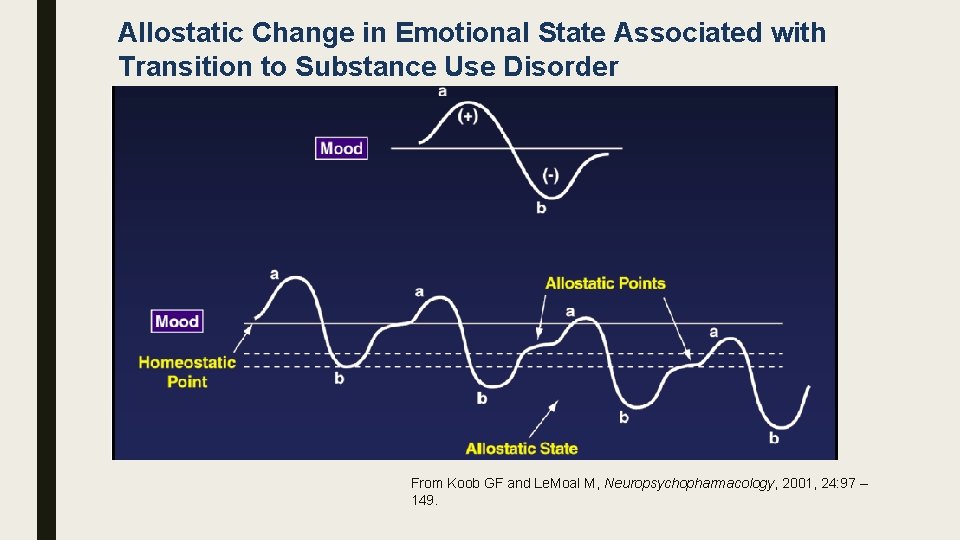
Allostatic Change in Emotional State Associated with Transition to Substance Use Disorder From Koob GF and Le. Moal M, Neuropsychopharmacology, 2001, 24: 97 – 149.
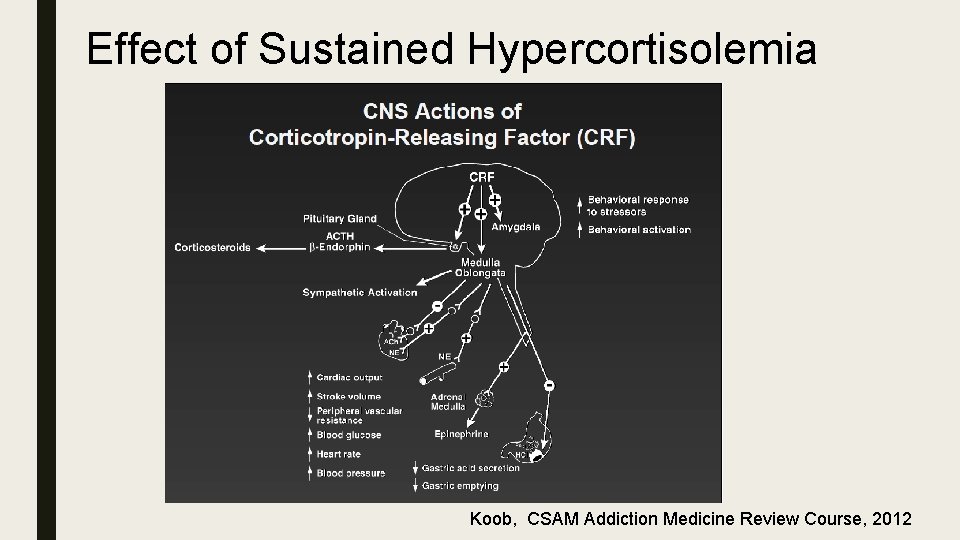
Effect of Sustained Hypercortisolemia Koob, CSAM Addiction Medicine Review Course, 2012
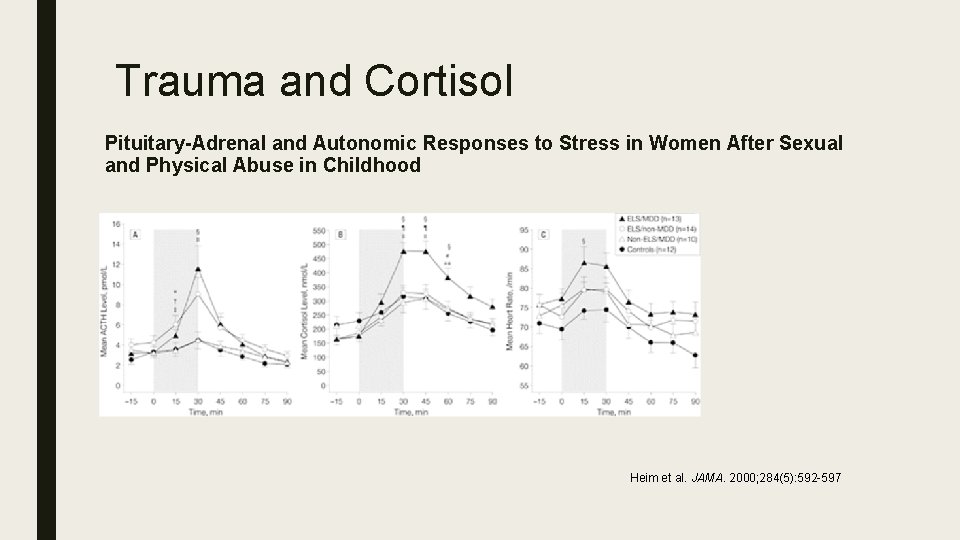
Trauma and Cortisol Pituitary-Adrenal and Autonomic Responses to Stress in Women After Sexual and Physical Abuse in Childhood Heim et al. JAMA. 2000; 284(5): 592 -597

Trauma is: : Inescapable powerlessness A “blow out” of your fight or flight system “The result of exposure to an inescapably stressful event that overwhelms a person’s coping mechanism” – Bessel Van der Kolk

Effects of Prolonged Hypercortisolemia o Decreased ability of nucleus accumbens to synthesize dopamine o o o Functional severing of connection between medial prefrontal cortex and limbic system, allowing limbic system to run amok further Decreased hippocampal volume: o decreased ability to “calm” amygdala o decreased negative feedback to HPA access o decreased reparative process (consolidating memories and making sense of them) CNS suppressants (Et. OH, benzodiazepines, opioids) further impair ability to process memories and emotions

So What’s the Problem? o The reward system is crucial for survival; if out of balance, it takes over: impulsivity, search for immediate gratification, unable to tolerate distress o Addictive drugs and search for pain relief can dump tons of dopamine into these circuits o Addictive drugs increase activity in these neurons, or prolong actions of neurotransmitters they release o New research show pain relief activates these neurons to drive habitual relief seeking Navratilova E et al, 2012

What Happens Over Time? Chronic dopamine firing reshapes these circuits, making them very fast and hard to control. Accelerator: D 1 receptor Brakes: D 2 receptor D 1 Receptors: Dopamine in the receptors tells Nucleus Accumbens to say “Yes!” D 2 Receptors: Activation of these receptors slows decision-making; allows frontal cortex time to step in

Too Much Accelerator is a Bad Thing When DA neurons are chronically over-active, they activate D 1 receptors – D 1 pathway becomes more efficient, speeding up decisions to seek relief – Activate anti-reward circuits (dynorphin, CRF, NE) – Increase stress response and worsen mood – both amplify pain signals – Pain severity increases and relief-seeking behaviors become compulsive

What Happened to the Brakes? Excessive or chronic dopamine input inhibits the indirect/D 2 pathway ■ Big spikes in dopamine desensitize and internalize D 2 receptors ■ D 2 receptor can’t work again until it is recycled (takes an hour) or a new receptor is synthesized You may ultimately wind up with a system that has no brakes

Case continued… ■ Jessica completes a three day stay in the hospital for detoxification from opioids and states she feels excited to go look for a job and new place to live. ■ The counselor discusses treatment options with her and she states, “This was a huge wake-up call for me. I do not even like heroin. I do not need to go to treatment, and I don’t like groups…I’m not like those people at AA meetings. Starting today I promise that I will never use heroin again. ” ■ Jessica is discharged with a pamphlet about AA groups in the area

Clinical Behaviors of a Substance Use Disorder ■ Denial – Not a lie; it is a belief created by the reward system – There is little motivation to change when the behavior is not believed to be a problem, hence the substance use disorder is protected

Case continued…. let’s pretend for a moment ■ Jessica was admitted to the hospital for DKA from brittle type 1 DM ■ Jessica completes a three day stay in the hospital for blood sugar management and states she feels excited to go look for a job and new place to live. ■ The provider discusses treatment options with her and she states, “This was a huge wake-up call for me. I do not even like diabetes. I do not need to go to treatment, and I don’t like groups…I’m not like those people in the endocrine clinic. Starting today I promise that I will never have diabetes again. ” ■ Jessica is discharged with a pamphlet about diabetes support groups in the area

Case continued… § Consider another perspective—you are Jessica’s SO or sibling. You explain that you love her and want her to be a part of your life. But you are no longer able to tolerate heroin use. § You explain to her that she must choose; either heroin or her family. § She says, “I love you more than anything in this world. I promise you that I will never use heroin again. I do not want to lose you. ” § Two days later you arrive unexpectedly to her home and find her intoxicated. § As the loving family member how do you feel?

Clinical Behaviors of a Substance Use Disorder ■ “Broken” Promises – Help the patient to continue their substance use disorder by denying a need for treatment or other interventions – As with denial, this is usually not the patient “lying” but a symptom of the disease process, which the patient truly believes – Broken promises can destroy relationships and families and make it very difficult to regain trust

Case continued… ■ Jessica presents to the emergency room following a relapse on heroin. ■ She says he was doing well and managed to stay sober for three weeks. Then about a week ago she found out that her friend from high school overdosed on fentanyl. She was very upset by this news and relapsed. ■ Her SO states that she and this friend were not close.

Clinical Behaviors of a Substance Use Disorder: Excuses ■ Addiction is cunning and baffling ■ Excuses will be created to convince the cortex to continue the use ■ Sometimes the excuses are negative (stressful event, rainy day), other times they are positive (happy event, sunny day) ■ A patient with a substance use disorder will always find a way to create excuses to enable the disorder ■ At times, the excuse is legitimate, i. e, spouse tragically dies and patient relapses after 5 years of sobriety

WHAT CAN BE DONE?

Clinical Implications for Addiction Recovery Interventions that increase D 2 pathway activity facilitate recovery o Need just enough DA to activate D 2 receptors, get some inhibition but not knock them out o Consistent low level DA input to build back inhibition o Lots of tiny opportunities for little reward o Medication treatments The tiny things in life are what make life good, and allow D 2 receptors to give your brain time to make a choice.

Summary o Addiction behaviors activate, and over-stress, the reward system o In both addiction and pain, when the reward system is over-activated, anti-reward neurotransmitters in the limbic system are enhanced o In both addiction and chronic pain, the executive function of the prefrontal cortex is impaired o Denial, promises and excuses are clinical manifestations of the cunning and baffling nature of the addictive process o Healing process involves re-wiring the frontal cortex to the limbic system and ventral striatum o You can’t engage in Recovery (from trauma, pain, addiction) without an alert, engaged prefrontal cortex

References ■ Shape shifting pain: chronification of back pain shifts brain representation from nociceptive to emotional circuits. Hashmi JA, et al. Brain 2013: 136; 2751 -2768 ■ Neurocircuitry of addiction. Koob GL et al. Neuropsychopharmacology. 2010 Jan; 35(1): 21738 ■ Is there a common molecular pathway for addiction? Nestler EJ. Nat Neurosci. 2005 Nov; 8(11): 1445 -9. ■ The opponent-process theory of acquired motivation: the costs of pleasure and the benefits of pain. Solomon RL. Am Psychol. 1980 Aug; 35(8): 691 -712. ■ Pain relief produces negative reinforcement through activation of mesolimbic reward– valuation circuitry. Navratilova et al. Proc Natl Acad Sci U S A. 2012 Dec 11; 109(50): 2070913. ■ New concepts in the neurobiology of pain and addiction. Trafton J. Lecture at CSAM Addiction Medicine State of the Art Conference, 2015. ■ Pituitary-Adrenal and Autonomic Responses to Stress in Women After Sexual and Physical Abuse in Childhood. Heim et al. JAMA. 2000; 284(5): 592 -597 ■ Van der Kolk, B. (2014). The Body Keeps the Score: Brain, Mind, and Body in the Healing of Trauma. New York, NY: Viking.
- Slides: 41