INTRODUCTION TO SCIENCE LAB SAFETY BRANCHES OF SCIENCE

INTRODUCTION TO SCIENCE
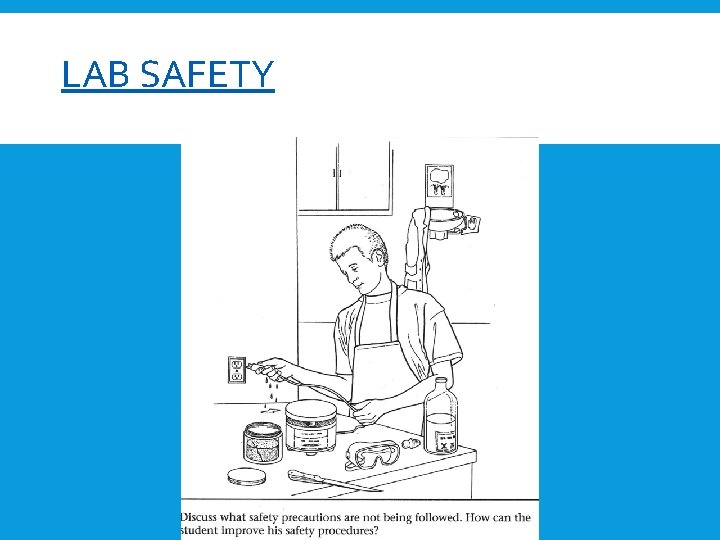
LAB SAFETY
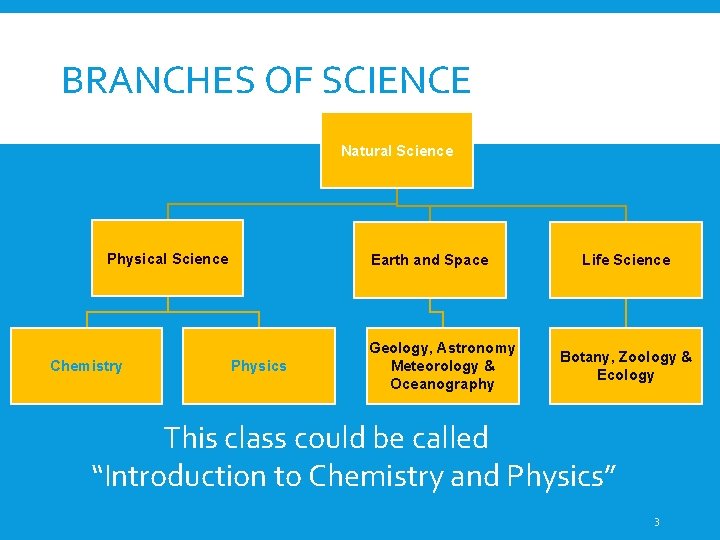
BRANCHES OF SCIENCE Natural Science Physical Science Chemistry Earth and Space Physics Geology, Astronomy Meteorology & Oceanography Life Science Botany, Zoology & Ecology This class could be called “Introduction to Chemistry and Physics” 3

PHYSICAL SCIENCE • Science is a process of gathering knowledge about the natural world • There are 2 main branches of physical science that cover the two main categories studied • The branch of chemistry covers the category of matter • Matter makes up the “stuff” in our world – The branch of physics covers the category of energy • Energy is the ability to do work

THE BIG IDEAS OF PHYSICAL SCIENCE 1. Space and Time- The universe is very old and big. It is about 13. 7 billion years old and we can observe 700 million billion meters in diameter. The four dimensions (Length, width, height and time). 2. Matter and Change - Matter is anything with volume and mass. It can be a solid, liquid, or gas. The building blocks are called atoms. Matter has mass and volume and can change forms. 5

THE BIG IDEAS OF PHYSICAL SCIENCE 3. Forces and Motion- Forces cause motion. If you push an object it will move. The motion of cars on a city street is captured in this time-exposure photograph. Forces govern changes in the motion of each car. 6

THE BIG IDEAS OF PHYSICAL SCIENCE 4. Energy- Energy exists in many forms. Moving objects have a kind of energy called kinetic energy. An object that is not moving has stored energy, or potential energy. Energy can be transferred from one form to another Panels on a solar convert energy from the sun into the mechanical energy of its moving parts 7

STEPS OF SCIENTIFIC INQUIRY A. Questioning 1. Based on observations using senses and tools. 2. Inferring- explain observations using prior knowledge. B. Hypothesizing 1. Must be testable 2. If------, then ------. C. Testing a hypothesis 1. Prediction- tells what you expect to happen under a particular set of conditions Educated guess
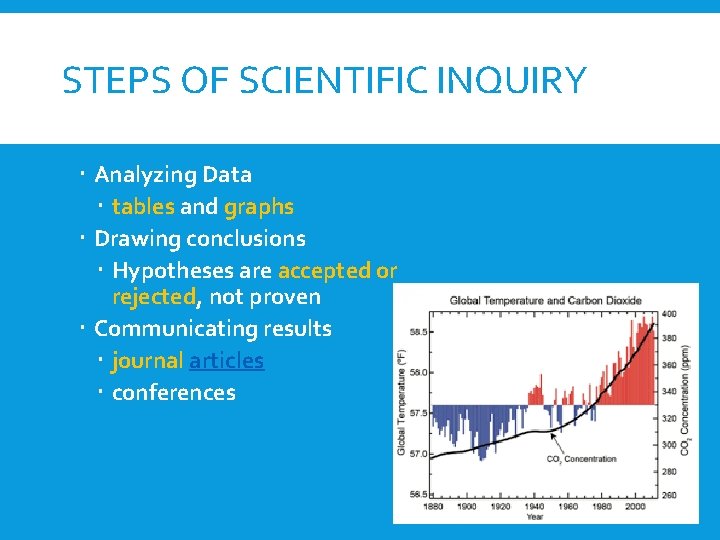
STEPS OF SCIENTIFIC INQUIRY Analyzing Data tables and graphs Drawing conclusions Hypotheses are accepted or rejected, not proven Communicating results journal articles conferences

RESULTS OF SCIENTIFIC INQUIRY • Theories are well-tested explanations of observations • Laws are summaries of patterns of observations
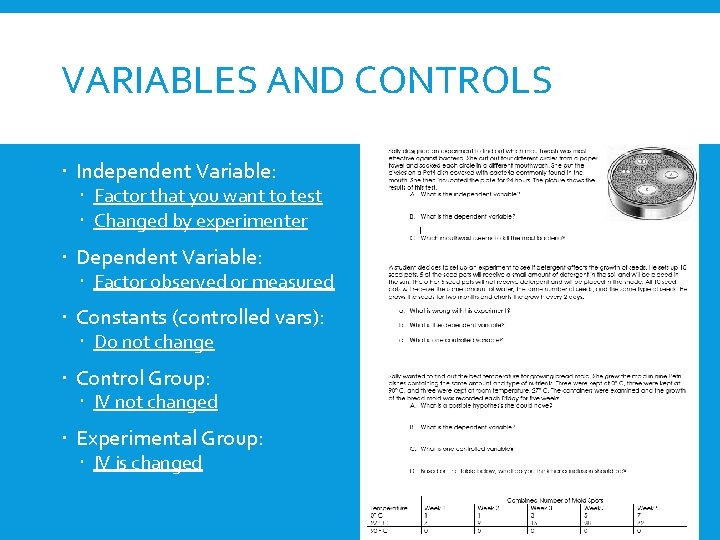
VARIABLES AND CONTROLS Independent Variable: Factor that you want to test Changed by experimenter Dependent Variable: Factor observed or measured Constants (controlled vars): Do not change Control Group: IV not changed Experimental Group: IV is changed
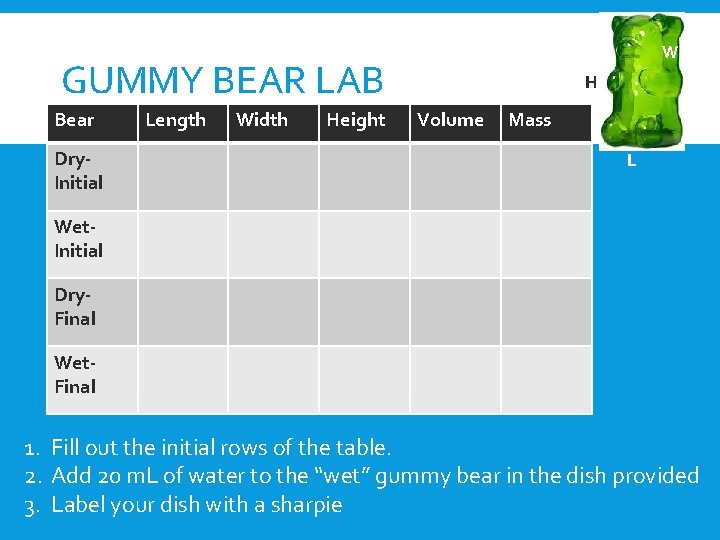
W GUMMY BEAR LAB Bear Dry. Initial Length Width Height H Volume Mass L Wet. Initial Dry. Final Wet. Final 1. Fill out the initial rows of the table. 2. Add 20 m. L of water to the “wet” gummy bear in the dish provided 3. Label your dish with a sharpie

SCIENTIFIC NOTATION A way to express very small or very large numbers Example: 12, 345 = 1. 2345 x 104 _ base unit__ – must be between 1 and 9 _ten_ 0. 00456 = 4. 56 x 10 -3 Exponent – the # of times the decimal was moved (+) to the left (-) to the right

SCIENTIFIC NOTATION PROBLEMS Reverse it! (+) right (-) left 56934 = 8. 98736 x 104 = 1280 = 0. 0345 0. 0000037 = 2. 347 x 10 -3 = = 3. 48763 x 10 -4 = 5. 7654 x 105 =
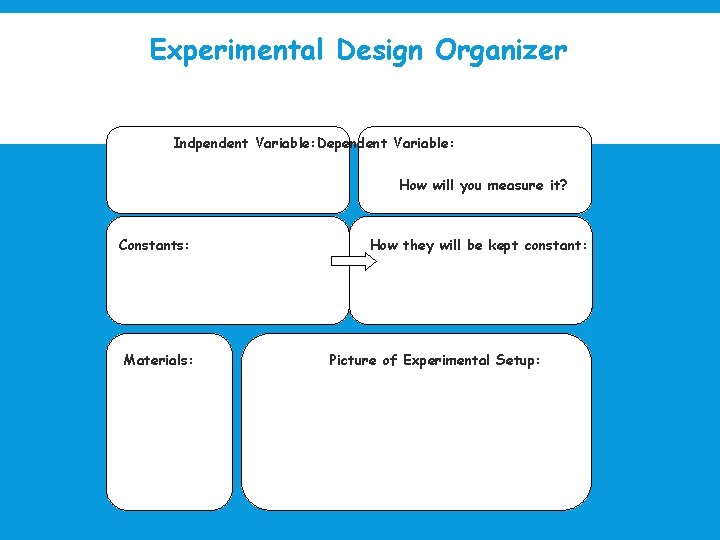
Experimental Design Organizer Indpendent Variable: Dependent Variable: How will you measure it? Constants: Materials: How they will be kept constant: Picture of Experimental Setup:

MEASUREMENT • The International System of Units (SI Units) are what scientists generally use Quantity Unit Symbol Length meter m Mass Kilogram kg *Volume cubic meter/liter M 3 /L Temperature Kelvin K Time Second s • a set of units based on the powers of ten 1. NO NAKED NUMBERS 2. Metric is BIGGER and therefore BETTER
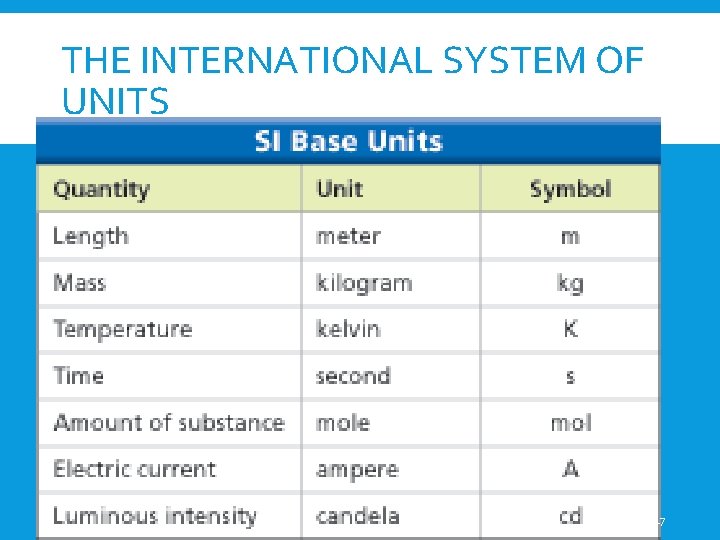
THE INTERNATIONAL SYSTEM OF UNITS 17

SI UNITS OF MEASURE All measurements need a number and a unit. Example: 5 ft 3 in or 25ºF NO!!!!!!! Scientists usually do not use these units. They use a unit of measure called SI or International System of Units. Base Units – more examples on following slide Length- straight line distance between 2 points is the meter (m) Mass- quantity of matter in an object or sample is the kilogram (kg) 18

3 cm 1 cm 7 cm METRIC CONVERSION FOLDABLE

METRIC SYSTEM Let’s do some more practice! Based on powers of ten We use prefixes to change the value Let’s try! 1. 5 dm = ______ m 2. 9 mg = ______ g 3. 4 cm = ______ m 4. 7. 02 h. L = _____ L 5. 0. 32 cm = _____ m 6. 4 m. L = _____ L 7. 0. 03 hg = ____ dg 8. 6035 mm = ______ cm 9. 6. 5 DL = _____ L 10. 3. 82 g = ______ kg

9/16 DO NOW 1. Please have your HW out on your desk 2. Convert: a) 13 mg = ______ g b) 4, 000 cm = ______ Hm c). 56 Km = ______ m d) 903 DL = _____ c. L 22

9/16 AGENDA Do Now and go over Do Now / label new packet Go over HW answers Scientific Notation HW: None Learning Objectives: Convert numbers using the rules of scientific notation 23

SECTION 1. 3 Scientific Notation – A way of expressing a value as the product of a number between 1 and 10 and a power of 10. Example: 300, 000 = 3. 0 x 108 The exponent 8 tells you the decimal point is really eight places to the right of 3. Example: 0. 00086 = 8. 6 x 10 -4 The exponent -4 tells you the decimal point is really four places to the left of 8 Scientists estimate that there are more than 200 billion stars in the Milky Way galaxy. 24

MATH PRACTICE 1. Convert the following to scientific notation. . 00823 300, 000 2. Convert these to normal number form: 7. 6 × 10− 4 1. 5 × 107 25

MORE PRACTICE 1. 4. 39 x 10 -2 = 2. 8. 52 x 100 = 3. 2792. 38 = 4. 7. 23 x 10 -3 = 5. 5480= 6. . 0005 = 26

9/19 DO NOW Write down 5 things you recall we learned or talked about last week 27

9/19 AGENDA 1. Do Now and go over Do Now 2. Review and Practice 3. Measurement Lab Tomorrow – No Sandals!! HW: 1) HW Packet 2) Dress for lab Test on Friday!! Learning Objectives: Review 28

SCIENCE FOUNDATIONS 1) The volume of a tissue box can be found by multiplying __ x __ 2) The difference between a theory and a law is that a _______ is something that is known for certain, and a _____ is something that is uncertain 3) Physical science is the study of: a) matter and energy b) matter and motion c) motion and energy 4) Match the unit with the type of measurement: ___ mass a) Hectometers (Hm) ___ density b) milliliters (m. L) ___ volume c) Kilograms (Kg) ___ length d) grams/milliliter (g/m. L)

SCIENTIFIC FOUNDATIONS CONT. 5) Chemistry is the study of a) energy b) matter c) math d) life 6) Sam uses her senses to gather information. This is called a) experiment b) conclusion c) observation d) conclusion 7) Explain why it is important for experiments to be “controlled”? 8) Why are SI units important? 9) List 4 examples of SI units. 1) 2) 3)

METRIC PRACTICE 1) Draw a picture of the metric “Ladder” we used last week 2) Which metric measurement would you use to measure the length of a car? a) millimeter b) centimeter c) meter d) kilometer 3) Which does the prefix “Deka” mean? a). 01 b) 1000 4) Convert 45, 786 meters to Hectometers 5) Convert 62 millimeters to Dekameters c) 100 d) 10

VOCAB PRACTICE 1. SCIENCE 2. PHYSICAL SCIENCE 3. ENERGY A. A possible explanation or answer to a question / problem B. A process of gathering knowledge about the natural world 4. MATTER C. Any pieces of information acquired through experimentation 5. SCIENTIFIC METHODS D. The “stuff” that everything is made of 6. OBSERVATIONS E. The ability to do work F. The study of matter and energy 7. HYPOTHESIS 8. DATA G. The ways in which scientists answer questions and solve problems H. Using the senses (when appropriate) to gain information

SCIENTIFIC METHOD PRACTICE Arrange the steps of the scientific method in order. “ 1” is the first step… _____ Analyze Results _____ Repeat the experiment if needed _____ Draw conclusions _____ Form hypothesis _____ Test your hypothesis

9/21 DO NOW 1. Please pick up a Ch 1 Review packet 2. Convert: 9. 83 x 104 = 4. 23 x 10 -3 = 76500. 4 = . 0004364 = 34

9/21 AGENDA 1. Do Now and go over Do Now 2. Lab Safety 3. Measurement Lab HW: Study for Test on Friday Learning Objectives: Apply your learning of metric measurement and conversion in a lab 35

9/22 DO NOW Please have your HW out 1. On a scale of 1 -10, how ready are you for our test tomorrow? 2. What topics are easy for you? Which are you good at? 3. Which topics are you nervous about/hard for you? 36

9/22 AGENDA 1. Do Now and go over Do Now 2. Go over the HW 3. Review Jeopardy game ** Get into a team with the people in your “Column” HW: Study for Test on Friday Learning Objectives: Review 37

9/23 DO NOW TEST DAY! 1. Convert 3, 006. 2 to scientific notation 2. Convert 3. 00 x 10 -4 from scientific notation to standard form 3. Draw and label the steps of the metric ladder 38

9/22 AGENDA 1. Do Now and go over Do Now 2. Turn in your Do Now 3. Questions? From the Review packet? 4. Test HW: None Learning Objectives: Assessment 39

9/26 DO NOW Write down 5 things you recall we learned or talked about last week 40

9/26 AGENDA 1. Do Now and go over Do Now 2. Go Over Ch 1 Test 3. Current Events! 4. No HW Learning Objectives: 41
- Slides: 40