Introduction to Science Chapter 1 Section 1 1

































- Slides: 44

Introduction to Science Chapter 1

Section 1. 1 NATURE OF SCIENCE

How do scientists do science? • Perform experiments to: – Find a new aspect of the natural world – Explain a known phenomenon – Check the results of other experiments – Test the predictions of current theories

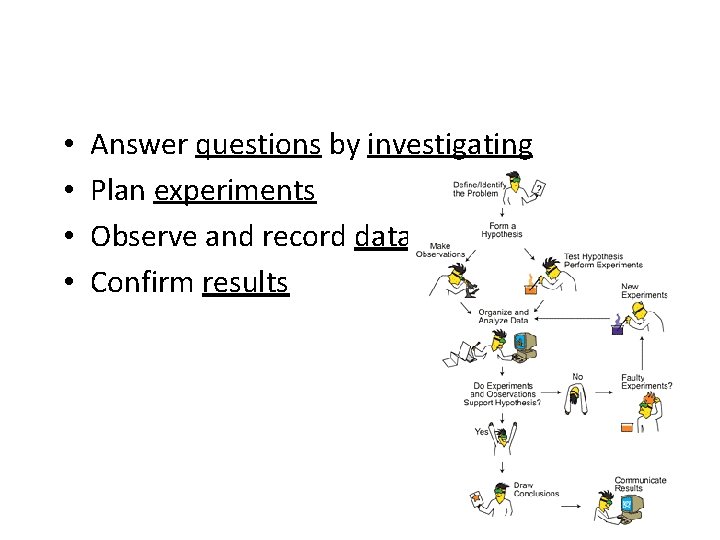
• • Answer questions by investigating Plan experiments Observe and record data Confirm results

The Branches of Science • Biological Science – Science of living things – Examples: botany, zoology, ecology

• Physical Science – Science of matter and energy – Examples: chemistry and physics

• Earth Science – Science of the earth – Examples: geology, meteorology, astronomy

• The branches of science work together. – Biochemistry: study of chemicals that make up living things – Geophysics: study of the forces that affect Earth

• Technology: the application of science for practical uses – Example: computers

Scientific Laws and Theories • Law – Describes a process in nature that can be tested by repeated experiments – Allows predictions to be made about how a system will behave under a wide range of conditions – A descriptive statement or equation that reliably predicts events under certain conditions – Example: E = mc 2

• Theory – An explanation of how a natural process works – A system of ideas that explains many related observations and is supported by a large body of evidence acquired through scientific investigation – Example: atomic theory

• Theories must pass several tests to be considered valid: – Must explain observations clearly and consistently – Experiments that illustrate theory must be repeatable – You must be able to predict results from theory • A theory can never be proved absolutely; there is always the possibility that it will be revised or replaced by a better theory

Quantitative vs. Qualitative Statements • Quantitative – Mathematical equations – Mass, height, length, volume, density, etc. – Example: A = l x w length is 12 m

• Qualitative – Description of something using words – Color, shape, physical characteristics, things you can identify using your senses – Example: color of the object is blue, my books smell old

Scientific Models • A representation of an object or event that can be studied to understand the real object or event • Types: – Drawings on paper – Real objects – Mental “picture” or set of rules that describes how something works.

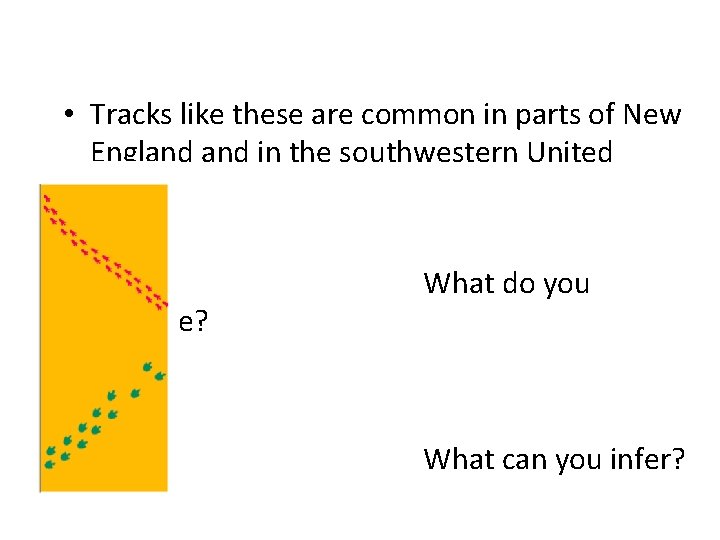
Observation vs. Inference • Observation: Any information collected with the senses. • Inference: Conclusions or deductions based on observations

• Tracks like these are common in parts of New England in the southwestern United States. observe? What do you What can you infer?

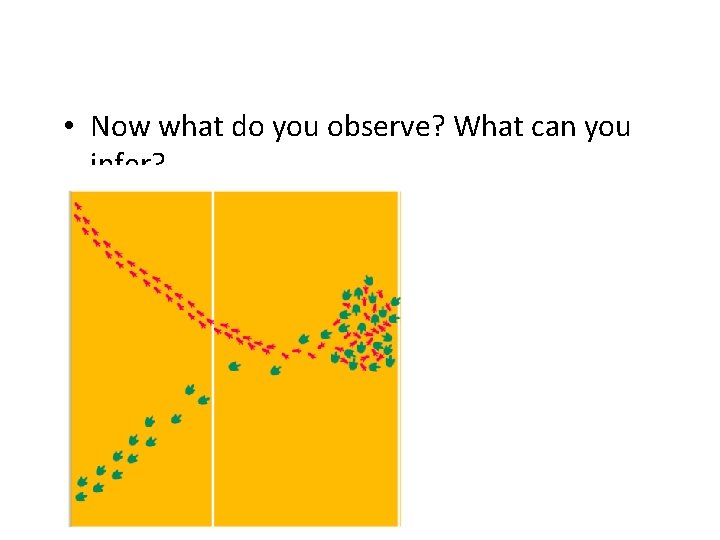
• Now what do you observe? What can you infer?

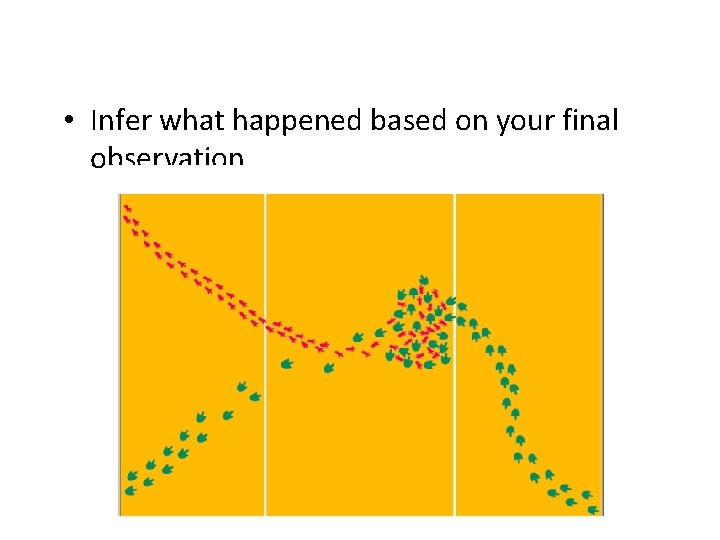
• Infer what happened based on your final observation.

Section 1. 2 THE WAY SCIENCE WORKS

Science Skills • Critical thinking: to approach a problem by asking questions, making observations, and using logic • Identify problems/make observations • Planning experiments • Recording observations • Correctly reporting data

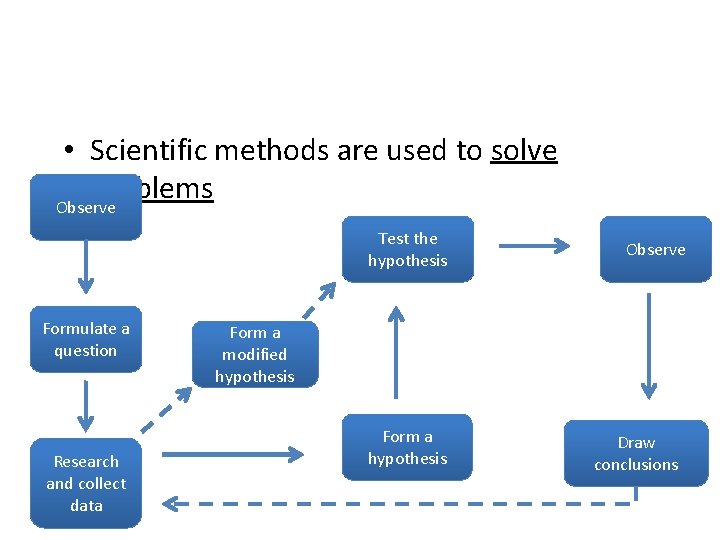
• Scientific methods are used to solve problems Observe Test the hypothesis Formulate a question Research and collect data Observe Form a modified hypothesis Form a hypothesis Draw conclusions

• Variable: factor that changes in an experiment in order to test a hypothesis • Controlled experiment: all variables are kept constant except the one that you want to measure • Example: Suppose your plant is dying and you want to figure out what is killing it. What are the possible things to look for? – Amount of water – Amount of sunlight – Amount of fertilizer These are variables

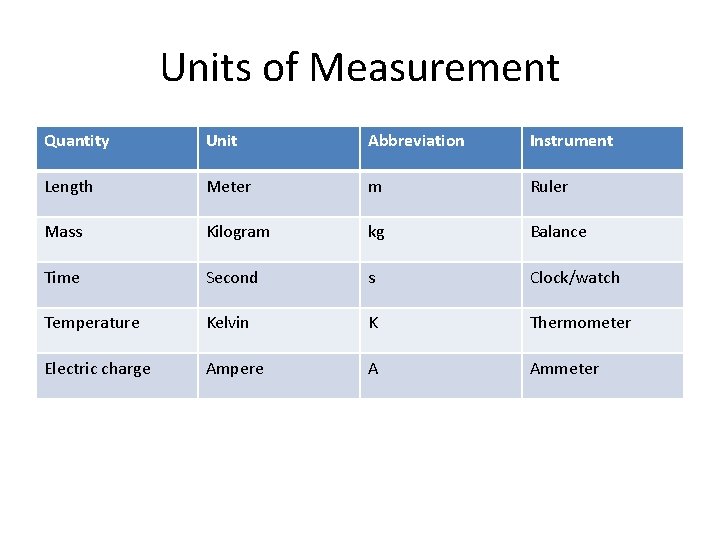
Units of Measurement Quantity Unit Abbreviation Instrument Length Meter m Ruler Mass Kilogram kg Balance Time Second s Clock/watch Temperature Kelvin K Thermometer Electric charge Ampere A Ammeter

Derived Units • • • Combinations of base units Area = Length x Width Volume = Length x Width x Height Pressure Force Speed

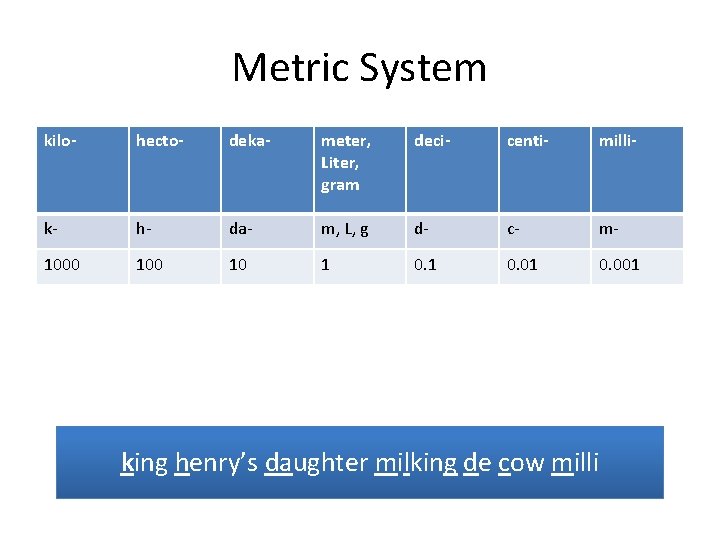
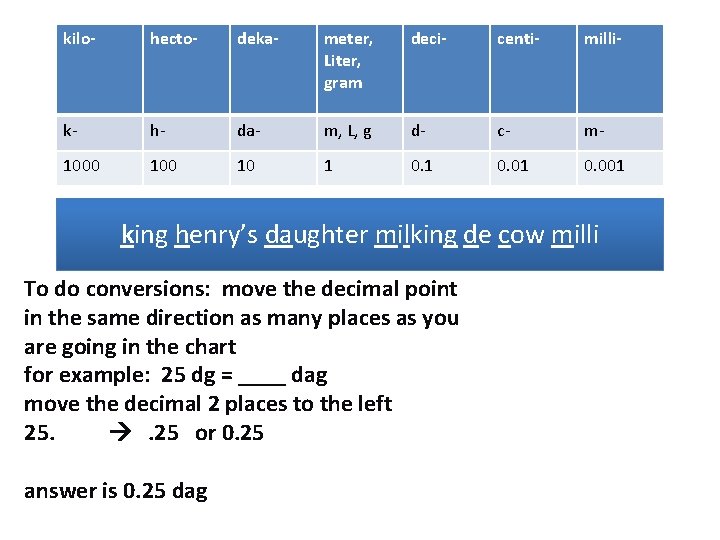
Metric System kilo- hecto- deka- meter, Liter, gram deci- centi- milli- k- h- da- m, L, g d- c- m- 1000 10 1 0. 01 0. 001 king henry’s daughter milking de cow milli

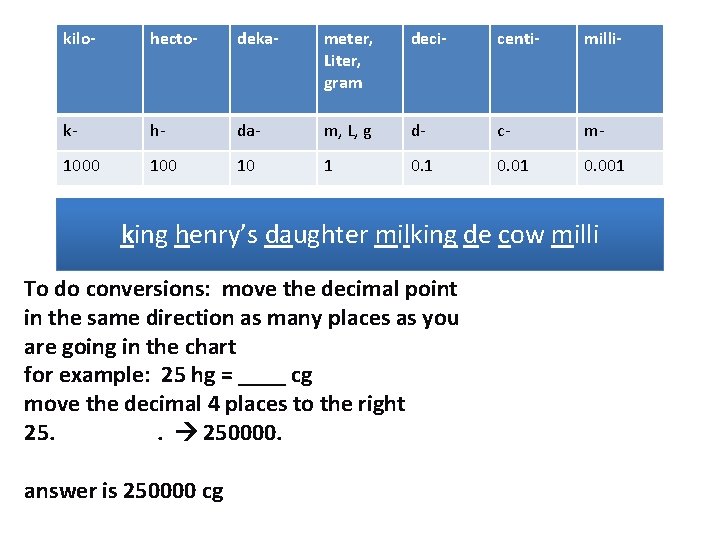
kilo- hecto- deka- meter, Liter, gram deci- centi- milli- k- h- da- m, L, g d- c- m- 1000 10 1 0. 01 0. 001 king henry’s daughter milking de cow milli To do conversions: move the decimal point in the same direction as many places as you are going in the chart for example: 25 hg = ____ cg move the decimal 4 places to the right 25. . 250000. answer is 250000 cg

kilo- hecto- deka- meter, Liter, gram deci- centi- milli- k- h- da- m, L, g d- c- m- 1000 10 1 0. 01 0. 001 king henry’s daughter milking de cow milli To do conversions: move the decimal point in the same direction as many places as you are going in the chart for example: 25 dg = ____ dag move the decimal 2 places to the left 25. . 25 or 0. 25 answer is 0. 25 dag

king henry’s daughter milking de cow milli Metric Conversions 1. ) 1 m = ___ mm 1000 mm 2. ) 3 mm = ___ cm 0. 3 cm 3. ) 43 cm = ___ dm 4. 3 4. ) 2. 5 km = ___ m 2500

king henry’s daughter milking de cow milli 5) 0. 5 m = ___ cm 50 cm 6) 0. 9 cm = ___ mm 9 mm 7) 15 cm = ___ m 0. 15 m 8) 9. 8 mm = ___ m 0. 00098

king henry’s daughter milking de cow milli 9) 10) 11) 12) 13) 14) 15) 16) 0. 03 cm = _______ mm 73 m = _______ km 1. 7 g = _______ cg 24 mg = _______ g 0. 05 cg = _______ mg 0. 00006 g = ____ cg 0. 03 kg = _______ g 215 mg = ____ g 9. ) 10. ) 11. ) 12. ) 13. ) 14. ) 15. ) 16. ) 0. 3 mm 0. 073 km 1700 cg 0. 024 g 0. 5 mg 0. 006 cg 30 g 0. 215 g k – h – da – (m, g, L) – d – c – m

king henry’s daughter milking de cow milli 17) 18) 19) 20) 21) 22) 23) 24) 4. 9 cg = _____ mg 0. 7 g = _____ cg 300 m. L = ______ L 2500 m. L = ______ L 500 m. L = ______ d. L 3. 75 m. L = ______ c. L 2. 5 g = _____ mg 3. 07 m. L = _______ L 17. ) 18. ) 19. ) 20. ) 21. ) 22. ) 23. ) 24. ) 49 mg 70 cg 0. 300 L 2. 500 L 5 d. L 0. 375 c. L 2500 mg 0. 00307 L k – h – da – (m, g, L) – d – c – m

king henry’s daughter milking de cow milli 25) 10. 76 m = _______ km 26) 27) 28) 29) 30) 31) 32) 760 cm = _____ dm 45 m. L = ______ da. L 1. 45 hg = ______ g 0. 07 L = _______ k. L 62 dam = ______ cm 71 kg = _____ hg 22 cm= _______ mm 25. ) 26. ) 27. ) 28. ) 29. ) 30. ) 31. ) 32. ) 0. 01076 km 76 dm 0. 0045 da. L 145 g 0. 0007 k. L 62000 cm 710 hg 220 mm k – h – da – (m, g, L) – d – c – m

king henry’s daughter milking de cow milli 33) 34) 35) 36) 37) 38) 39) 40) 17 dm = _______ m 4. 8 kg= _____ g 4 L = ______ m. L 73 cm= ______ mm 420 mm= _______ km 79 kg= ____ dg 59 cg = ____ kg 7. 5 kg = _______ mg 33. ) 34. ) 35. ) 36. ) 37. ) 38. ) 39. ) 40. ) 1. 7 m 4800 g 4000 m. L 730 mm 0. 000420 km 790000 dg 0. 00059 kg 7500000 mg k – h – da – (m, g, L) – d – c – m

king henry’s daughter milking de cow milli 41) 42) 43) 44) 10000 m = _______ km 25 mg= ____ kg 250 m. L = ______ L 425 cg = _______ kg 41. ) 42. ) 43. ) 44. ) 10 km 0. 00025 kg 0. 250 L 0. 00425 kg k – h – da – (m, g, L) – d – c – m

Section 1. 3 ORGANIZING DATA

Presenting Scientific Data • Organizing and presenting data are important science skills to have when you share your results with others • Independent Variable: the variable you control in an experiment; goes on x axis • Dependent Variable: depends on changes to the independent variable; goes on y axis

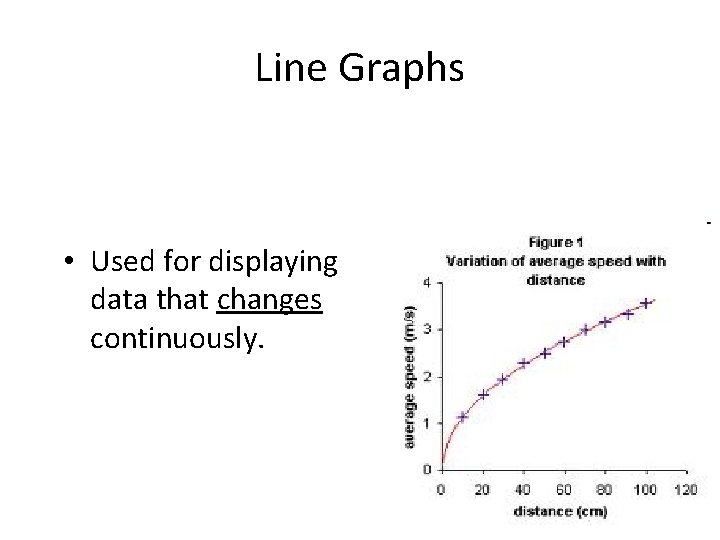
Line Graphs • Used for displaying data that changes continuously.

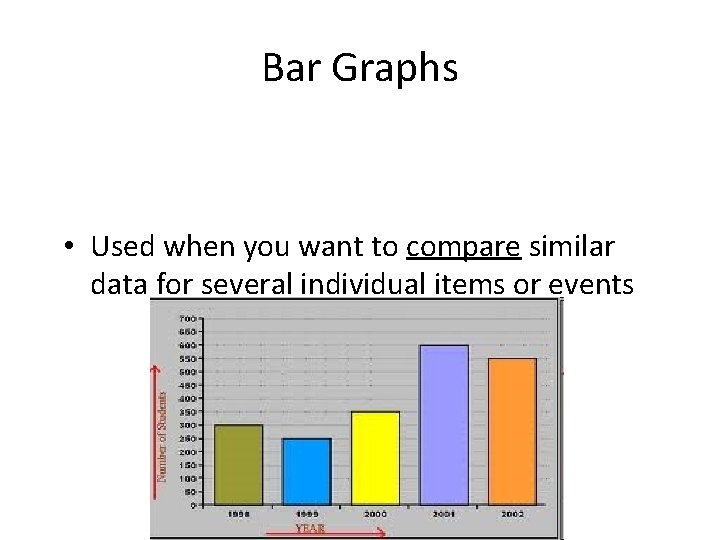
Bar Graphs • Used when you want to compare similar data for several individual items or events

Pie Graphs • Used for displaying data that are parts of a whole

Scientific Notation • A method of expressing a quantity as a number multiplied by 10 to an appropriate power • 2500 becomes 2. 5 x 103 • 0. 00036 becomes 3. 6 x 10 -4 • 9. 8 x 106 is? 9 800 000 • 2. 3 x 10 -2 is? 0. 023


Accuracy vs. Precision • Accuracy: a description of how close a measurement is to the true value of the quantity measured • Precision: the exactness of a measurement – how close a set of measurements are to each other

not accurate not precise not accurate precise