Introduction to Power Point Lectures for Biotechnology Second

Introduction to Power. Point Lectures for Biotechnology, Second Edition William J. Thieman and Michael A. Palladino Chapter 4 Proteins as Products Lectures by Lara Dowland Copyright © 2009 Pearson Education, Inc.

Chapter Contents • • • 4. 1 4. 2 4. 3 4. 4 4. 5 4. 6 4. 7 4. 8 4. 9 4. 10 Introduction to Proteins as Biotech Products Proteins as Biotechnology Products Protein Structures Protein Production Protein Purification Methods Verification Preserving Proteins Scale-Up of Protein Purification Postpurification Analysis Methods Proteomics Copyright © 2009 Pearson Education, Inc.

4. 1 Introduction to Proteins as Biotech Products • Proteins – large molecules that are required for the structure, function, and regulation of living cells • 2000 NIH launched Protein Structure Initiative – Effort to identify the structure of human proteins Copyright © 2009 Pearson Education, Inc.

4. 2 Proteins as Biotechnology Products • Use of proteins in manufacturing is a time-tested technology – Beer brewing and winemaking – Cheese making • Recombinant DNA technology made it possible to produce specific proteins on demand – Enzymes – proteins that speed up chemical reactions – Hormones – Antibodies Copyright © 2009 Pearson Education, Inc.

4. 2 Proteins as Biotechnology Products • Making a Biotech Drug – Produced through microbial fermentation or mammalian cell culture – Complicated and time-consuming process – Must strictly comply with FDA regulations at all stages of the procedure Copyright © 2009 Pearson Education, Inc.

4. 2 Proteins as Biotechnology Products • Applications of Proteins in Industry – – – – Medical applications Food processing Textiles and leather goods Detergents Paper manufacturing and recycling Adhesives: natural glues Bioremediation: treating pollution with proteins Copyright © 2009 Pearson Education, Inc.

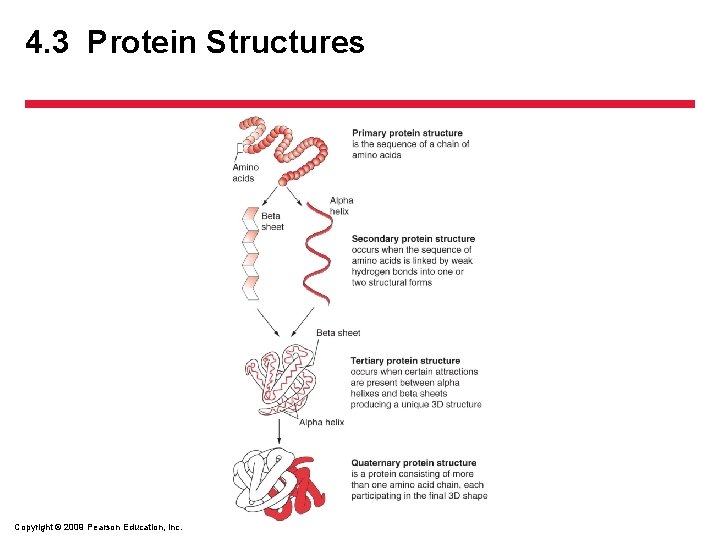
4. 3 Protein Structures • Proteins – Are complex molecules built of chains of amino acids – Have electrical charge that causes them to interact with other atoms and molecules • Hydrophilic – water loving • Hydrophobic – water hating Copyright © 2009 Pearson Education, Inc.

4. 3 Protein Structures • Structural Arrangement – four levels – Primary structure is the sequence in which amino acids are linked together – Secondary structure occurs when chains of amino acids fold or twist at specific points • Alpha helices and beta sheets – Tertiary structures are formed when secondary structures combine and are bound together – Quaternary structures are unique, globular, threedimensional complexes built of several polypeptides Copyright © 2009 Pearson Education, Inc.
4. 3 Protein Structures Copyright © 2009 Pearson Education, Inc.

4. 3 Protein Structures • Protein Folding – The structure and function of a protein depends on protein folding – If protein is folded incorrectly, desired function of a protein is lost and a misfolded protein can be detrimental – 1951 two regular structures were described • Alpha helices and beta sheets • Structures are fragile; hydrogen bonds are easily broken Copyright © 2009 Pearson Education, Inc.

4. 3 Protein Structures • Glycosylation – post-translational modification wherein carbohydrate units are added to specific locations on proteins • More than 100 post-translational modifications occur Copyright © 2009 Pearson Education, Inc.

4. 3 Protein Structures • Protein Engineering – Introducing specific, predefined alterations in the amino acid sequence through a process known as directed molecular evolution technology – Creating entirely new protein molecules Copyright © 2009 Pearson Education, Inc.

4. 4 Protein Production • Proteins are valuable • Proteins are complex and fragile products • Production of proteins is a long and painstaking process – Upstream processing includes the actual expression of the protein in the cell – Downstream processing involves purification of the protein and verification of the function; a stable means of preserving the protein is also required Copyright © 2009 Pearson Education, Inc.

4. 4 Protein Production • Protein Expression: The First Phase in Protein Processing – Selecting the cell to be used as a protein source • • • Microorganisms Fungi Plants Mammalian cell systems Whole-animal production systems Insect systems Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Protein Must Be Harvested – Entire cell is harvested if protein is intracellular • Requires cell lysis to release the protein • Releases the entire contents of the cell – Culture medium is collected if the protein is extracellular Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Separating the Components in the Extract – Similarities between proteins allow the separation of proteins from non-protein material • Protein precipitation – salts cause proteins to settle out of solution • Filtration (size-based) separation methods – Centrifugation – Membrane filtration – Microfiltration – Ultrafiltration Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Separating the Components in the Extract • Diafiltration and dialysis rely on the chemical concept of equilibrium Copyright © 2009 Pearson Education, Inc.

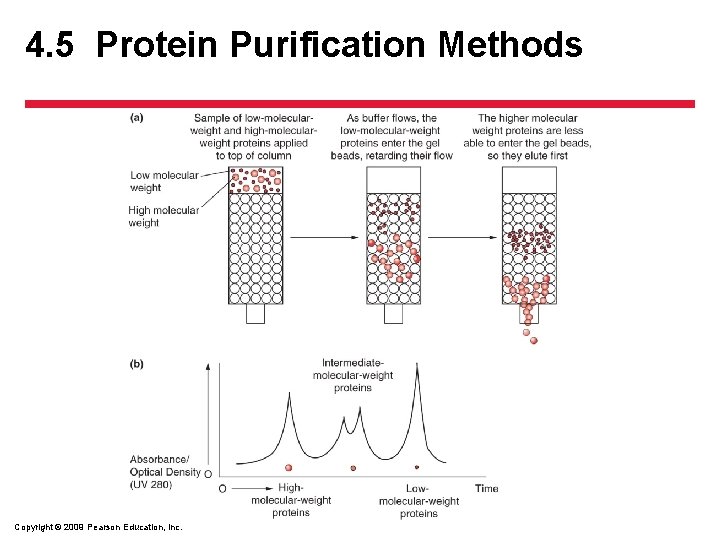
4. 5 Protein Purification Methods • Separating the Components in the Extract – Differences in proteins allows the separation of the target protein from other proteins • Chromatography – allows the sorting of proteins based on size or by how they cling to or dissolve in various substances Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Separating the Components in the Extract – Chromatography • Size exclusion chromatography (SEC) – uses gel beads with pores – Larger proteins move quickly around the beads and smaller proteins slip through the pores and therefore move more slowly through the beads Copyright © 2009 Pearson Education, Inc.
4. 5 Protein Purification Methods Copyright © 2009 Pearson Education, Inc.

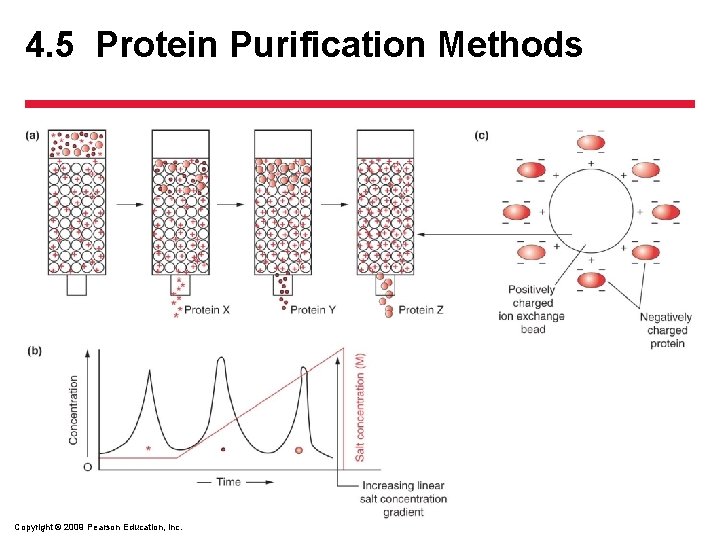
4. 5 Protein Purification Methods • Separating the Components in the Extract – Chromatography • Ion exchange chromatography – relies on the charge of the protein – Resin is charged – Opposite charged proteins will stick to resin beads – Can be eluted by changing the charge with salts of increasing concentration Copyright © 2009 Pearson Education, Inc.
4. 5 Protein Purification Methods Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Separating the Components in the Extract – Chromatography • Affinity chromatography relies on the ability of proteins to bind specifically and reversibly to uniquely shaped compounds called ligands Copyright © 2009 Pearson Education, Inc.

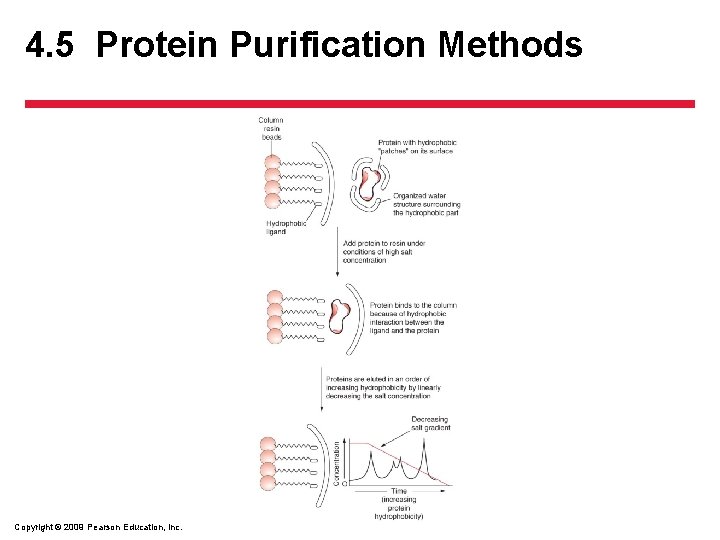
4. 5 Protein Purification Methods • Separating the Components in the Extract – Chromatography • Hydrophobic interaction chromatography (HIC) sorts proteins on the basis of their repulsion of water Copyright © 2009 Pearson Education, Inc.
4. 5 Protein Purification Methods Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Separating the Components in the Extract – Iso-electric focusing used in QC to identify two similar proteins that are difficult to separate by any other means • Each protein has a specific number of charged amino acids on its surface in specific places • Creates a unique electric signature known as its iso-electric point (IEP) where charges on the protein match the p. H of the solution Copyright © 2009 Pearson Education, Inc.

4. 5 Protein Purification Methods • Separating the Components in the Extract – Analytic methods • High-Performance liquid chromatography (HPLC) – uses high pressure to force the extract through the column in a shorter time • Mass spectrometry (mass spec) – highly sensitive method used to detect trace elements – Used to indicate the size and identity of most protein fragments Copyright © 2009 Pearson Education, Inc.

4. 6 Verification • The presence and concentration of the protein of interest must be verified at each step of the purification process – SDS-PAGE (polyacrylamide gel electrophoresis) – Western blotting – ELISA Copyright © 2009 Pearson Education, Inc.

4. 7 Preserving Proteins • Lyophilization (freeze-drying) – Protein, usually a liquid product, is first frozen – A vacuum is used to hasten the evaporation of water from the fluid – Will maintain protein structure and can be stored at room temperature for long periods of time Copyright © 2009 Pearson Education, Inc.

4. 8 Scale-Up of Protein Purification • Protocols are usually designed in the laboratory on a small scale • Must be scaled up for production – Process is approved by FDA so must make sure laboratory procedures can be scaled up Copyright © 2009 Pearson Education, Inc.

4. 9 Postpurification Analysis Methods • Protein Sequencing – Must determine the primary structure, the sequence of amino acids • X-ray Crystallography – Used to determine the complex tertiary and quaternary structures Copyright © 2009 Pearson Education, Inc.

4. 10 Proteomics • A new scientific discipline dedicated to understanding the complex relationship of disease and protein expression – Uses protein microarrays to test variation in protein expression between healthy and disease states Copyright © 2009 Pearson Education, Inc.
- Slides: 32