Introduction to Polymers Overview What are polymers How









- Slides: 25

Introduction to Polymers

Overview • What are polymers? • How are polymers named? • What is a polymer crosslinker?

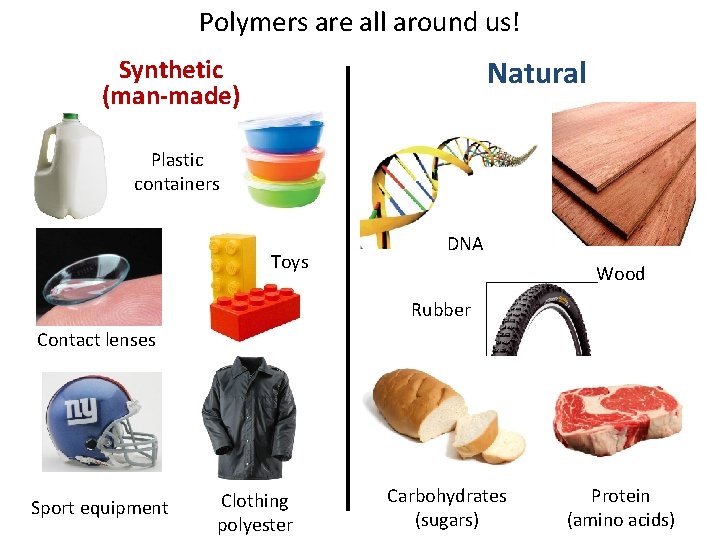
Polymers are all around us! Natural Synthetic (man-made) Plastic containers Toys DNA Wood Rubber Contact lenses Sport equipment Clothing polyester Carbohydrates (sugars) Protein (amino acids)

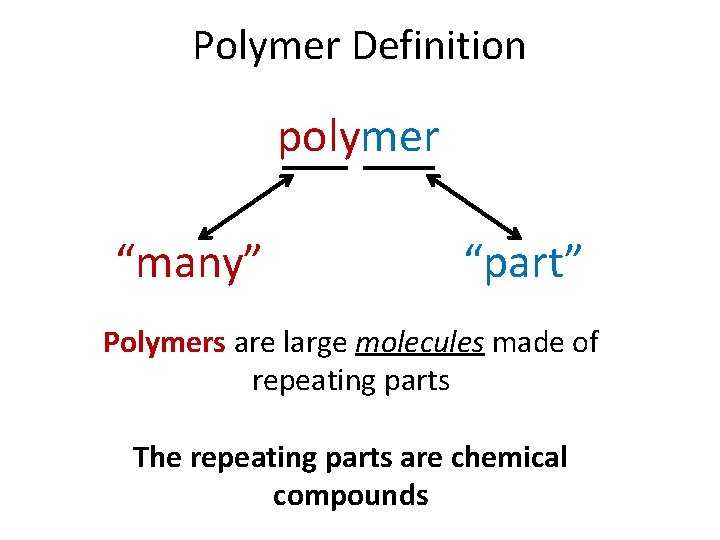
Polymer Definition polymer “many” “part” Polymers are large molecules made of repeating parts The repeating parts are chemical compounds

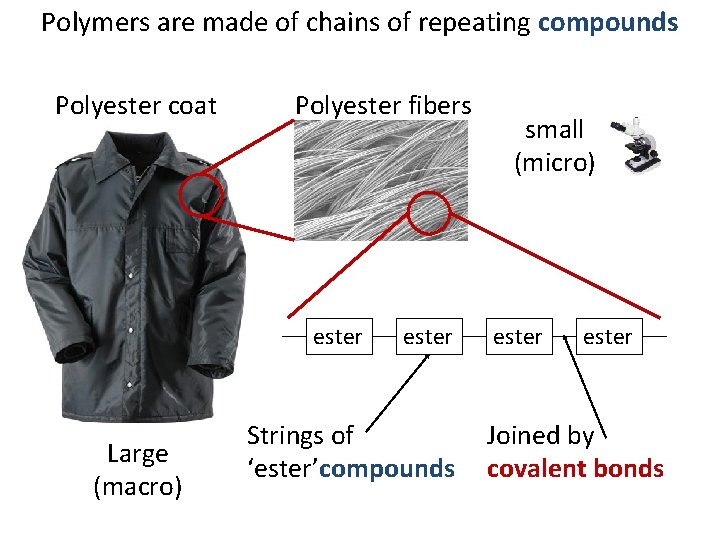
Polymers are made of chains of repeating compounds Polyester coat Large (macro) Polyester fibers small (micro)

Polymers are made of chains of repeating compounds Polyester coat Polyester fibers ester Large (macro) ester Strings of ‘ester’compounds small (micro) ester Joined by covalent bonds

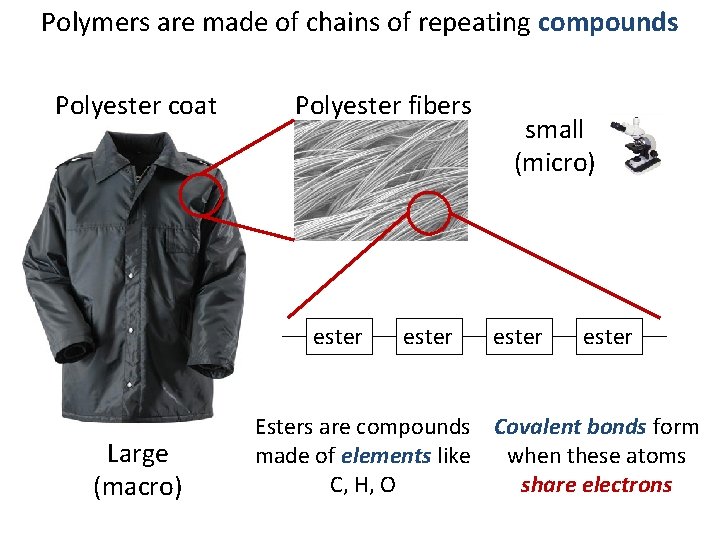
Polymers are made of chains of repeating compounds Polyester coat Polyester fibers ester Large (macro) ester small (micro) ester Esters are compounds Covalent bonds form made of elements like when these atoms C, H, O share electrons

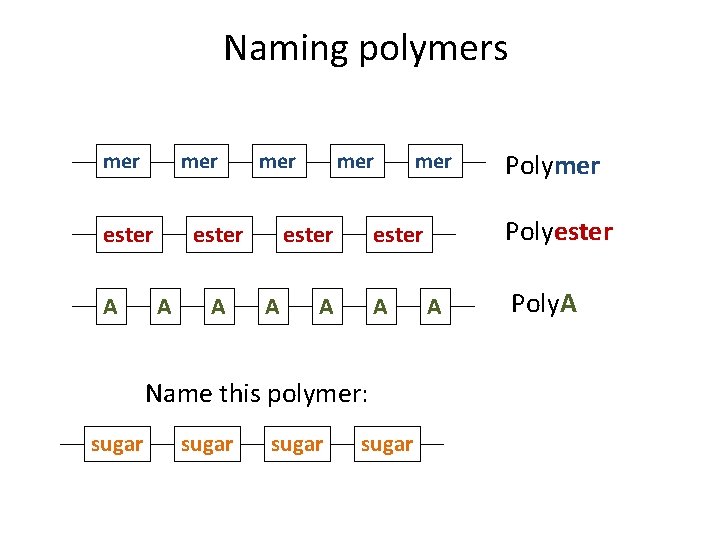
Naming polymers mer ester A A A sugar Polymer Polyester Name this polymer: sugar mer A Poly. A

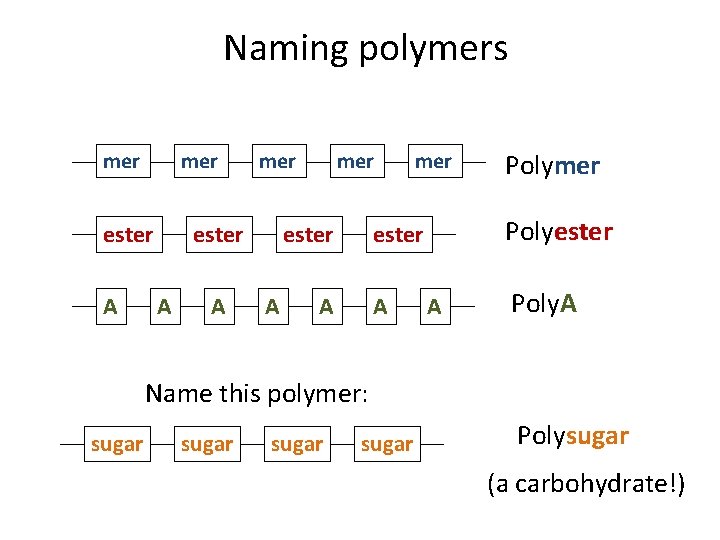
Naming polymers mer ester A mer Polyester A A Polymer A Poly. A Name this polymer: sugar Polysugar (a carbohydrate!)

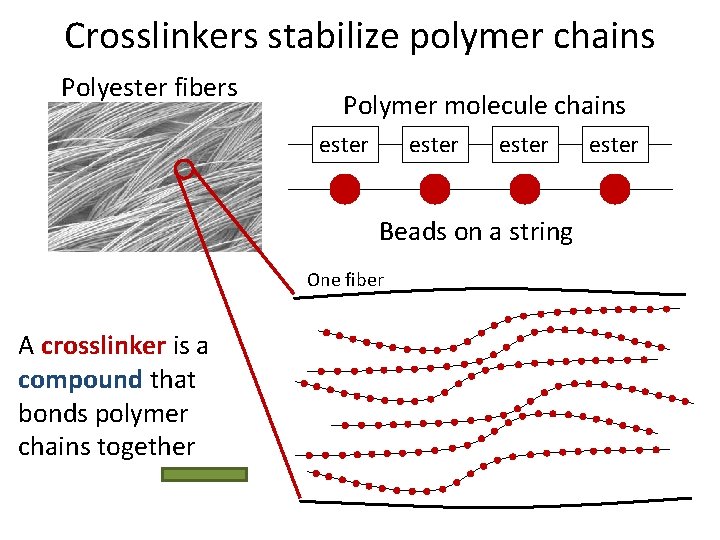
Crosslinkers stabilize polymer chains Polyester fibers Polymer molecule chains ester Beads on a string One fiber A crosslinker is a compound that bonds polymer chains together ester

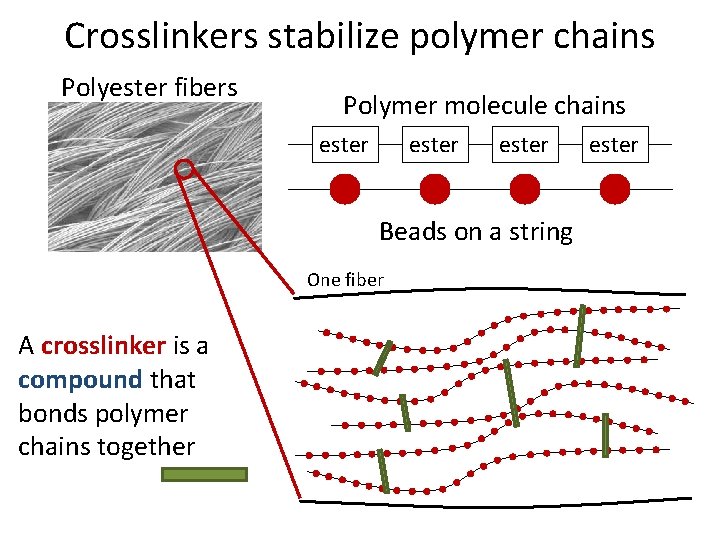
Crosslinkers stabilize polymer chains Polyester fibers Polymer molecule chains ester Beads on a string One fiber A crosslinker is a compound that bonds polymer chains together ester

What are some properties of polymers? vs. How can we change the properties of polymers?

The chemical properties of repeat units and their crosslinkers determine the physical properties of polymers • Density • Melting temperature See-through Flexible • Transparency • Stiffness Can’t see through Flexible Can’t see through Rigid, stiff

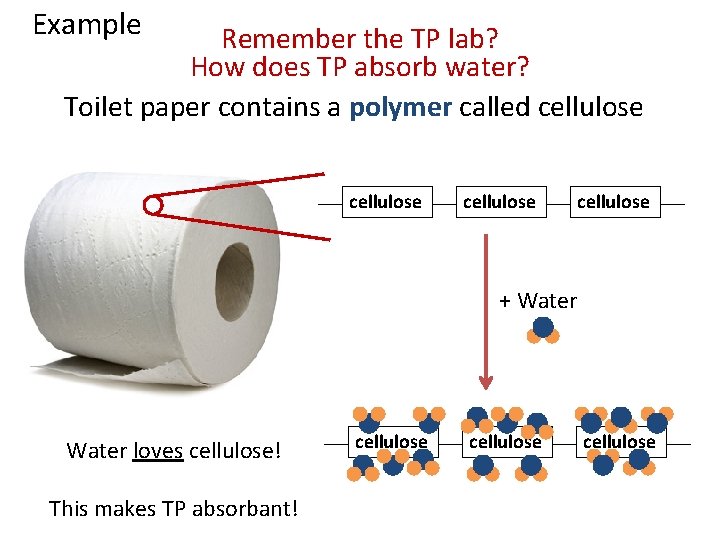
Example Remember the TP lab? How does TP absorb water? Toilet paper contains a polymer called cellulose + Water Remember, paper comes from wood, a natural polymer!

Example Remember the TP lab? How does TP absorb water? Toilet paper contains a polymer called cellulose + Water loves cellulose! This makes TP absorbant! cellulose

Example Remember the TP lab? How does TP absorb water? So, the chemical properties of a polymer (cellulose) in paper help determine its physical properties (absorbancy)

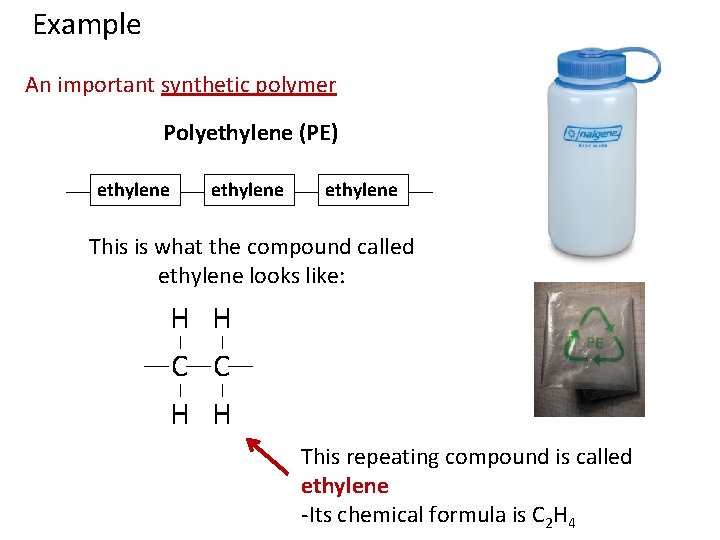
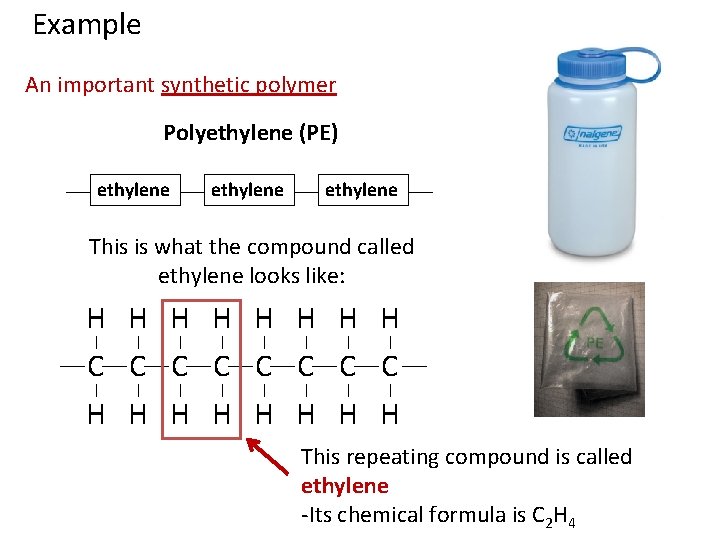
Example An important synthetic polymer Polyethylene (PE) ethylene

Example An important synthetic polymer Polyethylene (PE) ethylene This is what the compound called ethylene looks like: H H C C H H This repeating compound is called ethylene -Its chemical formula is C 2 H 4

Example An important synthetic polymer Polyethylene (PE) ethylene This is what the compound called ethylene looks like: H H H H C C C C H H H H This repeating compound is called ethylene -Its chemical formula is C 2 H 4

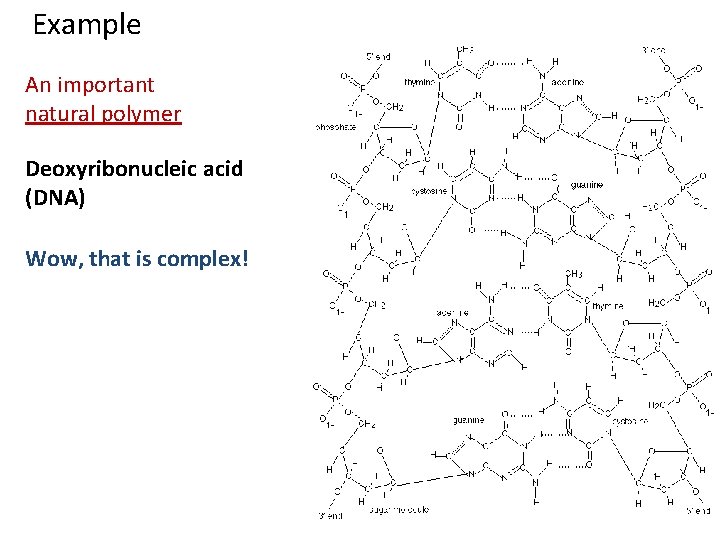
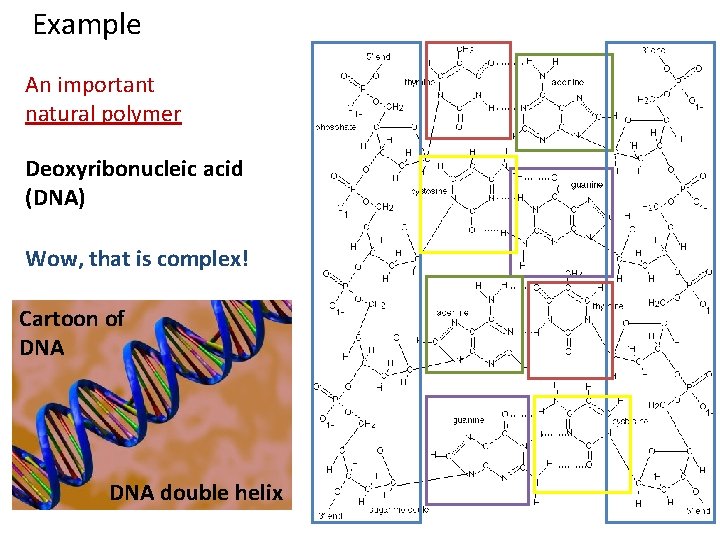
Example An important natural polymer Deoxyribonucleic acid (DNA) Wow, that is complex!

Example An important natural polymer Deoxyribonucleic acid (DNA) Wow, that is complex! Cartoon of DNA double helix

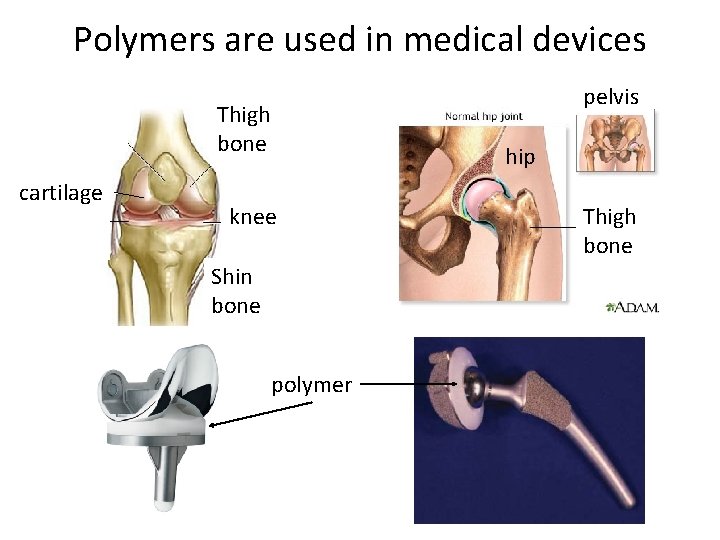
Polymers are used in medical devices pelvis Thigh bone cartilage hip knee Shin bone polymer Thigh bone

Polymer Synthesis Lab!

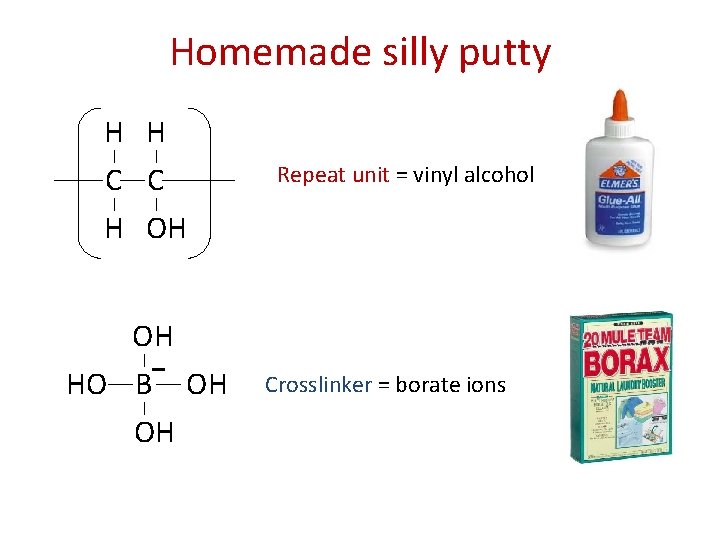
Homemade silly putty H H C C H OH OH HO B OH OH Repeat unit = vinyl alcohol Crosslinker = borate ions

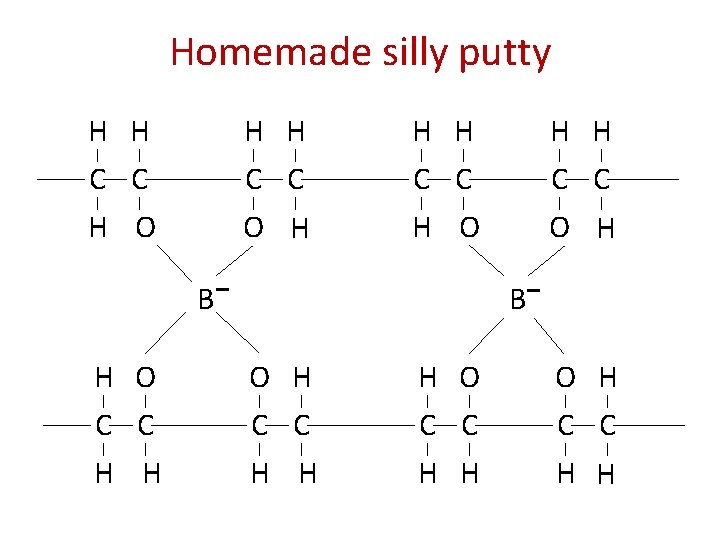
Homemade silly putty H H H H C C H O C C O H B H O C C H H B O H C C H H H O C C H H O H C C H H