Introduction to polymer science Polymer synthesis and chemical















- Slides: 39

高分子科学导论 Introduction to polymer science 高分子的合成与化学性能 Polymer synthesis and chemical properties 第一讲 四川大学 化学学院

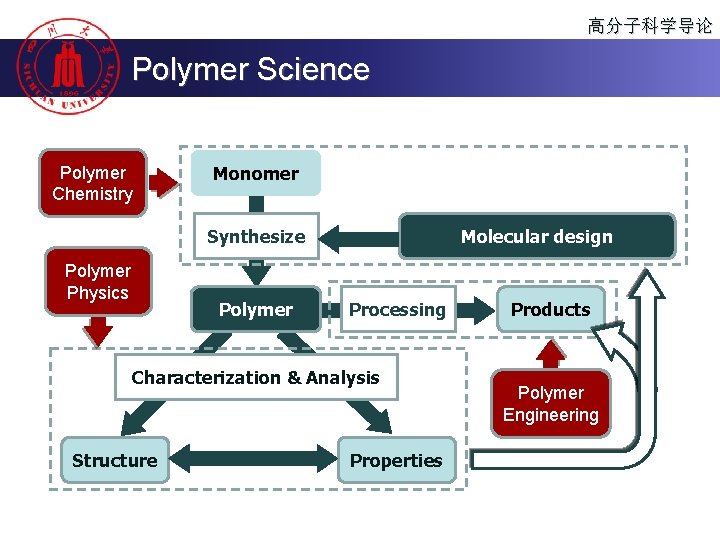
高分子科学导论 Polymer Science Polymer Chemistry Monomer Molecular design Synthesize Polymer Physics Polymer Processing Characterization & Analysis Structure Properties Products Polymer Engineering

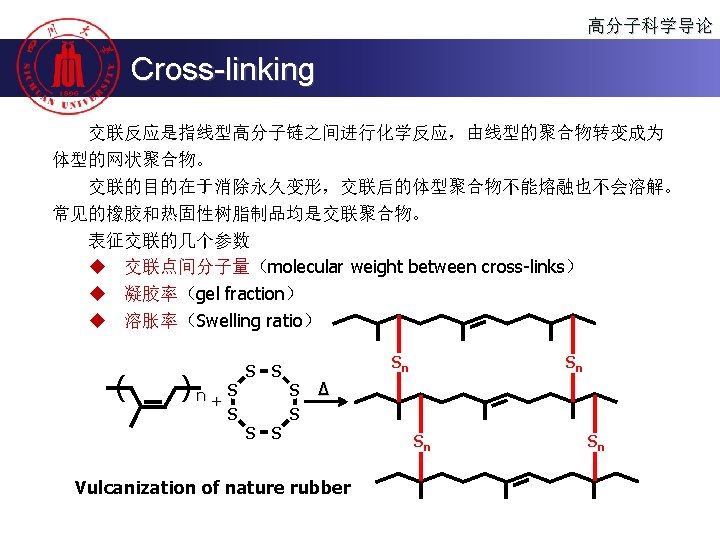
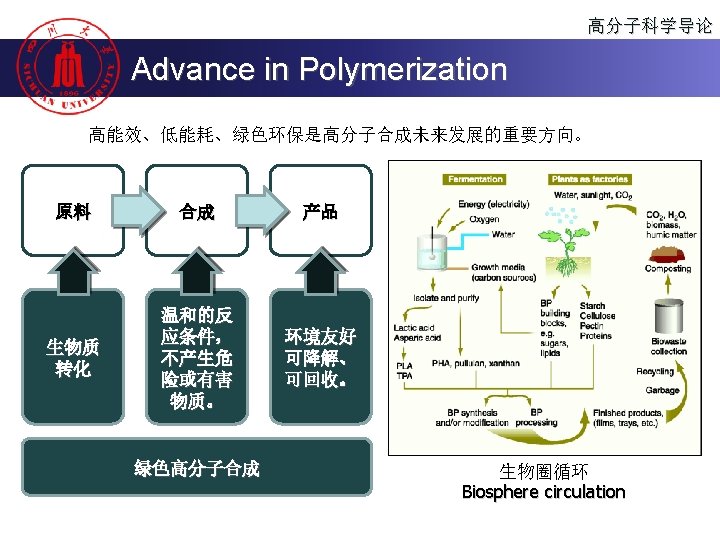
高分子科学导论 Content n 高分子的聚合方法 Polymerization n 聚合物的分子设计 Molecular design of the polymer n 聚合物的化学反应 Reactions of the Polymer n 高分子的老化、降解与回收 Polymer aging, degradation and recycling


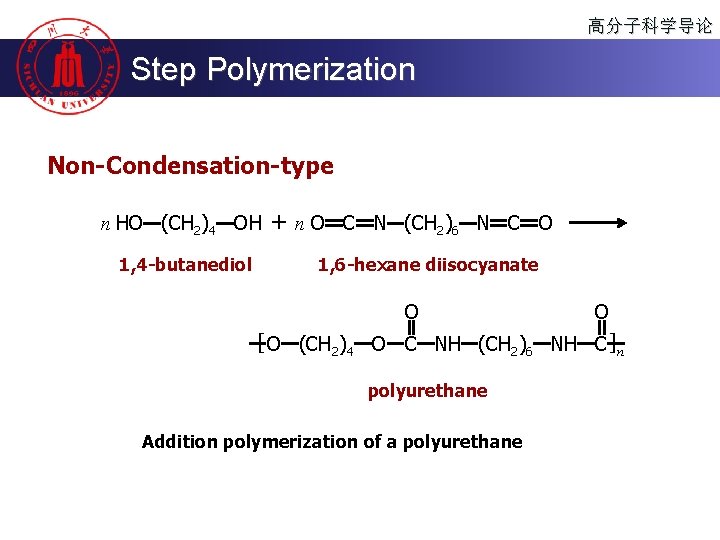
高分子科学导论 Step Polymerization Non-Condensation-type n HO (CH 2)4 OH 1, 4 -butanediol + n O C N (CH 2)6 N C O 1, 6 -hexane diisocyanate O O [ O (CH 2)4 O C NH (CH 2)6 NH C ]n polyurethane Addition polymerization of a polyurethane

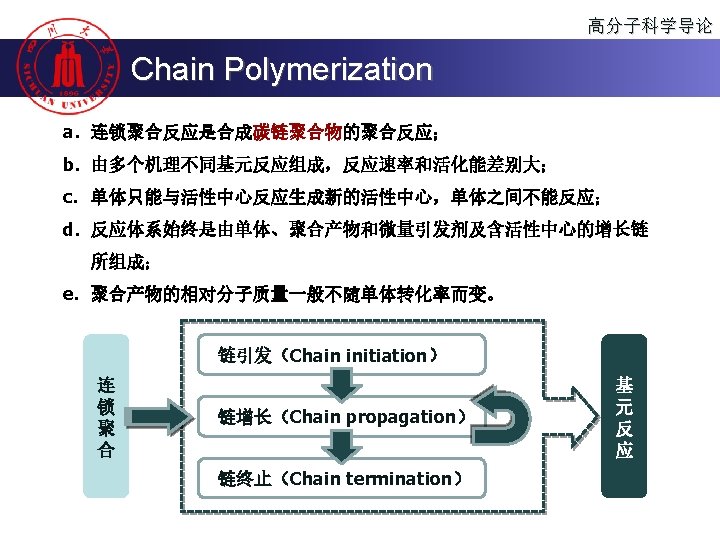
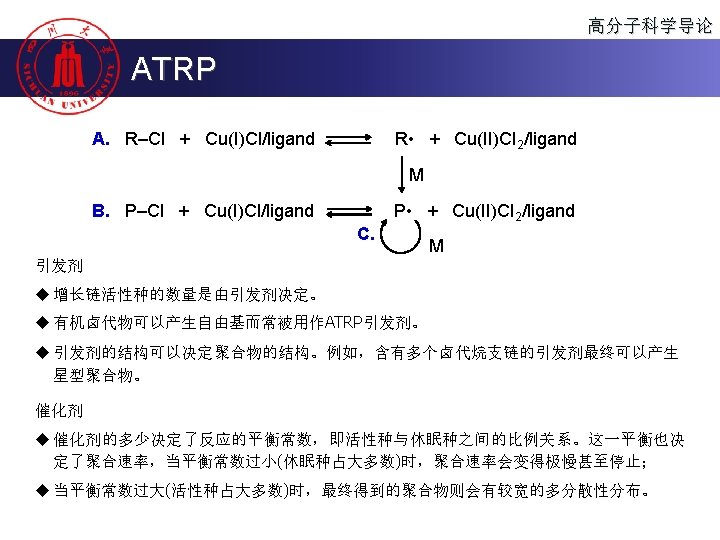
高分子科学导论 Polymerization 按聚合机理或动力学分类 u 连锁聚合(chain polymerization) u Chain (growth) polymerization is a polymerization technique where unsaturated monomer molecules add on to a growing polymer chain one at a time. It can be represented with the chemical equation: u n. M ( M )n u 逐步聚合(Step polymerization) u Step (growth) polymerization refers to a type of polymerization mechanism in which bi-functional or multifunctional monomers react to form first dimers, then trimers, longer oligomers and eventually long chain polymers.



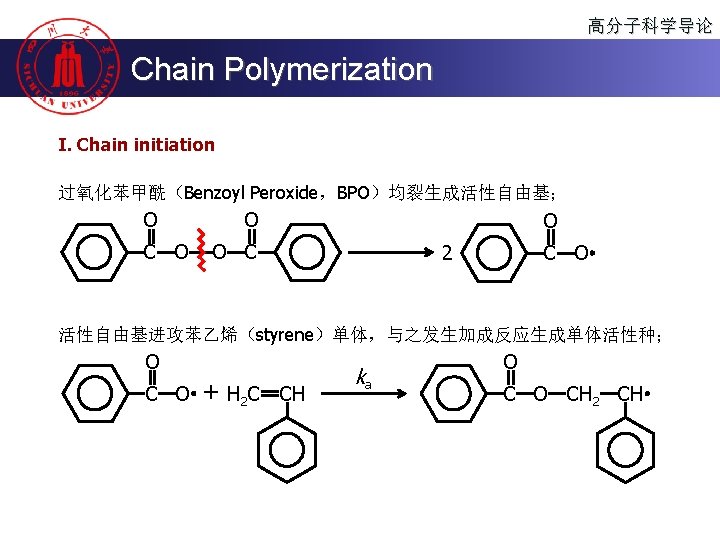
高分子科学导论 Chain Polymerization 烯类单体 u 自由基聚合 CH 2=CH 2 聚合类型 自由基 阴离子 阳离子 + + free radical polymerization CH 2=CH 2 CH 3 u 阳离子聚合 cationic polymerization u 阴离子聚合 anionic polymerization u 配位聚合 配位 + CH 2=CHCH=CH 2 + + ∆ + CH 2=C(CH 3)CH=CH 2 ∆ + CH 2=CHC 6 H 5 + ∆ ∆ ∆ CH 2=CHCl + CH 2=CCl 2 + CH 2=CHF + CH 2=CF 2 + coordination polymerization CH 2=CHOCOCH 3 CH 2=C(CH 3)COOCH 3 CH 2=CHCN ∆: available +: industrialized CH 2=CH-OR ∆ ∆ + + ∆ ∆ + ∆


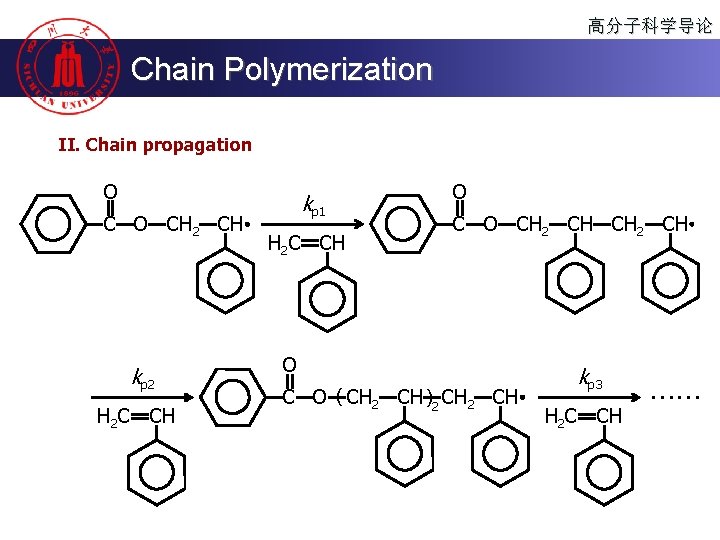
高分子科学导论 Chain Polymerization II. Chain propagation O CH 2 CH kp 2 H 2 C CH kp 1 H 2 C CH O CH 2 CH O C O ( CH 2 CH )2 CH kp 3 H 2 C CH ……

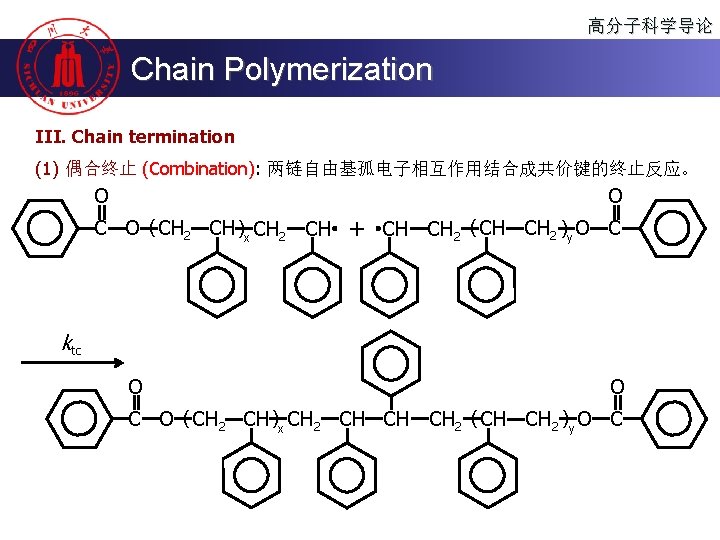
高分子科学导论 Chain Polymerization III. Chain termination (1) 偶合终止 (Combination): 两链自由基孤电子相互作用结合成共价键的终止反应。 O O C O ( CH 2 CH )x CH 2 CH + CH CH 2 ( CH CH 2 )y O C ktc O O C O ( CH 2 CH )x CH 2 CH CH CH 2 ( CH CH 2 )y O C

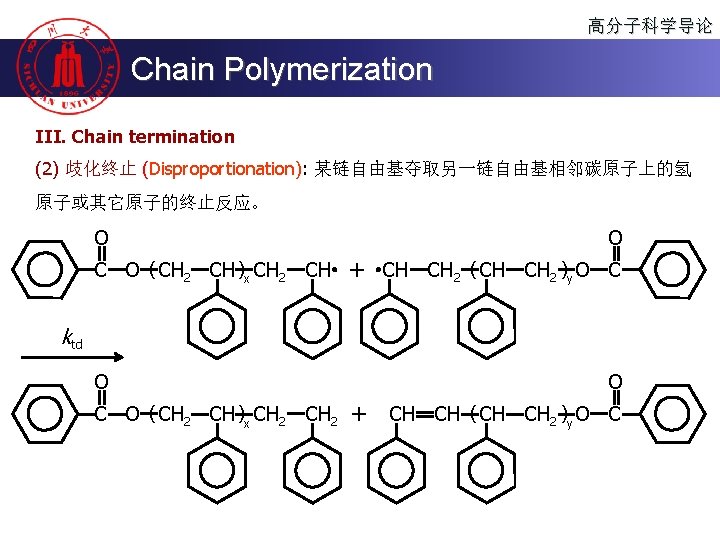
高分子科学导论 Chain Polymerization III. Chain termination (2) 歧化终止 (Disproportionation): 某链自由基夺取另一链自由基相邻碳原子上的氢 原子或其它原子的终止反应。 O C O ( CH 2 CH )x CH 2 CH O + CH CH 2 ( CH CH 2 )y O C ktd O C O ( CH 2 CH )x CH 2 O + CH CH ( CH CH 2 )y O C



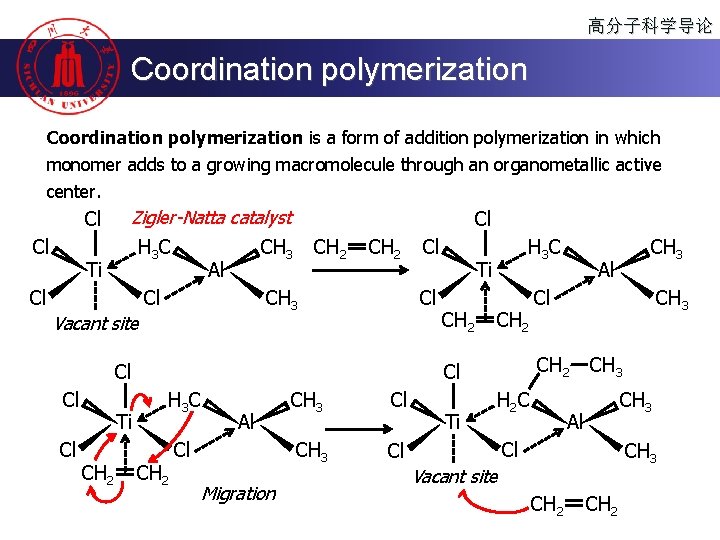
高分子科学导论 Coordination polymerization is a form of addition polymerization in which monomer adds to a growing macromolecule through an organometallic active center. Cl Cl Zigler-Natta catalyst H 3 C Ti CH 3 Al Cl CH 2 CH 3 Vacant site Cl Cl Ti CH 2 Cl H 3 C Ti CH 2 H 3 C CH 2 Al Cl Migration CH 3 Cl Ti Al Cl CH 3 CH 2 Cl CH 3 H 2 C CH 3 Al Cl CH 3 Vacant site CH 2

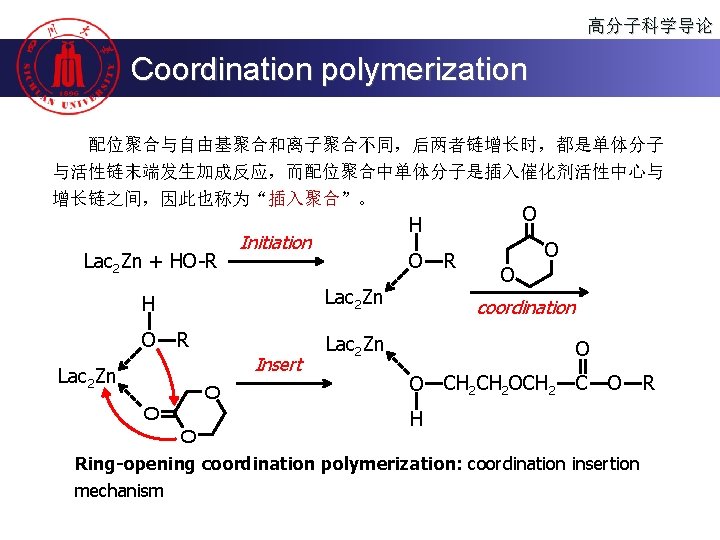
高分子科学导论 Coordination polymerization 配位聚合与自由基聚合和离子聚合不同,后两者链增长时,都是单体分子 与活性链末端发生加成反应,而配位聚合中单体分子是插入催化剂活性中心与 增长链之间,因此也称为“插入聚合”。 Lac 2 Zn + HO-R Initiation O O Lac 2 Zn H R Insert O O O Lac 2 Zn O H R O O coordination Lac 2 Zn O O CH 2 OCH 2 C O H Ring-opening coordination polymerization: coordination insertion mechanism R


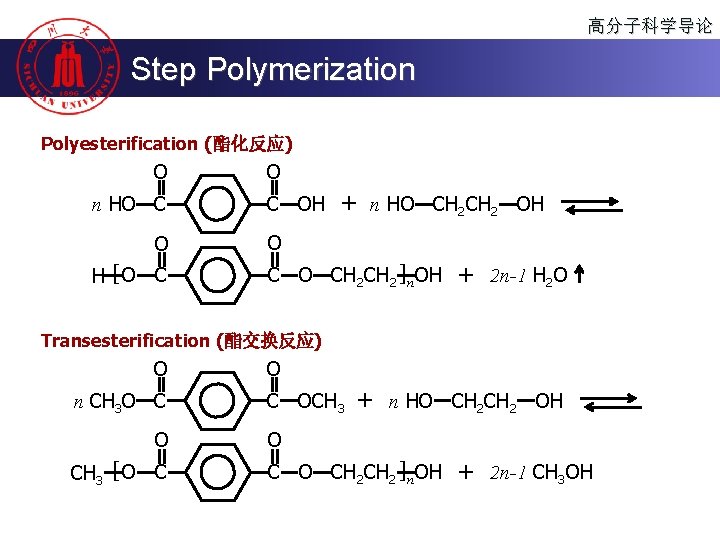
高分子科学导论 Step Polymerization Polyesterification (酯化反应) O n HO C O H [O C OH + n HO CH 2 OH O CH 2 CH 2 ]n OH + 2 n-1 H 2 O Transesterification (酯交换反应) O n CH 3 O C O CH 3 [ O C OCH 3 + n HO CH 2 OH O CH 2 CH 2 ]n OH + 2 n-1 CH 3 OH

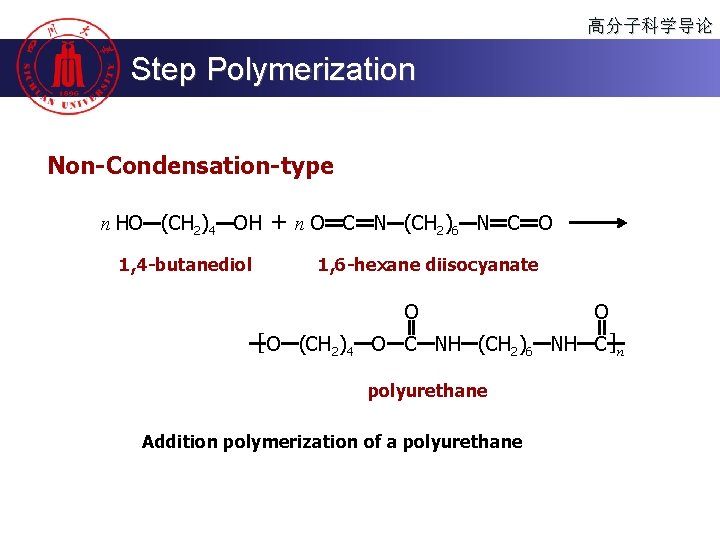
高分子科学导论 Step Polymerization Non-Condensation-type n HO (CH 2)4 OH 1, 4 -butanediol + n O C N (CH 2)6 N C O 1, 6 -hexane diisocyanate O O [ O (CH 2)4 O C NH (CH 2)6 NH C ]n polyurethane Addition polymerization of a polyurethane



高分子科学导论 Polymerization Methods u 界面聚合 (Interface polymerization) u 固相聚合 (Solid polymerization) u 超临界流体聚合 (Polymerization in supercritical fluids) u 辐射聚合 (Radiation polymerization) u 等离子体聚合 (Plasma polymerization) u 微波聚合 (Microwave-assisted polymerization) u 超声波聚合 (Ultrasonic-assisted polymerization)

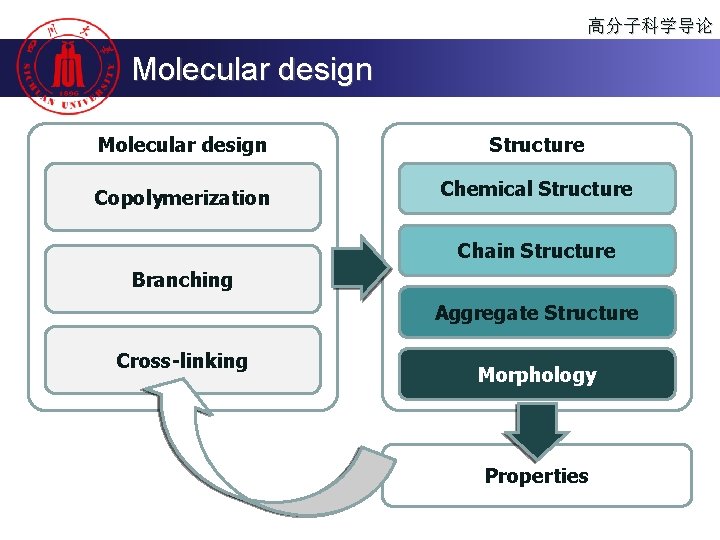
高分子科学导论 Molecular design Structure Copolymerization Chemical Structure Chain Structure Branching Aggregate Structure Cross-linking Morphology Properties

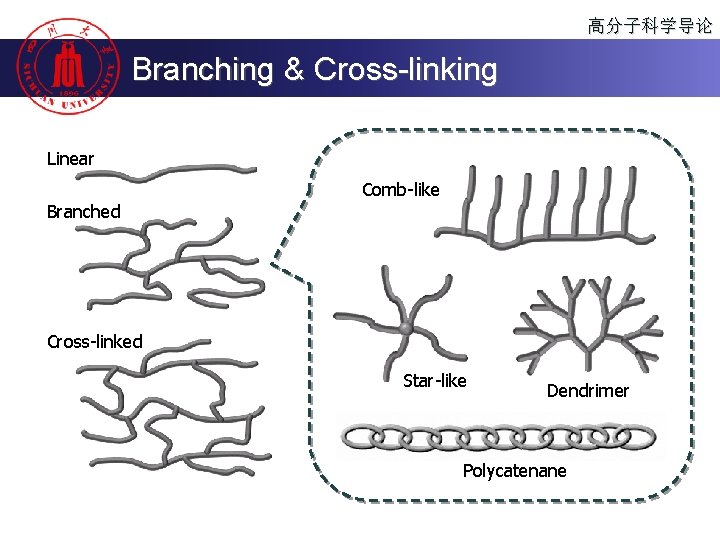
高分子科学导论 Branching & Cross-linking Linear Comb-like Branched Cross-linked Star-like Dendrimer Polycatenane

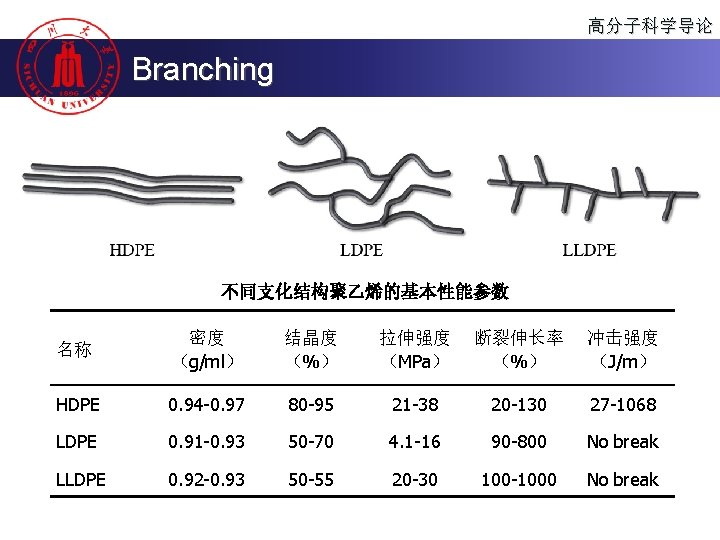
高分子科学导论 Branching 不同支化结构聚乙烯的基本性能参数 名称 密度 (g/ml) 结晶度 (%) 拉伸强度 (MPa) 断裂伸长率 (%) 冲击强度 (J/m) HDPE 0. 94 -0. 97 80 -95 21 -38 20 -130 27 -1068 LDPE 0. 91 -0. 93 50 -70 4. 1 -16 90 -800 No break LLDPE 0. 92 -0. 93 50 -55 20 -30 100 -1000 No break




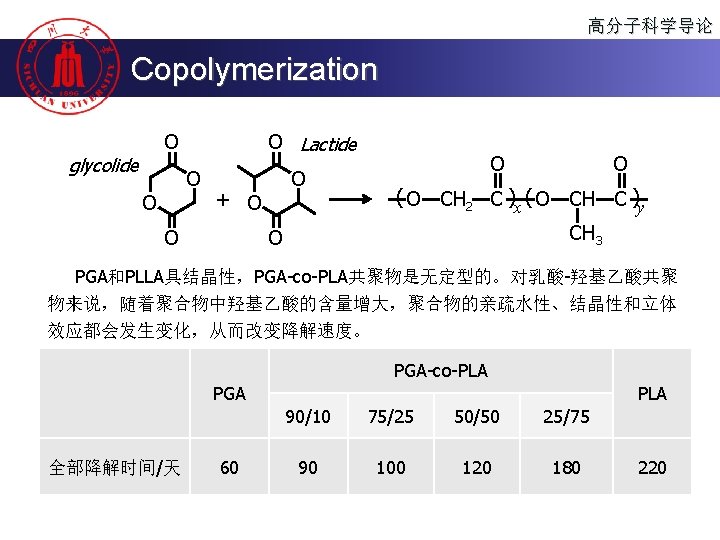
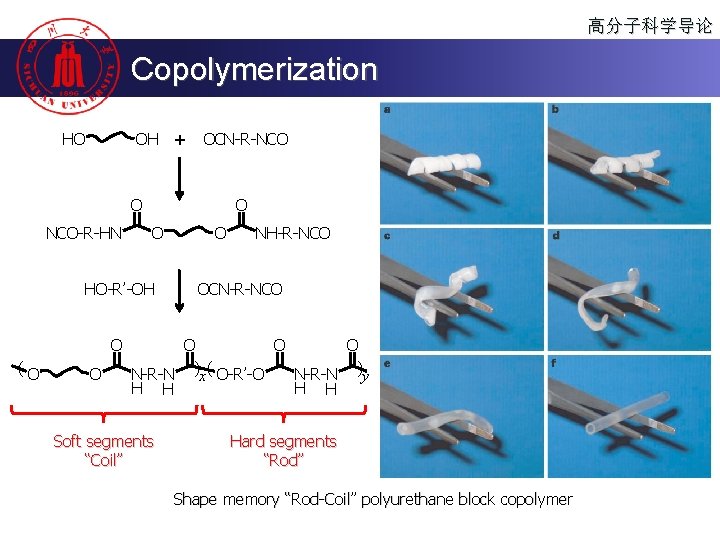
高分子科学导论 Copolymerization OH + HO OCN-R-NCO O NCO-R-HN O O HO-R’-OH O (O O O NH-R-NCO OCN-R-NCO O N-R-N )x ( O-R’-O N-R-N )y H H Soft segments “Coil” Hard segments “Rod” Shape memory “Rod-Coil” polyurethane block copolymer

高分子科学导论 Copolymerization Self-assembly “Crystalline-Coil” block copolymer of PPDO and PEG Si-Chong Chen, Gang Wu et. al. Chem. Commun. , 2011, 47, 4198– 4200

高分子科学导论 Copolymerization F. Y. Zhai, W. Huang, G. Wu, X. K. Jing, M. J. Wang, S. C. Chen* et. al. ACS Nano 2013, 7, 4892 -4901.

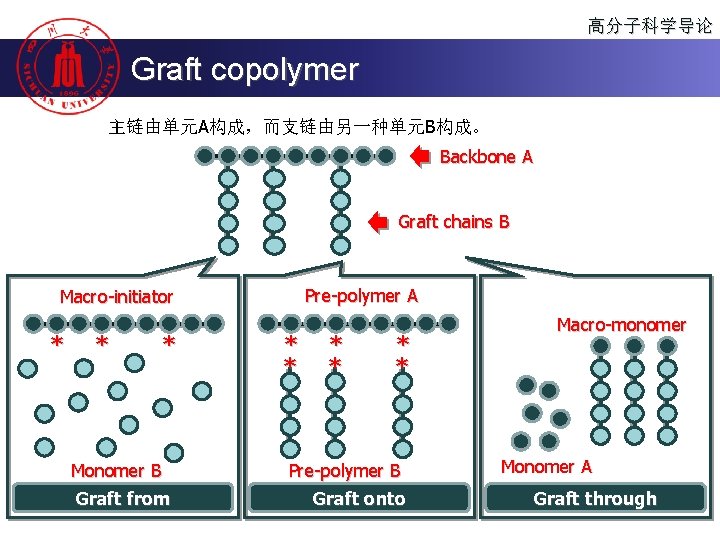
高分子科学导论 Graft copolymer 主链由单元A构成,而支链由另一种单元B构成。 Backbone A Graft chains B Pre-polymer A Macro-initiator * * * Monomer B Graft from * * * Pre-polymer B Graft onto Macro-monomer Monomer A Graft through





