Introduction to Physiology Acid Base Dr Barney Ward

Introduction to Physiology Acid & Base Dr. Barney Ward - Consultant Anaesthetist

Acid & Base • What are acids and bases ( for non-chemists) • Why is keeping acid / base balance important • How does the body manage this balance • How can we look at what is going on in the body?

What are acids? • Acids donate protons (H+ ions) Eg. Hydrochloric acid contains 1 hydrogen atom and 1 chlorine atom In solution HCl splits into ions (charged particles) HCl H+ + Cl. HCl is a strong acid, and almost all dissociates into H+ and Cl-

What are bases? • Bases can accept protons (H+ ions) Eg. sodium hydroxide (Na. OH) splits in solution to form Na+ and OHNa. OH Na+ + OHOH- ions can accept protons (H+) to form water OH- + H+ H 20

Strong vs. weak • In chemistry, we meet many strong acids & bases • In biology, most acids and bases are much weaker • Strong acids dissociate almost completely • Weaker acids do not… • CH 3 -COOH CH 3 COO- + H+ (cf. HCl H+ + Cl-)
![Measuring acidity • Concentration of H+ ions in solution - [H+] • Actual numbers Measuring acidity • Concentration of H+ ions in solution - [H+] • Actual numbers](http://slidetodoc.com/presentation_image/4e66c2140077f6e9dcbaba59a6d5c20f/image-6.jpg)
Measuring acidity • Concentration of H+ ions in solution - [H+] • Actual numbers not helpful! • Range 0. 1 - 0. 00000001 M • Taking negative base 10 logarithm gets rid of zeroes • Number known as p. H & gives range of 0 - 14
![Measuring acidity • For example - water has [H+] = 0. 0000001 M • Measuring acidity • For example - water has [H+] = 0. 0000001 M •](http://slidetodoc.com/presentation_image/4e66c2140077f6e9dcbaba59a6d5c20f/image-7.jpg)
Measuring acidity • For example - water has [H+] = 0. 0000001 M • - log 10 (0. 0000001) = 7 (neutral p. H) • Acids have p. H values less than 7 • Bases have p. H values greater than 7

• Acids have p. H values less than 7 • Bases have p. H values greater than 7

Why is maintaining p. H important? • Changes in acidity profoundly affect proteins • Protein function relies entirely on their shape • If p. H changes, so does the shape of proteins • Narrow limits of normal p. H 7. 35 - 7. 45 • Remember a 1 unit p. H change = 10 fold [H+] change

What disturbs the body’s p. H? • CO 2 production (respiration) – CO 2 is a weak acid when in solution – Produced when glucose metabolysed via glycolysis – Occurs in all cells, all the time

What disturbs the body’s p. H? • Metabolic acids – Under strenuous exercise, anaerobic metabolism occurs – Lactic acid produced – Also commonly accumulates in shock (poor perfusion state) – Protein breakdown produces acids

How does the body prevent p. H changes? • Buffers – Buffers allow acid to be ‘stored’ temporarily – Bicarbonate / CO 2 is the most important: H+ + HCO 3 - CO 2 + H 2 O – Excess acid (H+) drives the reaction to the right – CO 2 produced can be excreted by the lungs

How does the body prevent p. H changes? • Buffers – Other buffer systems: – Proteins - can reversibly accept H+ ions – Intracellular phosphate - HPO 42 - + H+ H 2 PO 4 -

How does the body prevent p. H changes? • Short term buffering requires a final solution • Respiratory acid (CO 2) is excreted by the lungs • Metabolic acids are excreted by the kidneys

Acid & Base in practice • Within the body, excess acid produces an acidosis • Excess base produces an alkalosis • Acidosis & alkalosis can be produced from either a respiratory or metabolic source

Acid & Base in practice • CO 2 too high = respiratory acidosis • CO 2 too low = respiratory alkalosis • Metabolic acids too high = metabolic acidosis • Metabolic acids too low = metabolic alkalosis

Acid & Base in practice - Blood gases • Arterial blood gas analysis reveals oxygenation & acid base status of the body • The analysis usually includes: p. H Pa. O 2 Pa. CO 2 BE Pa = Partial arterial pressure of O 2 or CO 2 BE = Base Excess (-ve values = acidosis, +ve values =

Acid & Base in practice - Blood gases Some normal values – – p. H Pa. O 2 Pa. CO 2 BE +ve=alkalosis 7. 35 – 7. 45 (no units) 10 – 13 KPa 4 – 4. 5 KPa -2 - +2 mmol/l -ve = acidosis,

Blood gases 1 - Oxygenation • To interpret Pa. O 2 result, you need to know what oxygen concentration the patient is breathing – – Pa. O 2 p. H Pa. CO 2 BE 9 (10 – 13 KPa) (7. 35 – 7. 45) (4 – 4. 5 KPa) (-2 - +2 mmol/l) • %age O 2 is often given as Fractional inspired O 2 • eg. 21% O 2 = Fi. O 2 0. 21

Blood gases 2 - Acid & Base This patient has severe sepsis – – Pa. O 2 p. H Pa. CO 2 BE 10 7. 0 4. 2 -19 (10 – 13 KPa) (7. 35 – 7. 45) low p. H= acid (4 – 4. 5 KPa) high CO 2= acid (-2 - +2 mmol/l) -ve = acid • First look at p. H - it tells you what the overall problem is • Next look at the Pa. CO 2 - it tells you what the respiratory side of things is doing • Then look at the base excess (BE) which tells you about the metabolic component • Here there is a metabolic acidosis (eg. severe sepsis)
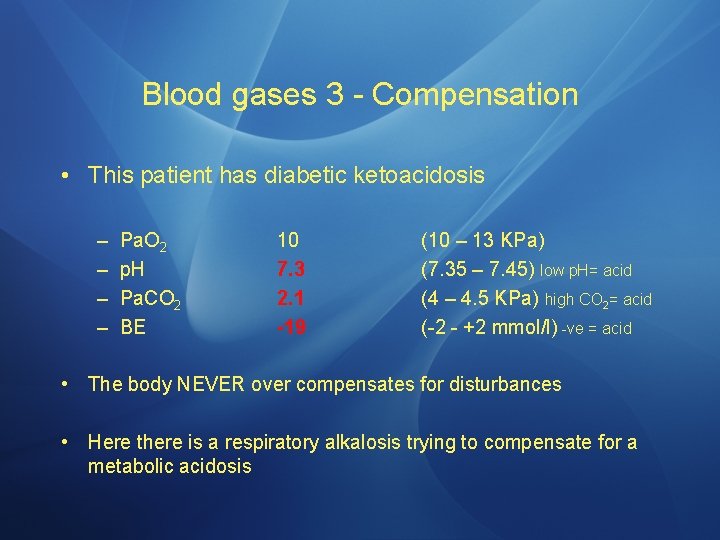
Blood gases 3 - Compensation • This patient has diabetic ketoacidosis – – Pa. O 2 p. H Pa. CO 2 BE 10 7. 3 2. 1 -19 (10 – 13 KPa) (7. 35 – 7. 45) low p. H= acid (4 – 4. 5 KPa) high CO 2= acid (-2 - +2 mmol/l) -ve = acid • The body NEVER over compensates for disturbances • Here there is a respiratory alkalosis trying to compensate for a metabolic acidosis
Blood gases 4 - Compensation • The lungs can compensate for disturbances QUICKLY • The kidneys can only compensate for metabolic disturbances SLOWLY BUT… • When things go wrong in the body, a metabolic acidosis can build up FAST

Acid / Base • Don’t worry if this is clear as mud! • More blood gas work in group session
- Slides: 23