Introduction to Pharmacy Ethics Background Pharmaceutical care involves

Introduction to Pharmacy Ethics

Background � Pharmaceutical care involves applied ethics in that it consists of the practical application of moral standards to specific ends. � Pharmaceutical care practitioners should not only be clinically competent, but must also adhere to the law and ethical standards. � The pharmaceutical care practitioner must learn to recognize when his/her personal values (political views, religious beliefs, or social expectations) interfere with professional responsibilities and behaviors. � As a general rule, clinical problems should be identified and resolved first, followed by legal issues.

Background � Pharmacists are the researchers, developers, producers, people who are trusted to give advice on drugs to all health professionals and persons who market drugs in the whole world. � The pharmaceutical industry is the most heavily regulated of all industries. � The law, as well as the professional regulation body and the public, will require reasons for professional actions. � Law and ethics are not static. This means that students, and eventual practitioners, need to remain alert throughout their professional life for changes and amendments to each and to be aware of how these will affect and impact on their chosen area of practice.
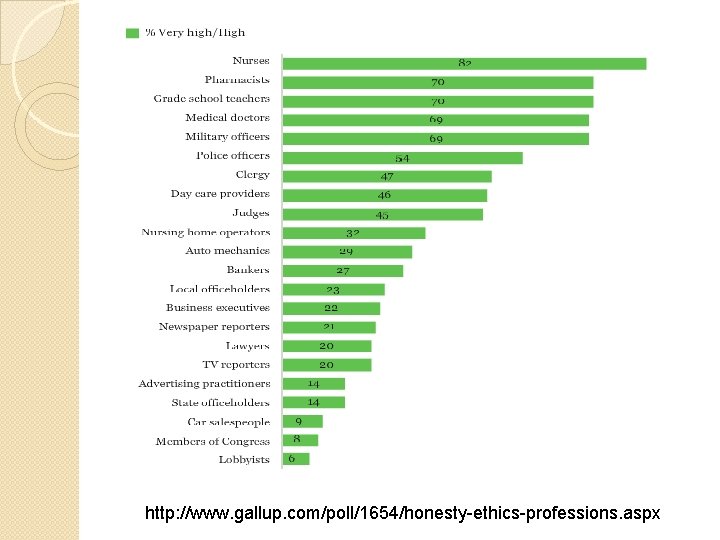
http: //www. gallup. com/poll/1654/honesty-ethics-professions. aspx

Ethics � Ethics is the study of the rightness or wrongness of human conduct. � Patient care involves applied ethics in that it consists of the practical application of moral standards to specific ends. � In pharmaceutical care, utilization of pharmaceuticals should depend on using the essential knowledge that should always accompany such clinical intervention.

Ethics � Pharmaceutical care is dependent upon human interactions, that include patients, family members, pharmaceutical care practitioners, other clinicians, support personnel, managers, and administrators. � These individuals are likely to have different values, beliefs, and preferences. Whenever two people with different value systems interact, there is the potential for an ethical problem to develop. � Because ethical problems are common in practice it is important that practitioners know how to identify and resolve them when they arise.

Ethics � There are three issues— clinical, legal, and ethical —which can be so closely associated. It will be helpful if the three issues can be separated, when possible, because a successful resolution to each issue is arrived at slightly differently. � Each situation requires different knowledge for its recognition and a somewhat different process for its resolution. � As a general rule, clinical problems should be identified and resolved first, followed by legal issues, and if an ethical problem remains, it can then be resolved effectively.

History 1906 Federal Food and Drug Act Ø Enacted to stop the sale of inaccurately labeled drugs Ø Manufacturers were required to put truthful information on the label before selling the drug Ø Manufacturers had to prove their drug’s effectiveness 1914 Harrison Narcotic Act Ø Enacted because of the excessive number of opium addicts in the United States Ø People could no longer obtain opium without a prescription 1938 Food, Drug, and Cosmetic Act Ø Enacted because the earlier Food and Drug Act was not worded strictly enough and did not include cosmetics Ø Required drug companies to include directions to the consumer regarding use and package inserts on drugs Ø All narcotics were required to be labeled “Warning:

History 1951 Durham-Humphrey Amendment ØRequired the labeling ”Caution: Federal law prohibits dispensing without a prescription” ØMade certain drugs require a doctor’s order and supervision ØMade the initial distinction between legend drugs (by prescription only) and over-the-counter (OTC) medications 1962 Kefauver-Harris Amendment Ø Enacted in an attempt to ensure the safety and effectiveness of all new drugs on the market ØBurden put on manufacturing companies to have good manufacturing practices (GMP) ØPrevented the sale of thalidomide in the United States because children were born with birth defects after

Issues of the law � Pharmacy law consists of rules, regulations, and actions that are disseminated by governments and are binding on its constituents. Example Should a pharmacist, based on his/her clinical judgment at the time of an emergency, provide a patient with a life-saving drug not authorized by a physician's prescription, when technically he/she is breaking the law, but is perhaps acting ethically? � There are many laws that direct what practitioners may and may not do with prescription drug products. � In those relatively few situations where the law is not clear, practitioners should always follow the professional and ethical mandate to do no harm and when it is possible, to do good for the patient.

Issues of the law � Example Is it ethical to intentionally dose a patient with an amount of drug that is so small to has little or no chance of successfully treating the medical problem? If the pharmacist's clinical judgment is that the patient's drug therapy problem is that the dosage is too low, then it is legally necessary to obtain the consent of the prescriber in order to increase the dose. � Being an active moral agent can conflict with the law and often does. � Ethical analysis requires critical examination of actions and their consequences and are not so easily differentiated as appropriate and inappropriate.

Legislation � Legislation sets out the rules or structure for what we can do and what we are not allowed to do and outlines the consequences of breaking the rules. � Every pharmacist has a duty to practise lawfully and the sanctions for failing to do so can be harsh. � The consequences of breaking the rules of professional practice can include criminal prosecution resulting in a fine or, in more serious cases, imprisonment, as well as removal of the right to continue to practise (removal from the register).

Pharmacist and Legislation � Pharmacists, are required to follow the requirements of the Code of Ethics and other guidance and standards set out by the professional regulator. � When applying clinical knowledge in practice, patients and the public will rightly wish to assume that the knowledge will be up to date, relevant and safe. � Early legislation relating to pharmacy was concerned with prohibiting harmful practices associated with the supply of medicines and restricting authority to a few types of practitioner. � In UK, Pharmacists were all required to achieve a set standard of education, to register with the Royal Pharmaceutical Society of Great Britain and pay an annual retention fee in order to be able to continue to practise.

Pharmacist and Legislation � Other recent changes have allowed pharmacists to utilise their clinical skills to become authorised prescribers as well as dispensers or suppliers of medicines. �A knowledge and understanding of pharmacy legislation are therefore crucial to be able to practise. � Pharmacy legislation sets out the framework of rules that govern what we can do legally.

Pharmacist and Legislation Accountable (responsible ) officer � In Health Act 2006 which relates to the supervision, management and use of controlled drugs. It introduces the concept of the ‘accountable officer’ � The aim was preventing patient deaths, take a systematic approach to the early detection of practitioners who might be misusing or abusing controlled drugs. � The Health Act places a requirement on specified organisations within the NHS to appoint an Accountable Officer for controlled drugs. � The role of this person is to ensure the safe use of controlled drugs within his or her organisation. � It also involves training, monitoring and assessment of people involved in their management and use.

Medicines and Pharmacies � The Health Act 2006 provides for powers in relation to supervision of sales of medicines in registered pharmacy site. � Replacing the term ‘personal control’ with ‘responsible pharmacist’, referring to both registered pharmacy premises and bodies corporate. � Since the implementation of the Medicines Act 1968, every registered pharmacy premises has been required to have a pharmacist in ‘personal control’. ‘Personal control’ is the legal term that has been replaced by the new term ‘responsible pharmacist’

Medicines and Pharmacies � The term ‘personal control’ meant that there had to be a pharmacist within each pharmacy who was responsible for the pharmacy’s systems and practices to ensure safe sale and supply of medicines to patients and the public. � Why is this control important? It meant that in every registered pharmacy premises, even those pharmacy building that were part of a body corporate and therefore had a manager pharmacist, there was an individual pharmacist responsible for the immediate running of each individual pharmacy. � The pharmacist had to be personally available to intervene in every sale or supply of a medicine whether in accordance with a prescription.

Medicines and Pharmacies � This requirement for a pharmacist to be present in the pharmacy at all times, put pharmacists at variance from other professions where supervision meant overseeing systems and/or practices of groups of professionals rather than personal presence and involvement. � For example, a supervisor of midwives is a senior midwife with professional responsibility. However, a supervisor of midwives does not personally have to attend every birth or every clinic visit undertaken by every member of his or her team of midwives.

Medicines and Pharmacies � In the early 20 th century, the pharmacy was the place many people went for advice about their healthcare. These were the places where free advice was readily available from a healthcare professional, but the National Insurance Act of 1911 changed health provision for many – mostly men. With the introduction of this Act of Parliament, employed men earning less than £ 170 per annum became entitled to free medical care provided by doctors who were ‘on the panel’. This meant the doctors were paid a retainer fee to see and treat men covered by the Act.

Conclusion � The art of pharmacy was first practiced in Ancient Babylon around 2600 BC. In this era the priest, physician and pharmacist was the same person. The Arabs were the first to separate the art of pharmacy from physician and in the eight century they establish the first private pharmacy in Baghdad. � When the European countries were exposed to Arabian influence, public pharmacies began to appear. However, it was not until about 1240 A. D. that pharmacy was separated from medicine. � The role of the pharmacist is changing drastically with the traditional activities such as compounding of medicines, and pharmacist becoming more like a walking encyclopedia for drugs, fulfilling the doctor’s needs by giving advice and information on use of drugs, providing correct dosage forms, assuring the efficacy and quality of

Conclusion � Now the pharmacy profession is not only related to dispensing and distribution of drugs or sometimes being regarded as a “glorified store keeper”. � Pharmacists now have a bigger role as global players and in the adoption of global standards so as not to be left out or left behind in the global race.
- Slides: 21