Introduction to Patient Safety Torbjrn Turland European Product

Introduction to Patient Safety Torbjörn Turland European Product Manager BD Pha. Seal. TM Pha. Seal Training 3 rd – 5 th December 2014 Oxford, UK

Healthcare Worker Safety PATIENT SAFETY Prescribing Administration Dispensing Information QUALITY = SAFETY Manufacturing STAFF SAFETY Monitoring

First question…… Do you think bacteria are killed by chemotherapy drugs?

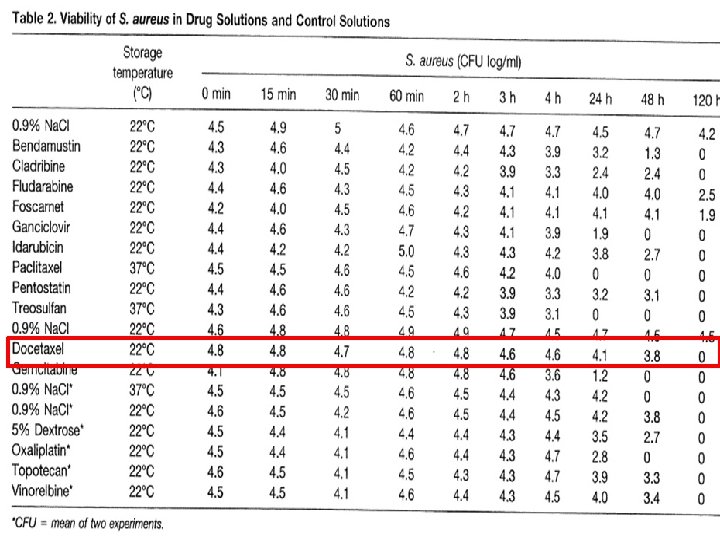
Why Quality? It’s microbiology for the patient! § Patients have an compromised immune system through: – Chemotherapy – Radiation § Much more need for sterile preparations than for e. g. TPN § Wrong perception that chemotherapy drugs would kill microorganism: – – Depends on organism Depends on product No short term activity Limited activity on long term

Microbiological challenge of four protective devices for the reconstitution of cytotoxic agents De Prijck, D’Haese, Vandenbroucke

Contamination of Vial Drop PSEUDOMONAS AEROGINOSA 4 X 103 (= Real Case) 4 X 105 (= Worst Case)
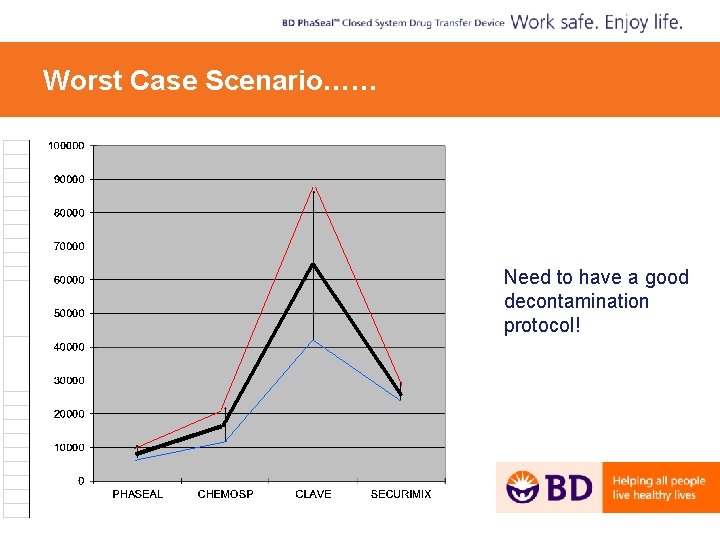
Worst Case Scenario…… § Need to have a good decontamination protocol!

Contamination of Transfer Device with Pseud Aer
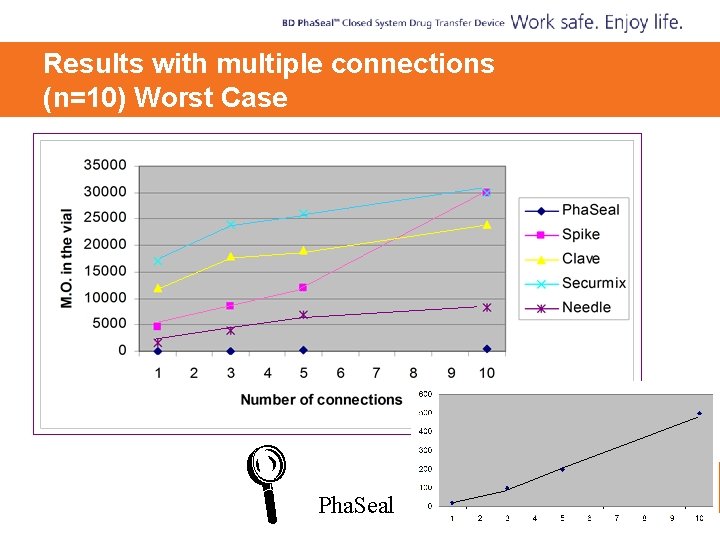
Results with multiple connections (n=10) Worst Case Pha. Seal

Study Conclusions § You can protect the patient by using a device which limits the amount of contamination going into the vial § Spraying or swabbing alone was inadequate, as opposed to a combination of spraying [0. 5% or 2. 0% (w ⁄ v) chlorhexidine in isopropanol] and swabbing [70% (v ⁄ v) isopropanol]. § Although Pha. Seal afforded the lowest transfer of micro-organisms, adequate disinfection of the vial prior to connection remains required.

First question…… Do you think bacteria are killed by chemotherapy drugs?
- Slides: 12