Introduction to Pathology Dr Ibrahim Bakhit Yousif Ph

Introduction to Pathology Dr. Ibrahim Bakhit Yousif Ph. D in Histopathology & Cytology
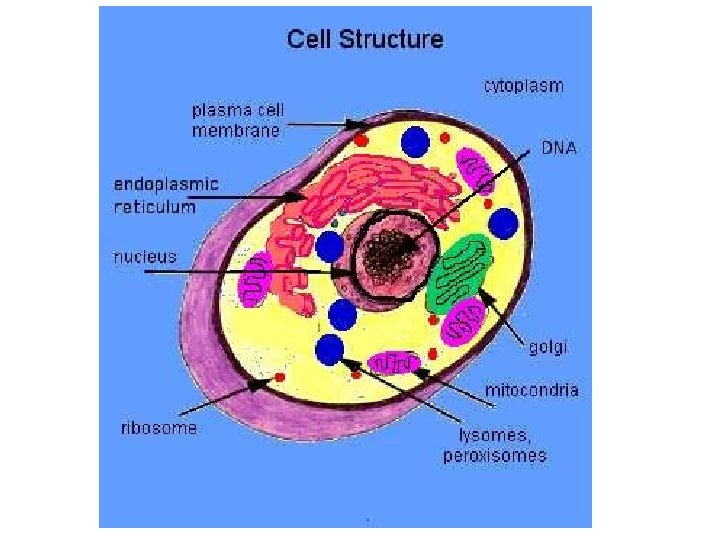

Learning Pathology: General Pathology Common changes in all tissues. e. g. . Inflammation, cancer, ageing, edema, hemorrhage …. etc. Systemic Pathology Discussing the pathologic mechanisms in relation to various organ systems e. g. CVS, CNS, GIT…. . etc.

What is the Disease? It is the “State in which an individual exhibits an anatomical, physiological, or biochemical deviation from the normal” Disease may be defined as : • an abnormal alteration of structure or function in any part of the body.

What is Pathology is the Scientific Study of Cell Injury and Disease: The structural and/or functional changes in cells, tissues, and organs. Scientific Study: applying one or more scientific methods to reach a diagnosis.

What we study in Disease: 1. Etiology: some diseases have a single etiology others have multiple etiological factors. Can be intrinsic or extrinsic causes. 2. Pathogenesis: sequence of events from the initial stimulus to ultimate expression of the disease (mechanisms). 3. Morphology: structural changes in cells and tissues (grossly and microscopically). 4. Clinical significance: course (signs and symptoms) and prognosis (outcome).

Four aspects of disease process Aetiology Pathogenesis Morphological changes Clinical significance

Aetiology (Causes of Injury and Disease) 1. Hypoxia (inadequate oxygenation of cells and tissues). 2. Physical agents (trauma, radiation, heat. . ). 3. Chemical agents (poisons, air pollutants, alcohol, cigarette smoking. . ). 4. Infectious agents (viruses, bacteria. . ). 5. Immunological reactions. e. g. drugs. 6. Nutritional imbalance (vitamin C deficiency and obesity). 7. Genetic factors : e. g; sickle cell anemia.

Pathogenesis Is the mechanism by which a certain aetiological factor causes disease (In Greek: pathos = disease, genesis = development).

Clinical Significance of Disease Ø Course: progression of disease. 1. Symptoms: complaints of the patient. 2. Signs: features of disease detected by clinical examination or investigations. 3. Prognosis: outcome of disease (good, poor, or unknown).

Clinical Symptoms & Signs Clinical symptoms are the patient’s complain l usually by its own words as pain. Clinical signs are seen only in the living individual as organomegaly.
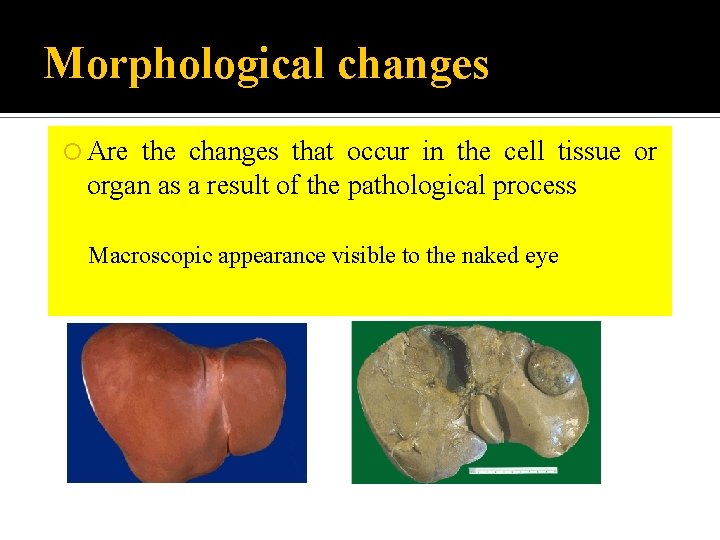
Morphological changes Are the changes that occur in the cell tissue or organ as a result of the pathological process Macroscopic appearance visible to the naked eye

Progression of a disease Complete cure Death Complication

Cell reacts to adverse influences by : All forms of cell injury start with molecular or structural alterations in cells. Under normal conditions, cells are in a homeostatic ‟steady state. ” Excess physiologic or pathologic stress may force the cell to a new steady state (Adaptation). Too much stress exceeds the cell’s adaptive capacity (Injury).

1) 2) 3) Adapting. Reversible injury. Irreversible injury and dying. Cellular adaptation : occurs when excessive physiologic stresses, or some pathologic stimuli, preserves the viability of the cell. Examples are: 1. Hypertrophy ( increase in mass of the cell). 2. Atrophy ( decrease in mass of the cell). Revesible cell injury: pathologic changes that can be reversed when the stimulus is removed.

Irreversible injury: pathologic changes that are permanent and cause cell death. There are two morphologic patterns of cell death: Necrosis versus apoptosis 1. necrosis. 2. Apoptosis. Necrosis: is more common type after exogenous stimuli and is manifested by: a. Cell swelling. b. Denaturation. c. Coagulation of proteins. d. Breakdown of cellular organelles. e. Cell rupture.

Apoptosis: ( programmed cell death ) is characterized by: 1) Occurs in single or small clusters of cells. 2) Results in elimination of unwanted cells during embryogenesis and in various physiologic and pathologic states.

Causes of cellular injury: 1. Hypoxia (decrease of oxygen), due to : a. Ischaemia (loss of blood supply). b. Inadequate oxygenation e. g. , (cardiorespiratory failure). c. Loss of oxygen- carrying capacity of the blood e. g (anaemia, carbon monoxide poisoning).

2. Physical agent, including : v Trauma. v Heat. v Cold. v Radiation. v Electric shock.
![3. Chemical agents and drugs: a) Therapeutic drugs (e. g. , acetaminophen[Tylenol]). b) Nontherapeutic 3. Chemical agents and drugs: a) Therapeutic drugs (e. g. , acetaminophen[Tylenol]). b) Nontherapeutic](http://slidetodoc.com/presentation_image_h2/4ee102aaf07b7e0854c1182f4b0e3d64/image-20.jpg)
3. Chemical agents and drugs: a) Therapeutic drugs (e. g. , acetaminophen[Tylenol]). b) Nontherapeutic agents (lead & alcohol). 4. Infections agents, including viruses, rickettsiae, bacteria, fungi and parasites. 5. Immunologic reactions. 6. Genetic derangements 7. Nutritional imbalances.

There are 4 main structures that may be targeted by injury: 1. Cell membrane 2. Mitochondria (ATP production) 3. Protein 4. DNA (genetic material)

Biochemical Mechanisms of Cell Injury ATP depletion. Intracellular accumulation of calcium. Accumulation of Oxygen-derived free radicals (Oxidative Stress). Mitochondrial Membrane Damage. damage with permeability loss.

1 -ATP depletion Hypoxia causes decreased ATP synthesis. This depletion results in: 1. Failure of active transport by cell membrane and accumulation of sodium and water into the cell. 2. Defects in protein synthesis, lipogenesis, and phospholipids turnover, leading to loss of integrity of plasma membrane.

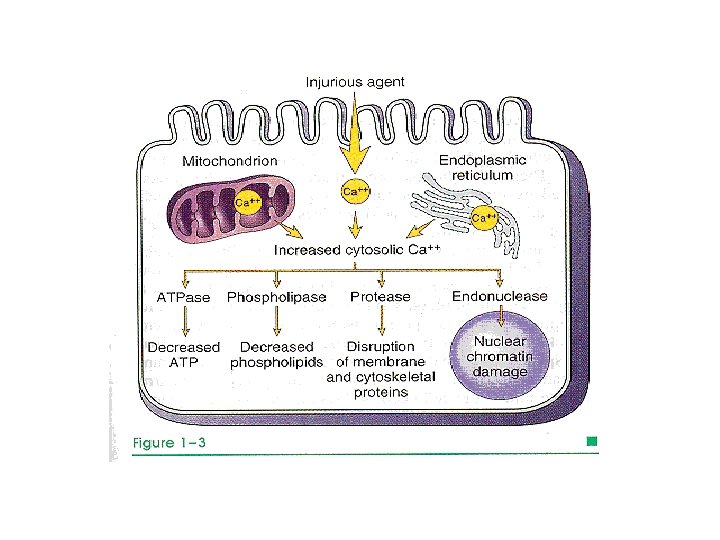
2 -Intracellular accumulation of calcium. Ischemia (deficient blood supply) increases concentrations of cytosolic calcium leading to activation of catabolic enzymes (phospholipase, protease, ATPase, and endonuclease). This results in cell membrane damage, breakdown of proteins, ATP depletion, and chromatin damage.

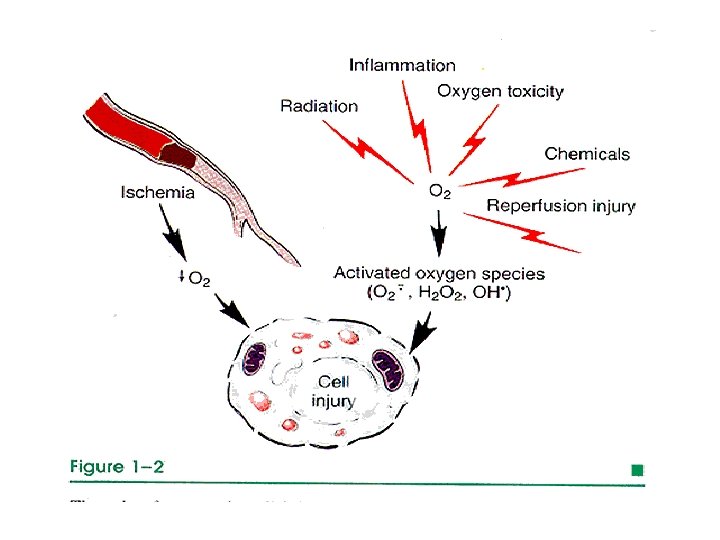
3 -Accumulation of Oxygen-Derived Free Radicals (Oxidative Stress) During generation of energy by cells, small amounts of reactive oxygen forms and free radicals are produced. These forms are can damage lipids, proteins, and nucleic acids. They are referred to as reactive oxygen species. Cells have defense systems to prevent injury caused by these products. An imbalance between free radical-generating and radical scavenging systems results in oxidative stress, leading to cell injury.

Important pathologic sources of free radicals include: 1. Prolonged 2. Bacterial ischemia (venous obstruction). infection 3. Radiation

Oxygen and oxygen derived free radicals affect cells as follows: 1. Lipid per oxidation of cell membrane, leading to cell damage. 2. Oxidative proteins, leading to damage of proteins and enzymes. 3. DNA damage, leading to death or cancer

Free radicals- induced cell injury: Free radicals are highly reactive, unstable species that interact with proteins, lipids, and carbohydrates and are Involved in cell injury induced by: a) Absorption of radiant energy (UV light, X-ray). b) Oxidative metabolic reactions. c) Enzymatic conversion of oxygenous chemicals & drugs (ccl 4→ccl 3). d) O-derived radicals are toxic species (e. g. OH, H 2 O 2, NO). e) Superoxide (Ȯ 2).

4 -Mitochondrial Damage Mitochondria can be damaged by: 1. Increased cytosolic calcium. 2. Oxidative stress. 3. Breakdown of phospholipids.

5 -Membrane damage with loss of Permeability This can be due to: 1. ATP depletion 2. Loss of phospholipids due to activation of phospholipases by increased calcium. 3. Toxicity of oxygen derived free radicals. 4. Various infectious, chemical, or physical factors.

Reversible injury Definition: cells or tissues return to normal structure and function after removal of the injurious factor. Morphology: 1. Hydropic change: accumulation of water and sodium into the cell. 2. Fatty change: accumulation of fat into the cell.

Hydropic Change Hypoxia results in ATP reduction, which affects the membrane Na- K pump, leading to accumulation of water and sodium into the cell and cellular swelling. Affects all types of cells and considered the first sign of cell injury. .

Fatty Change Hypoxia leads to release of fat from cell membrane, which appear as small or large lipid vacuoles in cytoplasm.

Irreversible cell injury Definition: cells and tissues affected do not return to normal structure and function. Morphology: 1. Severe cytoplasmic vacuolation. 2. Mitochondrial damage. 3. Severe damage of cell components and membrane.

Necrosis Definition: morphologic changes that follow cell death in living tissue. The morphologic appearance of necrosis is the result of denaturation of intracellular proteins and enzymatic digestion of the cell. These enzymes are derived either from the lysosomes of the dead cells themselves (autolysis), or from the lysosomes (heterolysis). leukocytes during inflammatory reactions

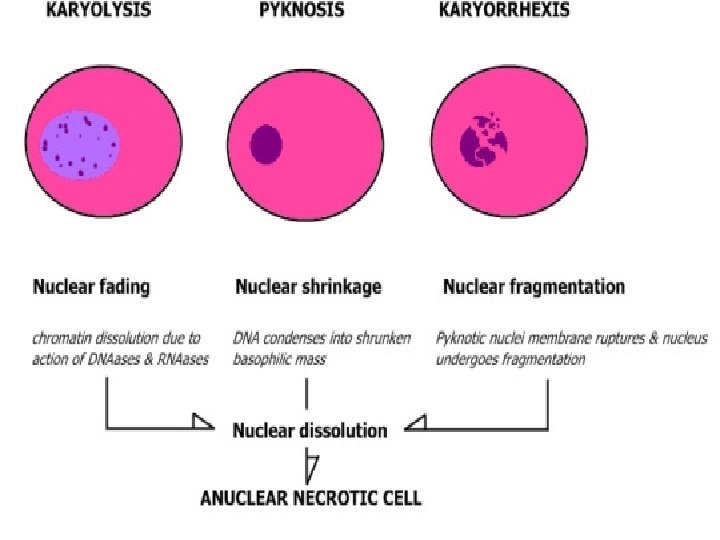
Nuclear changes assume one of three patterns: 1. karyolysis : fading of chromatin basophilia. 2. pyknosis: characterized by nuclear shrinkage and increased basophilia. 3. karyorrhexis: the nucleus undergoes fragmentation.
DEATH: LIGHT MICROSCOPY

Common types of necrosis: 1. Coagulative necrosis: common in the heart, kidney, and spleen. The affected tissue is swollen, firm and yellow with visible outlines of cells. 2. Liquefactive necrosis: results from rapid action of hydrolytic enzymes , common in the brain and with bacterial infections. The affected tissue is soft liquidlike with complete loss of architecture.
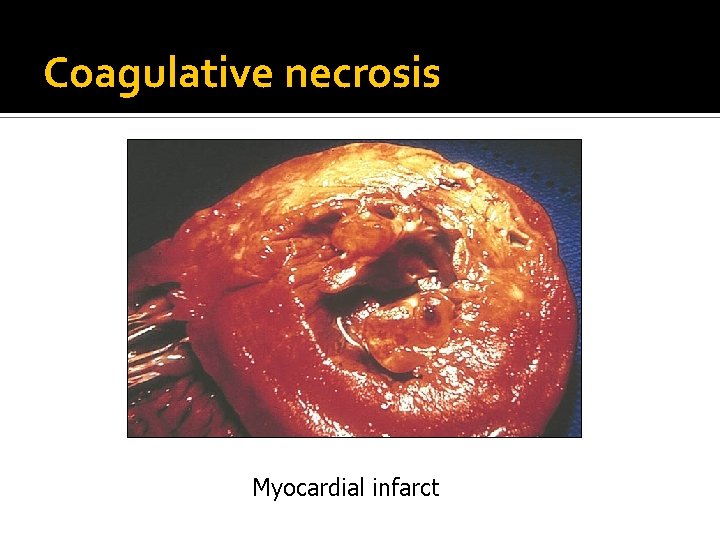
Coagulative necrosis Myocardial infarct

3. Caseous necrosis: chronic type of coagulative necrosis with fragile cheesy appearance. It is characteristic for TB. 4. Gangrenous necrosis: it is a combination between coagulative and liquefactive necrosis. Common in the legs. 5. Fat necrosis: due to action of activated lipases common in fat of breast (traumatic) and peritoneum (alcoholic).
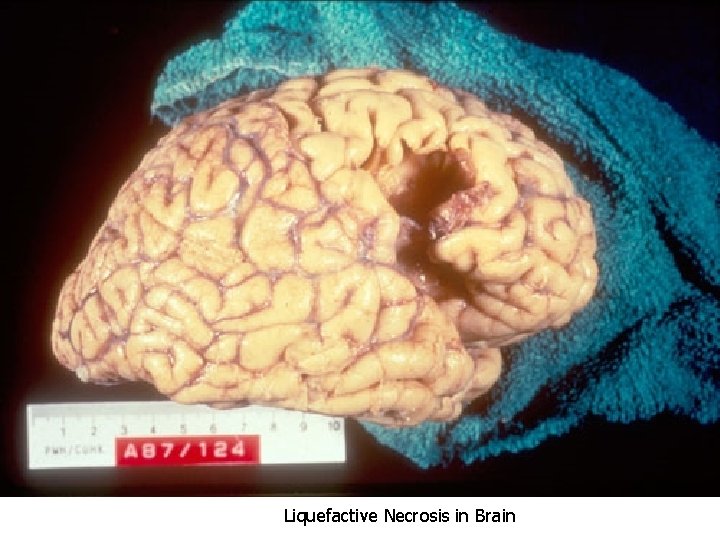
Liquefactive Necrosis in Brain
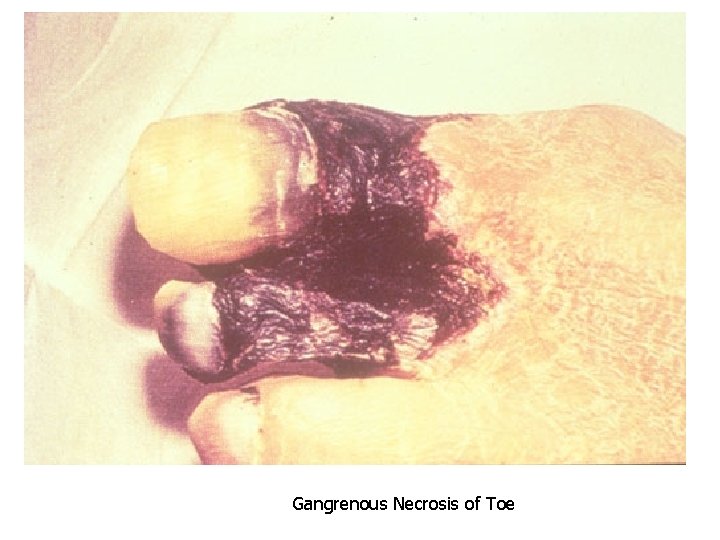
Gangrenous Necrosis of Toe
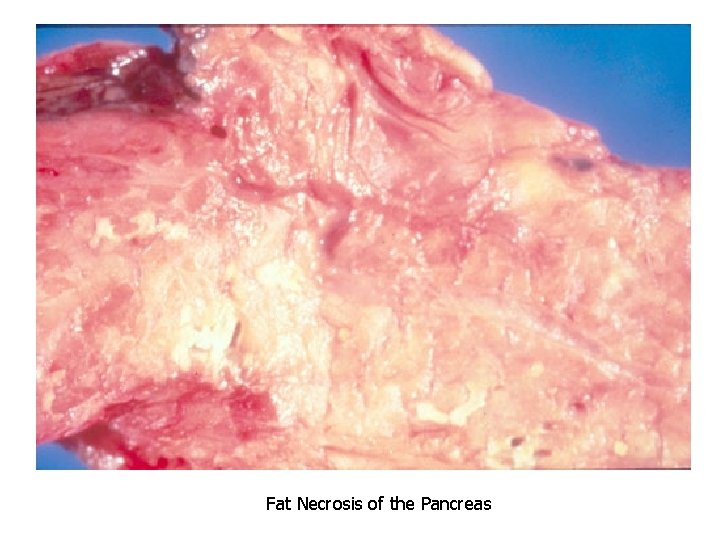
Fat Necrosis of the Pancreas

Fat Necrosis of the Pancreas

Apoptosis Is programmed cell death Can affect single cell or small group of cells Is not associated with inflammatory reaction Could be pathological or physiological like in thymus involution causes Physiological, embryogensis, hormon dependent, Pathological, injurious stimuli, viral infections, tumors

Morphological features Cell shrinking Chromatin Cellular Lack condensation& fragmentation destruction into apoptotic bodies of inflammation
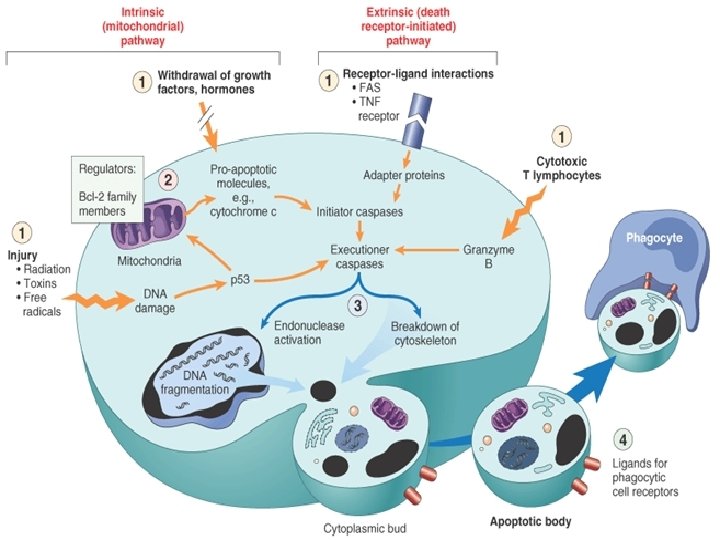

Removal of the dead cell Dying cells that undergo the final stages of apoptosis display phagocytotic molecules, such as phosphatidylserine on their cell surface. The process is so efficient as it disappear without leaving a trace of inflammation
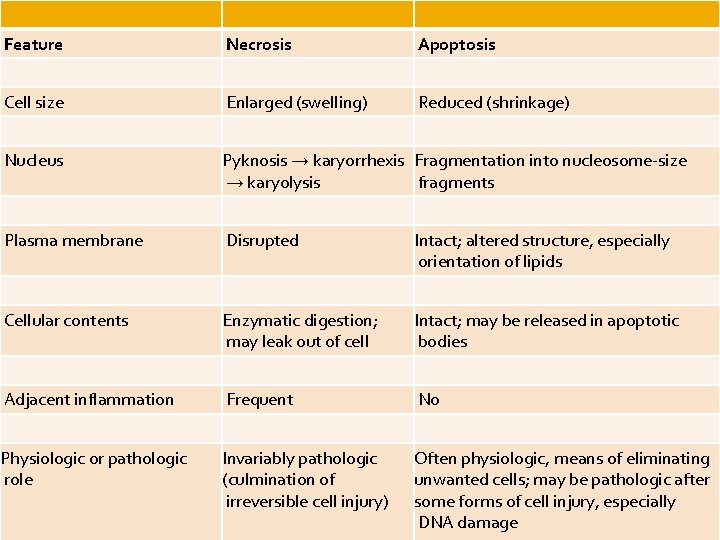
Feature Necrosis Apoptosis Cell size Enlarged (swelling) Reduced (shrinkage) Nucleus Pyknosis → karyorrhexis Fragmentation into nucleosome-size → karyolysis fragments Plasma membrane Disrupted Intact; altered structure, especially orientation of lipids Cellular contents Enzymatic digestion; may leak out of cell Intact; may be released in apoptotic bodies Adjacent inflammation Frequent No Physiologic or pathologic role Invariably pathologic (culmination of irreversible cell injury) Often physiologic, means of eliminating unwanted cells; may be pathologic after some forms of cell injury, especially DNA damage

Morphological Cellular Adaptations

Atrophy. Hypoplasia. Hypertrophy. Metaplasia.

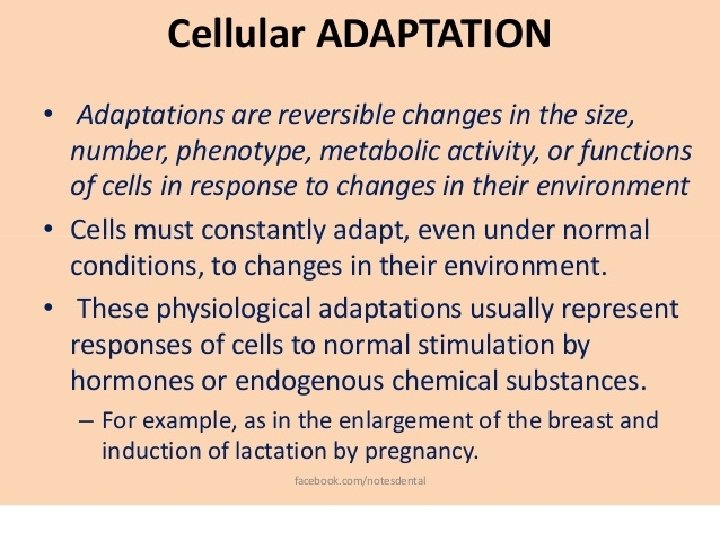
Atrophy Definition: decreased cell size and functions due to reduced number of cell organelles. Examples: 1. Brain atrophy due to aging. 2. Pressure atrophy due to mass lesions. 3. Nutritional atrophy: starvation. 4. Atrophy due to febrile disease.
Testicular Atrophy

Hypoplasia Definition: decreased cell numbers due to failure of proliferation during development 1. Examples: Dwarfism( hypo plastic tissues due to reduced levels of growth hormone or lack of its receptors).

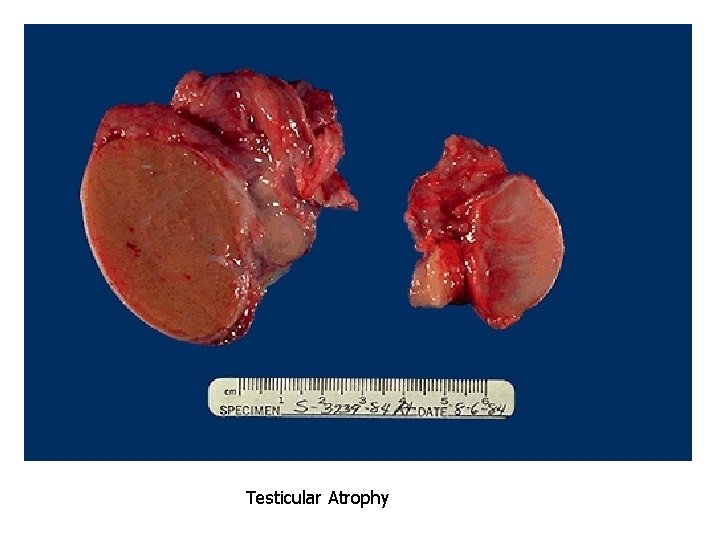
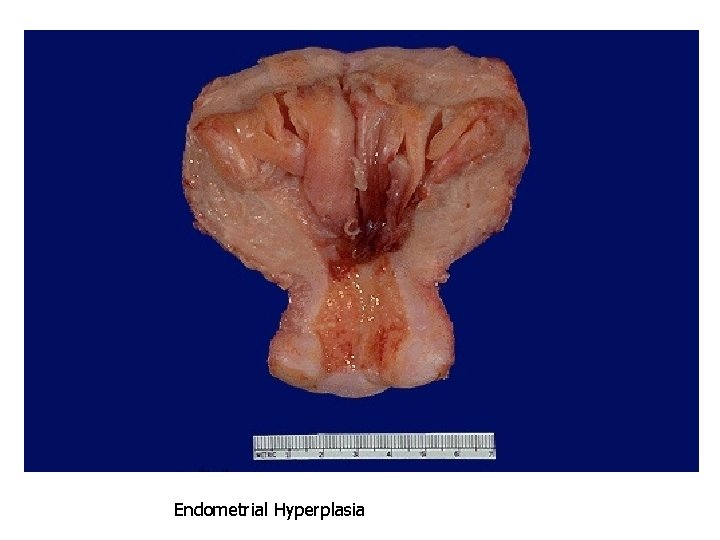
Hyperplasia Definition: increased total mass of an organ due to increased number of cells by frequent cell divisions. Examples: 1. Enlargement of breasts during pregnancy due to hormonal effects (physiological). 2. Endometrial hyperplasia (pathological). 3. Bone marrow hyperplasia (compensatory).
Endometrial Hyperplasia
Hyperplasia of Prostate

Hypertrophy Definition: increased total mass of an organ due to increased size of cells without cell division. Mainly occurs in organs composed of cells that can’t divide (cardiac muscles) Examples: 1. Increased functional demand hormonal stimuli result in enlargement of uterine fibers during pregnancy. 2. Compensatory hypertrophy in renal donors.

Hyperplasia and hypertrophy frequently occur together. For example , hormone-induced growth in uterus during pregnancy involves both increased number and size of smooth muscle and epithelial cells.

Metaplasia Definition: transformation of one type of differentiated tissue into another. Examples: 1. Cigarette smoking makes bronchial epith becomes squamous instead of columnar.
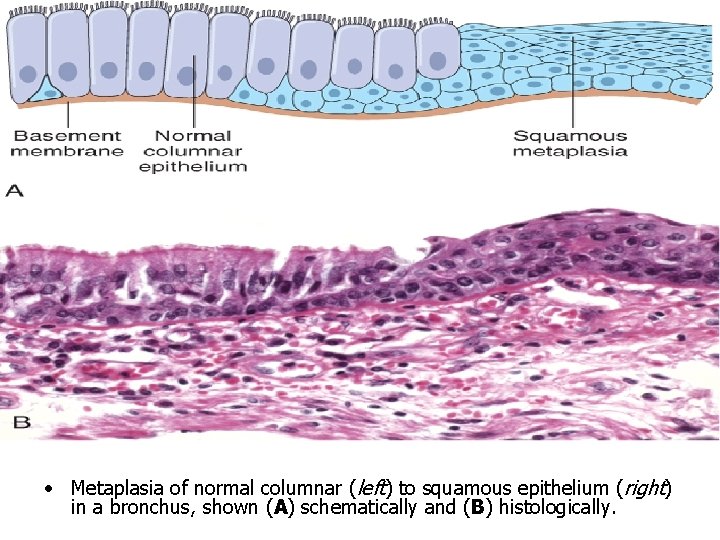
• Metaplasia of normal columnar (left) to squamous epithelium (right) in a bronchus, shown (A) schematically and (B) histologically.
- Slides: 65