INTRODUCTION TO PARASITOLOGY DR MOHIT BHATIA ASSISTANT PROFESSOR

INTRODUCTION TO PARASITOLOGY DR. MOHIT BHATIA ASSISTANT PROFESSOR DEPARTMENT OF MICROBIOLOGY AIIMS, RISHIKESH

KEY DEFINITIONS • Medical parasitology: “the study and medical implications of parasites that infect humans” • Parasite: “a living organism that acquires some of its basic nutritional requirements through its intimate contact with another living organism”. Parasites may be simple unicellular protozoa or complex multicellular metazoan • Eukaryote: a cell with a well-defined chromosome in a membrane-bound nucleus. All parasitic organisms are eukaryotes • Protozoa: unicellular organisms, e. g. Plasmodium spp. (Malaria) • Metazoa: multicellular organisms, e. g. helminths (worms) and arthropods (ticks, lice) • An endoparasite: “a parasite that lives within another living organism” – e. g. Plasmodium spp. , Giardia • An ectoparasite: “a parasite that lives on the external surface of another living organism” – e. g. lice, ticks

• Host: “the organism in, or on, which the parasite lives and causes harm” • Definitive host: “the organism in which the adult or sexually mature stage of the parasite lives” • Intermediate host: “the organism in which the parasite lives during a period of its development only” • Zoonosis: “a parasitic disease in which an animal is normally the host - but which also infects man” • Vector: “a living carrier (e. g. an arthropod) that transports a pathogenic organism from an infected to a non-infected host”. A typical example is the female Anopheles mosquito that transmits malaria

WHAT ARE PARASITISM, PARASITE AND HOST? Evolution of parasitism: Understanding start with basic concept of symbiosis • Symbiosis was first coined by the German de Bary in 1879 - to mean “living together”. It was originally coined to refer to all cases where dissimilar organisms or species (e. g. , heterogenetic associations) live together in an intimate association

INTERACTIONS OF SYMBIONTS In order to facilitate our understanding of symbiosis, 3 subordinate categories of symbiotic relationships are indicated. They are: commensalism (including phoresis), mutualism, and parasitism

SYMBIOSIS (CONT. ) 1. Mutualism: This occurs when each member of the association benefits the other • The mutuals are metabolically dependent on one another. Sometimes, one cannot survive in the absence of the other

SYMBIOSIS (CONT. ) 2. Commensalism means “eating at the same • table” and in many commensalistic relationships one organism (the commensal) is feeding on food that was not consumed by the host Commensalism occurs when one member of the associating pair, usually the smaller, receives all the benefit and the other member is neither benefited nor harmed • To carry -- phoresis

SYMBIOSIS (CONT. ) 3. Parasitism A parasitos (para: beside; sitos: grain or food): Original meaning from the Greek is a relationship in which "one eats at another's table or lives at another's expense. " • Parasitism is a relationship in which one of the participants, the parasite, either harms its host (the part that got harmed) or in some sense lives at the expense of the host. The true nature of parasitism involves an ecological relationship between the parasite and its host. A parasite is metabolically dependent on its host.
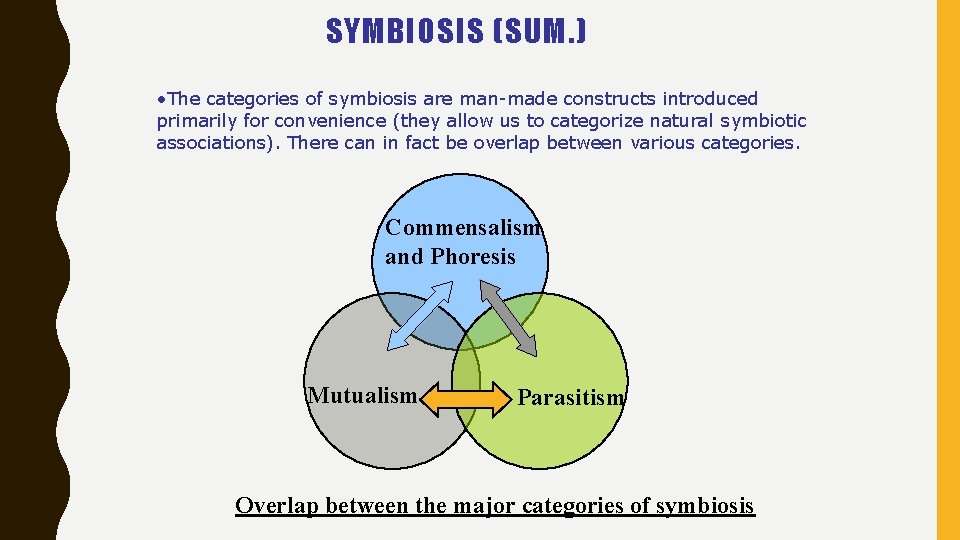
SYMBIOSIS (SUM. ) • The categories of symbiosis are man-made constructs introduced primarily for convenience (they allow us to categorize natural symbiotic associations). There can in fact be overlap between various categories. Commensalism and Phoresis Mutualism Parasitism Overlap between the major categories of symbiosis

Parasite — In the relationship known as parasitism, the partner lives in or on another from which it gains benefit, always smaller, is the parasite which to some degree injures its partner. — Parasites (animal parasites) are invertebrates that can not live independently and should depend upon others to maintain their lives (live at the expense of others).

—Parasites may be classified according to different ways: § residing site---endoparasite / ectoparasite § ecology---obligatory/facultative; accidental or opportunistic § duration of parasitism---permanent/intermittent

KINDS OF PARASITES • An organism that does not absolutely depend on the parasitic way of life, but is capable of adapting to it if placed in such a relationship is known as a facultative parasite • If an organism is completely dependent on the host during a segment or all of its life cycle the parasite is known as an obligatory parasite • Parasites that live within the body of their host (intestinal tract, liver, etc. ) are called endoparasites • Parasites that are attached to the outer surfaces of their hosts are called ectoparasites

HOST — Definition: In the parasitism, the partners which provide the food and shelter for parasites, and to some degree are injured by this association, are scientifically called as hosts. larger, more complex and better regulated bodies

TYPES OF THE HOST • A definitive host is the host in which the parasite become sexually mature (where the adult worm harbor or undergoing sexual reproduction). • An intermediate host is host in which the parasite undergoes larval development but does not reach sexual maturity, parasites often can undergo asexual reproduction in this type of host.

Definitions of Hosts (cont. ) Reservoir host is referred to those animals that harbor an infection that can be transmitted to humans. Even if the animal is the normal host of the parasite, it is the reservoir for the zoonotic infection of people. Thus, the reservoir host shares the same stage of the parasite with humans. Zoonosis: a disease of animals that may be transmitted to humans under natural conditions.

Definitions of Hosts (cont. ) A transport/paratenic/transfer host: When parasite enters the body of an abnormal host and does not undergo any development but continues to stay alive and be infective to the normal host.The host is called the transport host. Not necessary for the completion of the parasite’s life cycle

DEFINITIONS OF HOSTS (CONT. ) • Immune compromised hosts are persons who are considered to have reduced resistance to illness include: infants, hospital patients, pregnant women, frail, elderly people, malnourished individuals, people with controlled physical or metabolic disorders (e. g. , diabetes or high blood pressure), people with AIDS.

DEFINITIONS OF HOSTS (CONT. ) • Opportunistic parasitic infection: Any infection caused by a parasite that does not normally cause disease in humans; occurs in persons with abnormally functioning immune systems (as AIDS patients or transplant patients receiving immunosuppressive drugs). – For example: Toxoplasma gondii, Crypsosporidium Pneumocystis jeroveci (carinii)

Definitions of Hosts (cont. ) Vector parasite infections may be carried from one host to another by means of arthropod vectors. A vector may also be a host if development of the parasite takes place within its body. (If the arthropod is simply an instrument of passive transfer, we refer to it as a mechanical vector)

EPIDEMIOLOGY ü Although parasitic infections occur globally, the majority occur in tropical regions, where there is poverty, poor sanitation and personal hygiene ü Often entire communities may be infected with multiple, different organisms which remain untreated because treatment is neither accessible nor affordable ü Effective prevention and control requires "mass intervention strategies” and intense community education. Examples include: – General improved sanitation: pit latrines, fresh water wells, piped water – Vector control: insecticide impregnated bed nets, spraying of houses with residual insecticides, drainage, landfill – Mass screening and drug administration programmes which may need to be repeated at regular intervals
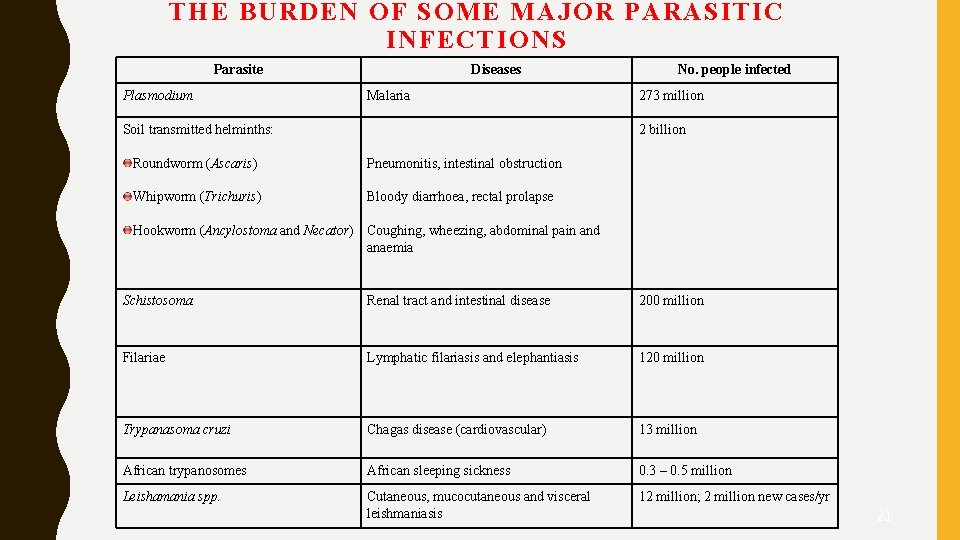
THE BURDEN OF SOME MAJOR PARASITIC INFECTIONS Parasite Plasmodium Diseases Malaria Soil transmitted helminths: No. people infected 273 million 2 billion Roundworm (Ascaris) Pneumonitis, intestinal obstruction Whipworm (Trichuris) Bloody diarrhoea, rectal prolapse Hookworm (Ancylostoma and Necator) Coughing, wheezing, abdominal pain and anaemia Schistosoma Renal tract and intestinal disease 200 million Filariae Lymphatic filariasis and elephantiasis 120 million Trypanasoma cruzi Chagas disease (cardiovascular) 13 million African trypanosomes African sleeping sickness 0. 3 – 0. 5 million Leishamania spp. Cutaneous, mucocutaneous and visceral leishmaniasis 12 million; 2 million new cases/yr 21

TAXONOMIC CLASSIFICATION OF PARASITIC ORGANISMS • Parasites form part of the animal kingdom which comprises some 800, 000 identified species categorised into 33 phyla (but it is estimated that there may be ~10 m species in total) • The parasitic organisms that are of importance for human health are eukaryotes - they have a well defined chromosome in a nuclear membrane (as opposed to prokaryotes which have no nuclear membrane, e. g. bacteria) • Parasites are classified into 2 sub-kingdoms: protozoa (unicellular) and metazoa (multicellular) • Protozoan (unicellular) parasites are classified according to morphology and means of locomotion. There are 45, 000 protozoa species. Most species that cause human disease belong to the phylum Sarcomastigophora and apicomplexa • Metazoa (multicellular) include the worms (helminths) and arthropods (posses an external skeleton) e. g. ticks, lice
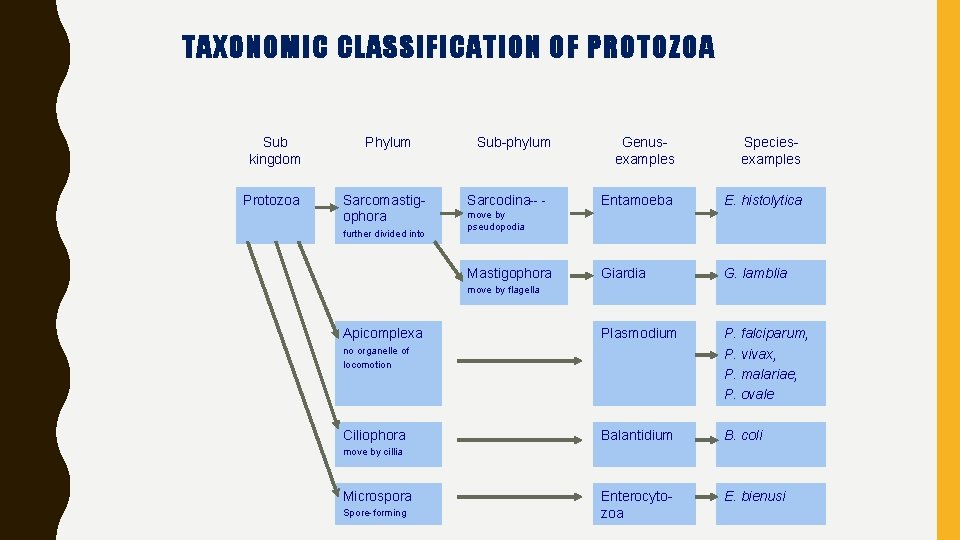
TAXONOMIC CLASSIFICATION OF PROTOZOA Sub kingdom Protozoa Phylum Sarcomastigophora further divided into Sub-phylum Sarcodina-- - Genusexamples Speciesexamples Entamoeba E. histolytica Giardia G. lamblia Plasmodium P. falciparum, P. vivax, P. malariae, P. ovale Balantidium B. coli Enterocytozoa E. bienusi move by pseudopodia Mastigophora move by flagella Apicomplexa no organelle of locomotion Ciliophora move by cillia Microspora Spore-forming
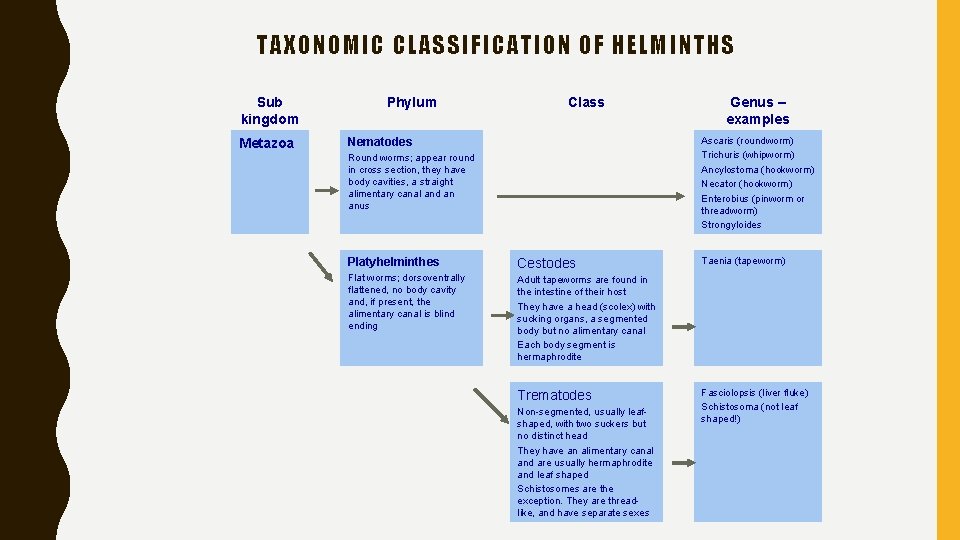
TAXONOMIC CLASSIFICATION OF HELMINTHS Sub kingdom Metazoa Phylum Class Genus – examples Ascaris (roundworm) Trichuris (whipworm) Ancylostoma (hookworm) Necator (hookworm) Enterobius (pinworm or threadworm) Strongyloides Nematodes Round worms; appear round in cross section, they have body cavities, a straight alimentary canal and an anus Platyhelminthes Cestodes Flat worms; dorsoventrally flattened, no body cavity and, if present, the alimentary canal is blind ending Adult tapeworms are found in the intestine of their host They have a head (scolex) with sucking organs, a segmented body but no alimentary canal Each body segment is hermaphrodite Trematodes Non-segmented, usually leafshaped, with two suckers but no distinct head They have an alimentary canal and are usually hermaphrodite and leaf shaped Schistosomes are the exception. They are threadlike, and have separate sexes Taenia (tapeworm) Fasciolopsis (liver fluke) Schistosoma (not leaf shaped!)

25
26

EXAMPLES OF IMPORTANT INTESTINAL PROTOZOA Transmitted by the faecal-oral route and cause diarrhoea Giardia lamblia: world-wide distribution, lives in the small intestine and results in malabsorption Entamoeba histolytica: may invade the colon and cause bloody diarrhoea – amoebic dysentery. Also causes amoebic liver abscess. Cryptosporidium parvum: more prevalent in the immunocompromised Cyclospora cyatenensis : parasitizes the small intestinal mucosa and may cause diarrhoea for several weeks Balantidium coli: A large motile ciliated parasite that lives in the colon of pigs, humans and rodents and can lead to colonic ulceration 27
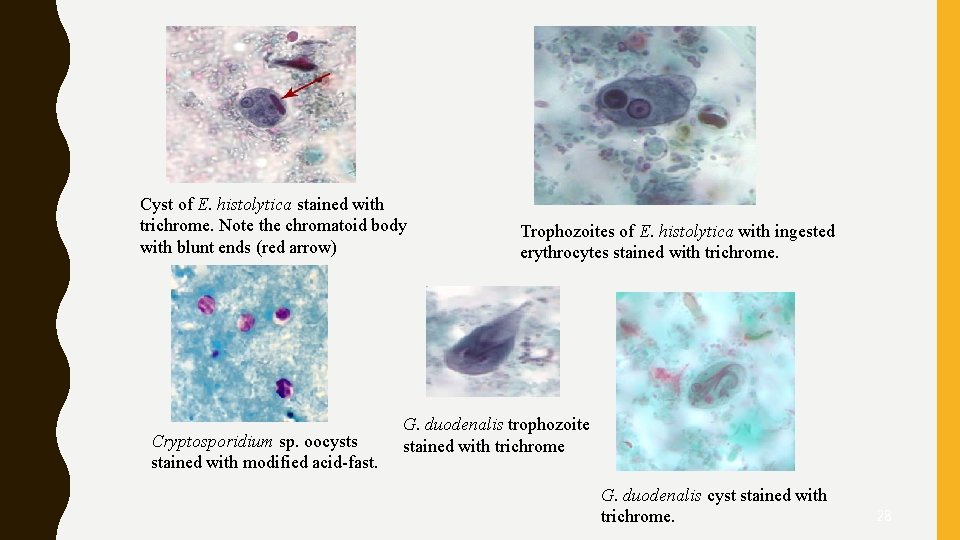
Cyst of E. histolytica stained with trichrome. Note the chromatoid body with blunt ends (red arrow) Cryptosporidium sp. oocysts stained with modified acid-fast. Trophozoites of E. histolytica with ingested erythrocytes stained with trichrome. G. duodenalis trophozoite stained with trichrome G. duodenalis cyst stained with trichrome. 28

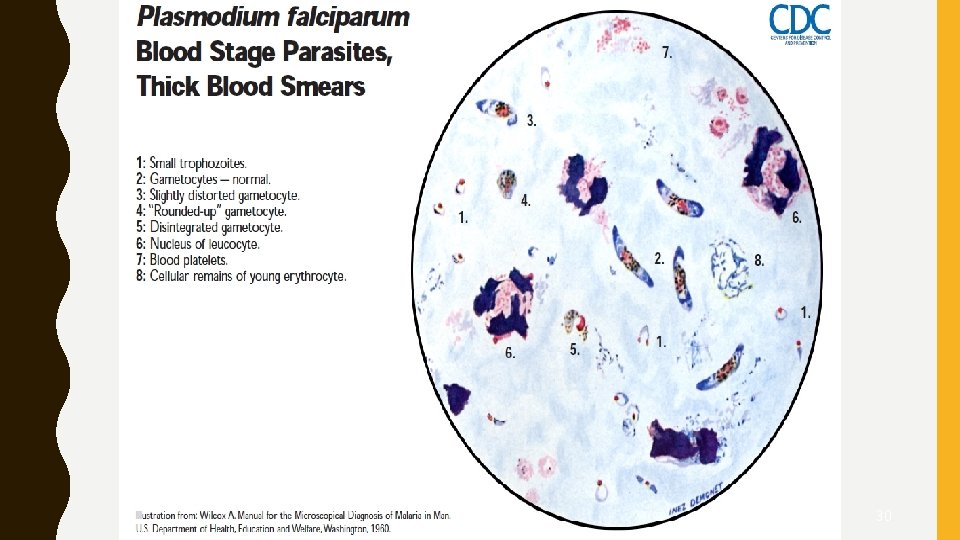
EXAMPLES OF IMPORTANT SYSTEMIC PROTOZOA Detected in the blood • Plasmodium: the cause of malaria. There are 4 species that infect man: P. falciparum, P. vivax, P. ovale and P. malariae • Toxoplasma gondii: transmitted by the ingestion of oocysts from cat faeces. Infection can lead to ocular problems and is also a cause of neonatal toxoplasmosis • Leishmania: transmitted by sand flies, can lead to visceral, cutaneous and mucocutaneous leishmaniasis Typical lesion of cutaneous leishmaniasis • Trypanosoma: haemoflagellates cause – In Africa - sleeping sickness (transmitted by the Tsetse fly) – In South America - Chagas disease (transmitted by the Reduviid bug) 29
30
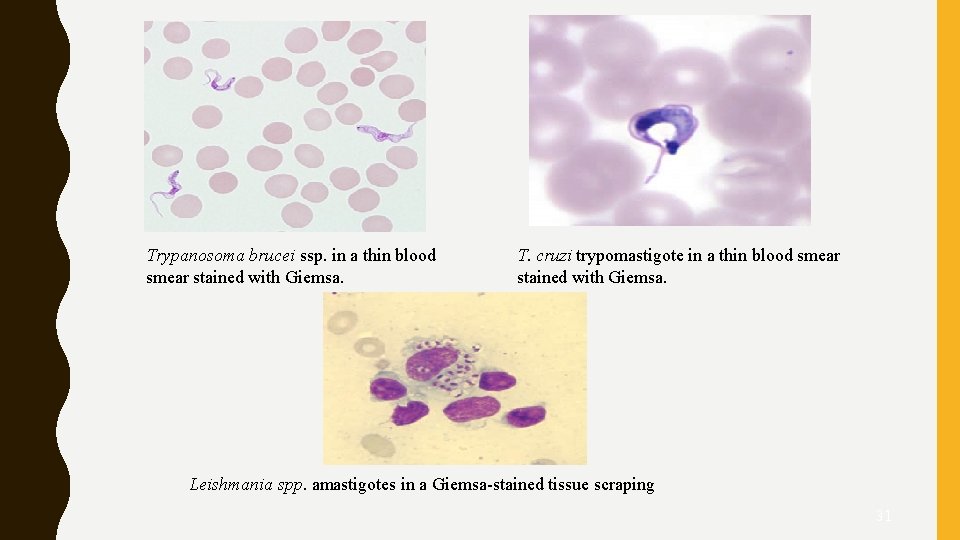
Trypanosoma brucei ssp. in a thin blood smear stained with Giemsa. T. cruzi trypomastigote in a thin blood smear stained with Giemsa. Leishmania spp. amastigotes in a Giemsa-stained tissue scraping 31

EXAMPLES OF IMPORTANT INTESTINAL NEMATODES • Trichuris (whipworm) – A soil transmitted helminth – Prevalent in warm, humid conditions – Can cause diarrhoea, rectal prolapse and anaemia in heavily-infected people • Ancylostoma and Necator (hookworms) – A major cause of anaemia in the tropics • Ascaris (roundworm) – Found world-wide in conditions of poor hygiene, transmitted by the faecal- oral route – Adult worms lives in the small intestine – Causes eosinophilia • Strongyloides • Enterobius (pinworm or threadworm) – Inhabits the small bowel – prevalent in cold and temperate climates but rare in the tropics – Infection more severe in immunospressed people (e. g. HIV/AIDS, malnutrition, – found mainly in children intercurrent disease) 32
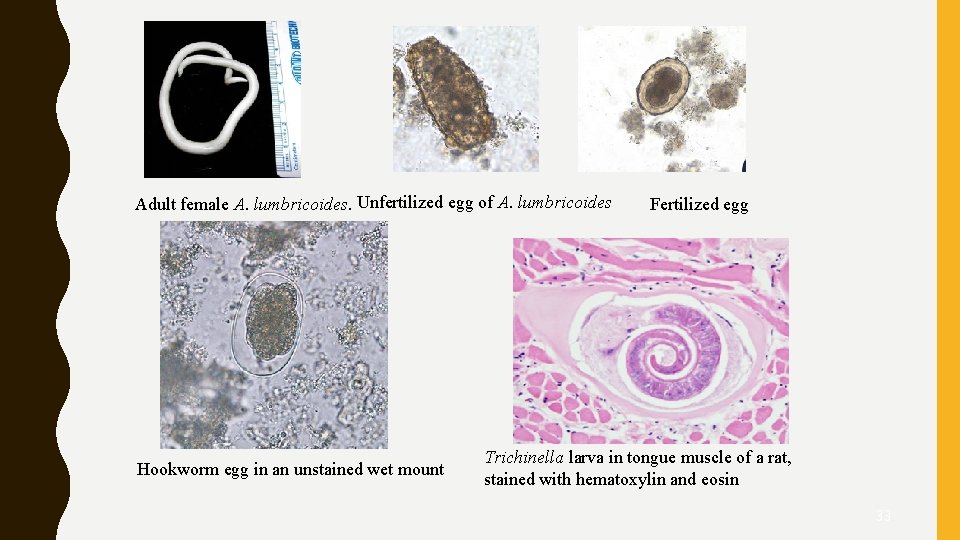
Adult female A. lumbricoides. Unfertilized egg of A. lumbricoides Hookworm egg in an unstained wet mount Fertilized egg Trichinella larva in tongue muscle of a rat, stained with hematoxylin and eosin 33

EXAMPLES OF IMPORTANT SYSTEMIC NEMATODES Filaria worms including: • Onchocerca volvulus : Transmitted by the simulium black fly, this microfilarial parasite can cause visual impairment, blindness and severe itching of the skin in those infected • Wuchereria bancrofti : The major causative agent of lymphatic filariasis Toxocara – A world-wide infection of dogs and cats – Human infection occurs when embryonated eggs are ingested from dog or cat faeces – It is common in children and can cause visceral larva migrans (VLM) • Brugia malayi : Another microfilarial parasite that causes Microfilaria of W. bancrofti in a thick blood lymphatic filariasis smear stained with Giemsa 34

EXAMPLES OF IMPORTANT FLATWORMS : CESTODES Intestinal : (“tapeworms”) Taenia saginata – worldwide – acquired by ingestion of contaminated, uncooked beef – a common infection but causes minimal symptoms Taenia solium – worldwide – acquired by ingestion of contaminated, uncooked pork that contains cystercerci – Less common, but causes cystercicosis – a systemic disease where cysticerci encyst in muscles and in the brain – may lead to epilepsy Systemic Echinococcus granulosus (dog tapeworm) and Echinicoccus multilocularis (rodent tapeworm) Hydatid disease occurs when the larval stages of these organisms are ingested The larvae may develop in the human host and cause spaceoccupying lesions in several organs, e. g. liver, brain 35
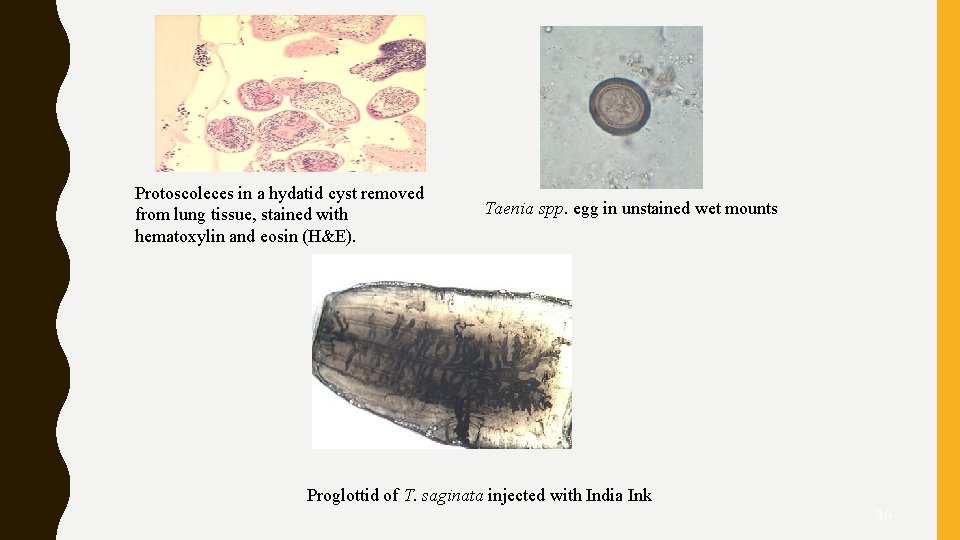
Protoscoleces in a hydatid cyst removed from lung tissue, stained with hematoxylin and eosin (H&E). Taenia spp. egg in unstained wet mounts Proglottid of T. saginata injected with India Ink 36

EXAMPLES OF IMPORTANT TREMATODES (FLUKES) Intestinal � Fasciolopsis buski : A common parasite of humans and pigs in South- east Asia. This parasite is one of the largest trematodes to infect man (8 cm in length) and lives in the upper intestine. Chronic infection leads to inflammation, ulceration and haemorrhage of the small intestine Paragonimus westermani ( lung fluke)Widespread in the Far East and South east Asia, the parasite is acquired by ingestion of infective metacercariae in raw or pickled crustaceans • Fasciola hepatica (liver fluke)- a parasite of sheep, humans become infected when ingest metacercariae that have encysted on watercress. The adult trematode lives in the intra-hepatic bile ducts of the liver. “Fascioliasis” can lead to severe anaemia in humans • Clonorchis sinensis (liver fluke): Widespread in China, Japan, Korea and Taiwan, this parasite is acquired by ingestion of infective metacercariae in raw or pickled fish • Schistosoma haematobium, S. mansoni and S. japonicum 37
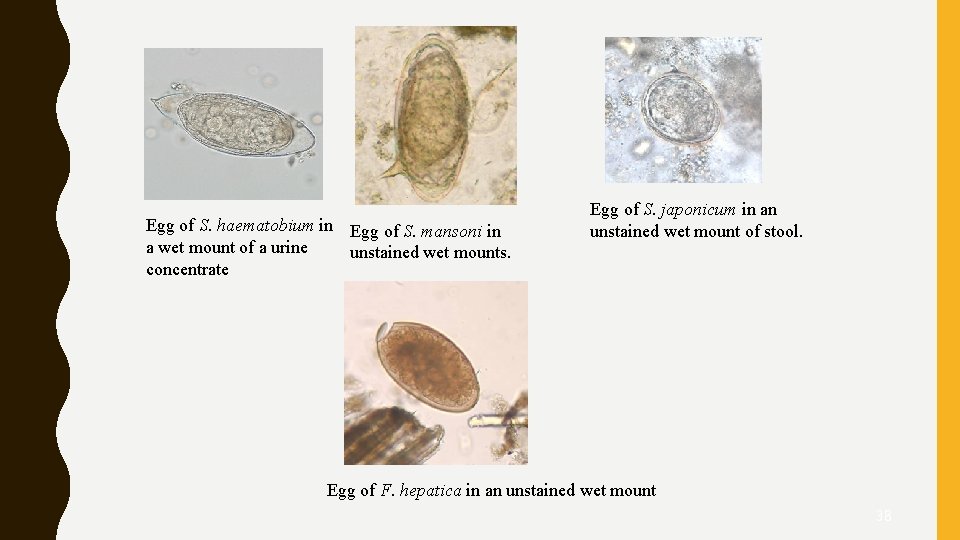
Egg of S. haematobium in Egg of S. mansoni in a wet mount of a urine unstained wet mounts. concentrate Egg of S. japonicum in an unstained wet mount of stool. Egg of F. hepatica in an unstained wet mount 38

WHAT IS LIFE CYCLE? Life cycle described the ontogenesis, development and reproduction of the parasite, tracking it through the various phases of its life history which will encompass both parasitic and non-parasitic stages. The key to understanding the transmission of a parasite species and parasitic disease is its life-cycle

GENERALIZED STAGES OF A PARASITE’S LIFE CYCLE Stage in human host (linking to pathogenesis) Stage to discharge (diagnostic stage) Stage developing outside human host: in intermediate host or insect host (transmission) Stage infecting men (infective stage) external environment,
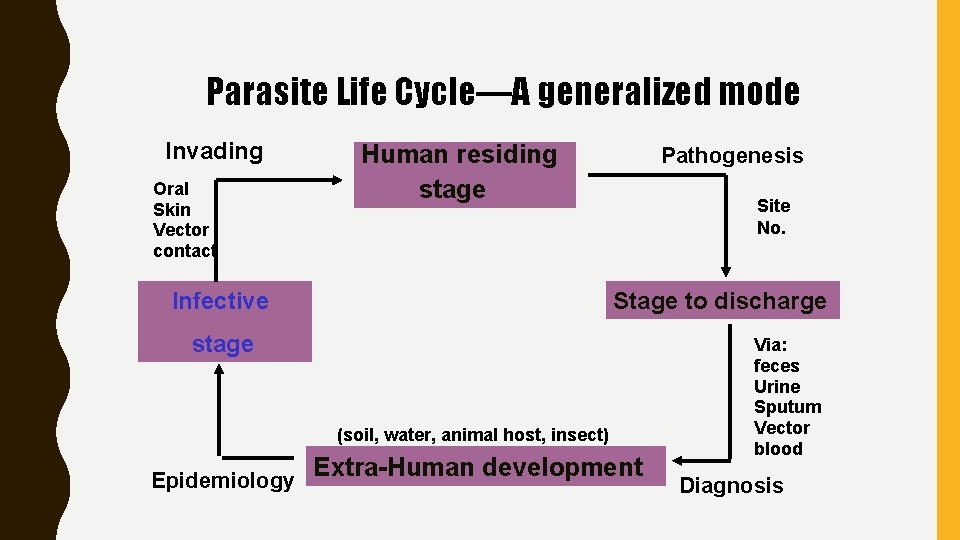
Parasite Life Cycle—A generalized mode Invading Oral Skin Vector contact Human residing stage Infective Pathogenesis Site No. Stage to discharge stage (soil, water, animal host, insect) Epidemiology Extra-Human development Via: feces Urine Sputum Vector blood Diagnosis

The types of life cycle of parasites Direct type: one host (definitive host) ---geo-helminth Indirect type: more one hosts(intermediate host(s) and definitive host) ---bio-helminth

LIFE CYCLE (CONT. ) • Simple or Direct Life Cycle (monoxenous) is one in which there is only one host where the parasite often spends most of its life, usually as an adult, and where it reproduces.
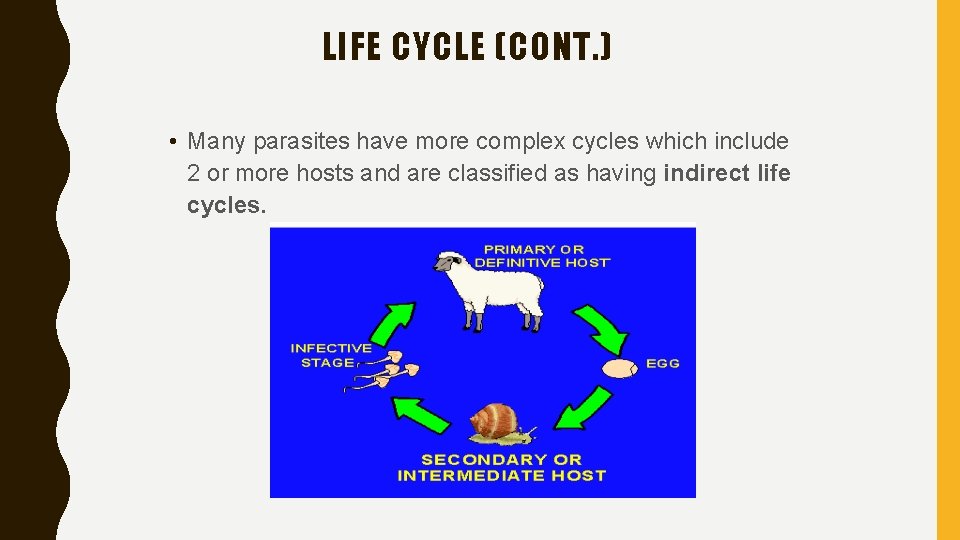
LIFE CYCLE (CONT. ) • Many parasites have more complex cycles which include 2 or more hosts and are classified as having indirect life cycles.

Host-parasite interactions Adaptations to parasitism l Profound morphological adaptation to their way of life l Organs not necessary to a parasitic existence are frequently lost or degenerated l Reproductive system is very highly developed in association with increased reproductive capacity l Specialized attachment organs in the form of suckers and hooks have been developed l Physiological and biochemical adaptations l Immune evasion

Host-parasite interactions THE HARMFUL EFFECTS ON THE HOST • Depriving for nutrition • Mechanical damage • Toxic effects • Immune-pathological consequences
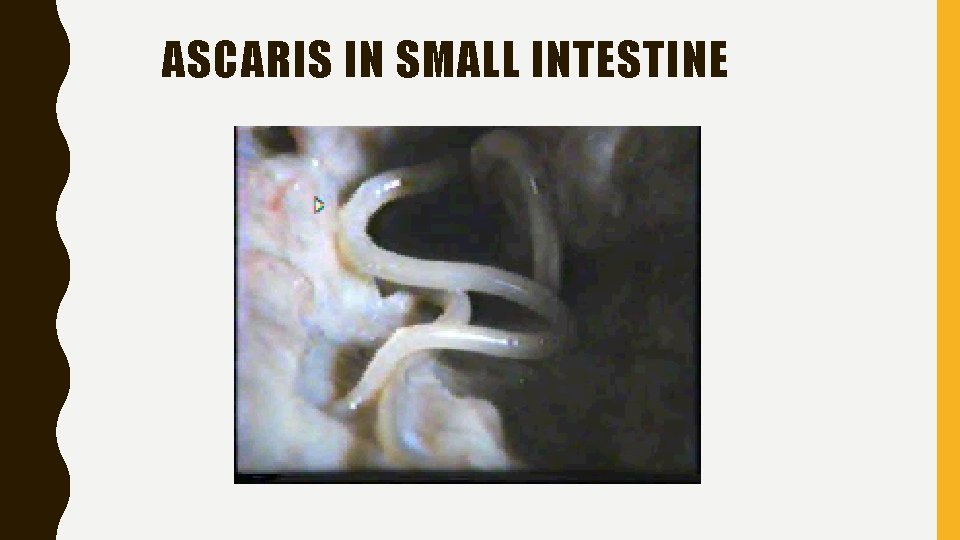
ASCARIS IN SMALL INTESTINE
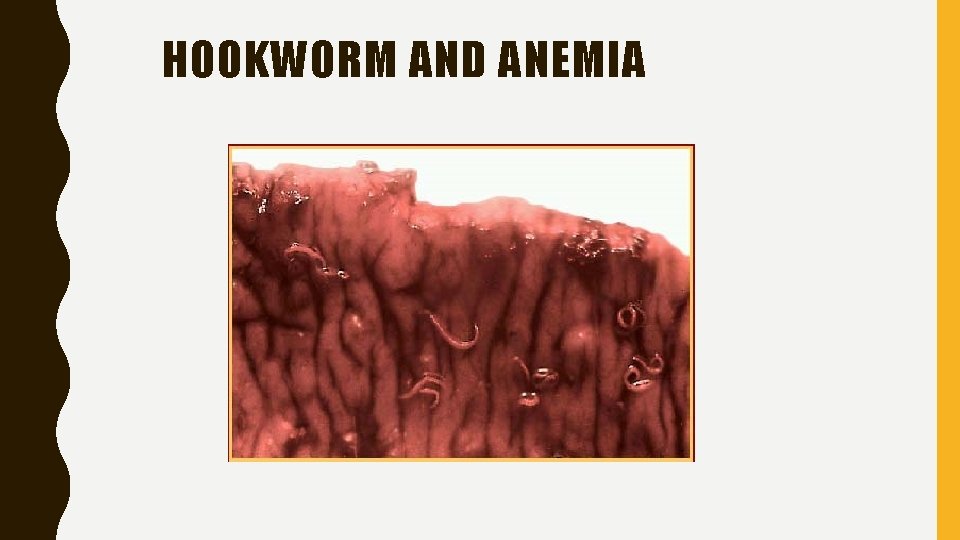
HOOKWORM AND ANEMIA
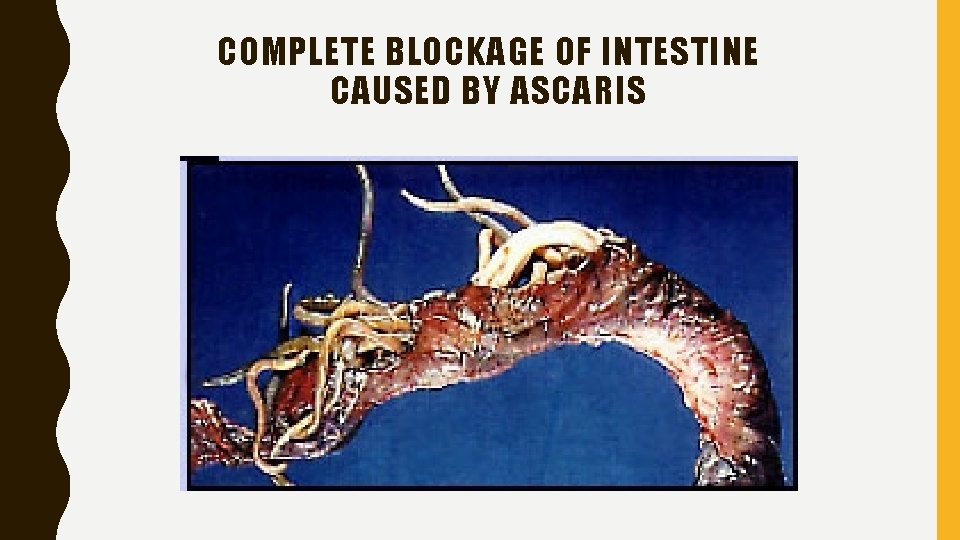
COMPLETE BLOCKAGE OF INTESTINE CAUSED BY ASCARIS
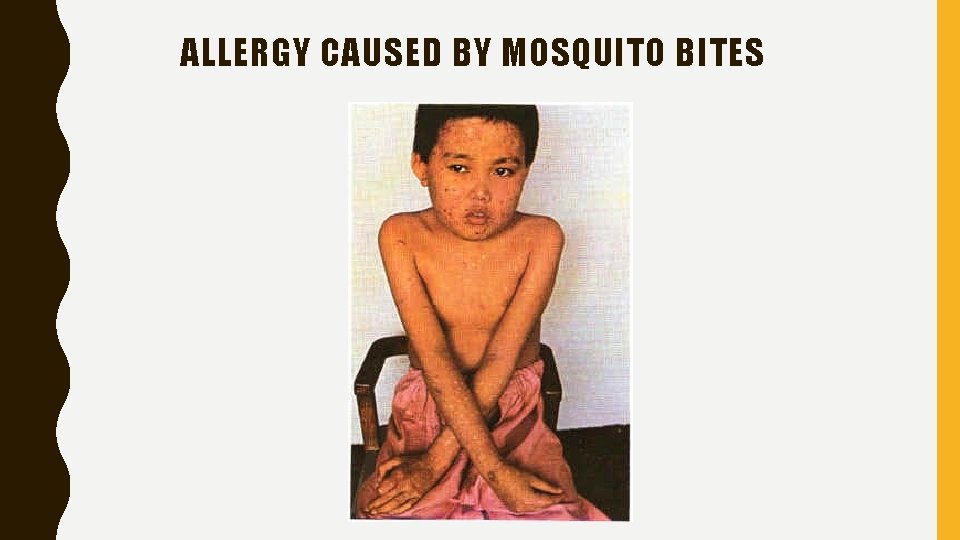
ALLERGY CAUSED BY MOSQUITO BITES
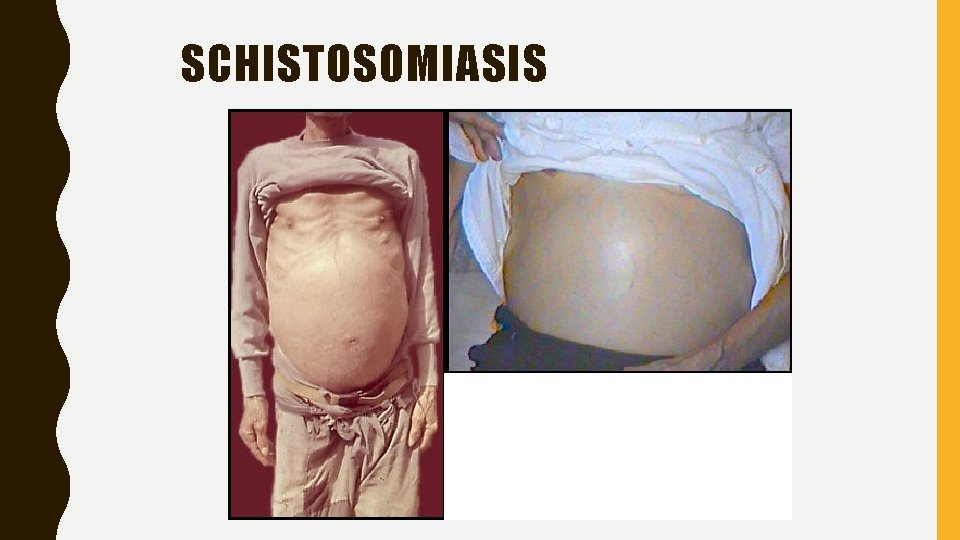
SCHISTOSOMIASIS

Host-parasite interactions Effects of the host to the parasite • Genetic constitution of the host may profoundly influence the host-parasite relationship (racial variations in resistance to certain strains of Plasmodium vivax; sickle cell trait increased resistance to infection with P. falciparum )

Host-parasite interactions Effects of the host to the parasite • Anti-parasitic immune responses üNatural immunity– muco-cutaneous barrier, blood brain barrier, phagocyte, complement, defensins… üAcquired immunity ØPremunition, concomitant immunity

Host-parasite interactions • Colonized (“infected”), asymptomatic – Differences in host susceptibility • Many people are asymptomatically infected with Toxoplasma gondii. – Capable of spreading microbe • Amoeba carrier • Colonized, infected, symptomatic • Infected, host death

CHARACTERISTICS OF PARASITIC DISEASE (INFECTION) • Acute/Chronic infection, carrier • Polyparasitism • Eosinophilia • Ig. E ↑ • Larva migrans • Ectopic parasitism

DIAGNOSIS OF PARASITIC INFECTIONS 1. Clinical 2. Laboratory Purpose of laboratory diagnosis : – Confirmation of clinical suspicion – Identification of unsuspected infection 56

Specimens v. Stool v. Blood v. Serum and plasma v. Others (anal swab, duodenal aspirate, sputum, urine, urogenital specimen) v. Tissues and aspirates 57

STOOL EXAMINATION Sample collection: • Sample is collected in clean, dry container • Handled carefully • Sometimes use preservative (10% formalin) • Samples in some cases fresh(amoeba, ciliates) • Liquid and soft stool examined within 15 min • Not mixed with urine or disinfectant (as they will kill trophozoites) • Specimens obtained by enema or laxatives are often positive for worm eggs or adult worm. 58

Examination of the stool sample: Gross examination: • Mucoid blood stained (acute amoebic dysentery), Parasites can be detected (nematodes, cestodes) Microscopic examination: • • Saline mount Iodine Mount Thick smears – not commonly used Permanent stained smears – Iron hematoxylene – Whearley’s trichrome stain – Concentration methods • Floatation techniques • Sedimentation techniques • Antigen detection • Molecular diagnosis 59

MICROSCOPIC EXAMINATION Direct wet mount: • Thin emulsion of small amount of faeces • Few drops of saline • Sometimes add lugol’s iodine (nuclear details, glycogen vacuole in cyst) • Protozoa (trophozoite), cyst, eggs and larva of helminths, crystals (charcot leyden) 60

CONCENTRATION METHODS • Scanty parasites in the sample • Floatation (eggs and cyst float , solution of high specific gravity) 1. Saturated sodium chloride (ascaris, hookworms) 2. Zinc sulphate centrifugation floatation (cyst, nematodes). • Sedimentation (solution of low specific gravity): Formol ether Egg count in 1 gram 61

Stoll’s technique for counting helminth egg 3 gm stool and 42 ml water 0. 15 ml on slid Multiply result in 100 Number in 1 gm 62

IMMUNODIAGNOSTIC (ANTIGEN DETECTION) • Fresh or preserved stool samples are the appropriate specimens Amebiasis • EIA kits are commercially available for detection of fecal antigens for the diagnosis of intestinal amebiasis. • These assays use monoclonal antibodies that detect the galactoseinhibitable adherence protein in the pathogenic E. histolytica. 63

Cryptosporidiasis Several kits are combined tests for Cryptosporidium, Giardia, and E. Histolytica. DFA test identifies oocysts in concentrated or unconcentrated fecal samples by using a fluorescein isothiocyanate (FITC)-labeled monoclonal antibody is the most sensitive. Giardiasis DFA assays may be purchased that employ FITC-labeled monoclonal antibody for detection of Giardia cysts. 64

MOLECULAR DIAGNOSIS (USIN G ST OO L SA MPLE) If an unequivocal identification of the parasite can not be made, the stool specimen can be analyzed using molecular techniques such as polymerase chain reaction (PCR). PCR amplified fragments can be analyzed by using restriction fragment length polymorphisms (RFLP) or DNA sequencing if further characterization is needed. 65

• Sample : Fresh stool should be kept cold or frozen till DNA extraction. Samples collected in a preservative should be compatible with molecular detection (Total. Fix, Unifix, modified PVA (Zn- or Cu-based), and Ecofix) • DNA Extraction better using commercially available kits (Qiagen) • PCR analysis: Conventional PCR: DNA is tested by PCR with diagnostic primers. Amplified DNA fragments are electrophoretically resolved on an agarose gel for analysis of results. Real-Time PCR The DNA amplification is monitored by measuring the fluorescence signal generated in the reaction vessel. The fluorescence signal is measured every 66 cycle and is proportional to the amount of accumulated PCR product.

BLOOD EXAMINATION • Fresh capillary blood of finger or ear lobe • Venous blood collected in EDTA (anticoagulant) Blood sample will be used for : – Microscopic examination(Thin Smear, Thick smear, Wet mount for microfilaria). • Molecular diagnosis • Detection of parasite antigen • Isolation of organisms • Special tests 67
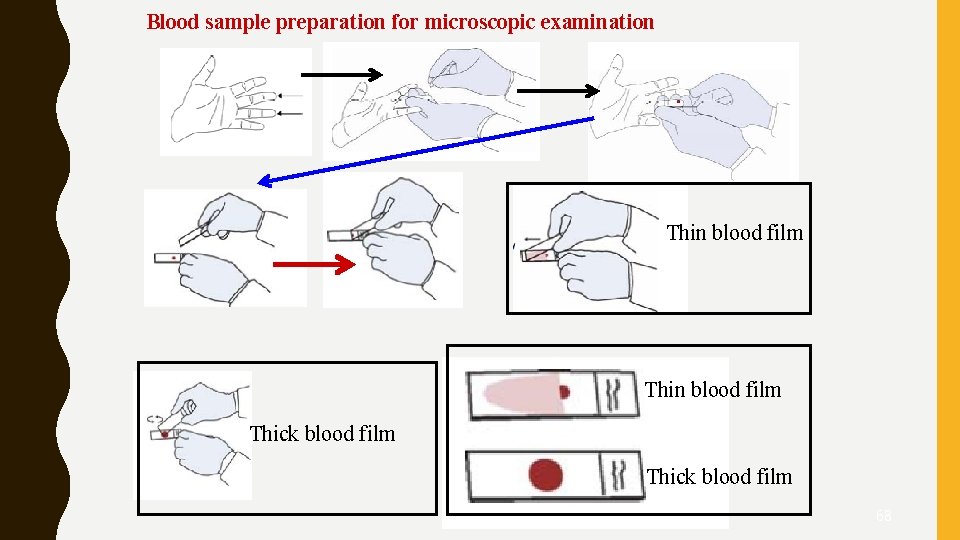
Blood sample preparation for microscopic examination Thin blood film Thick blood film 68
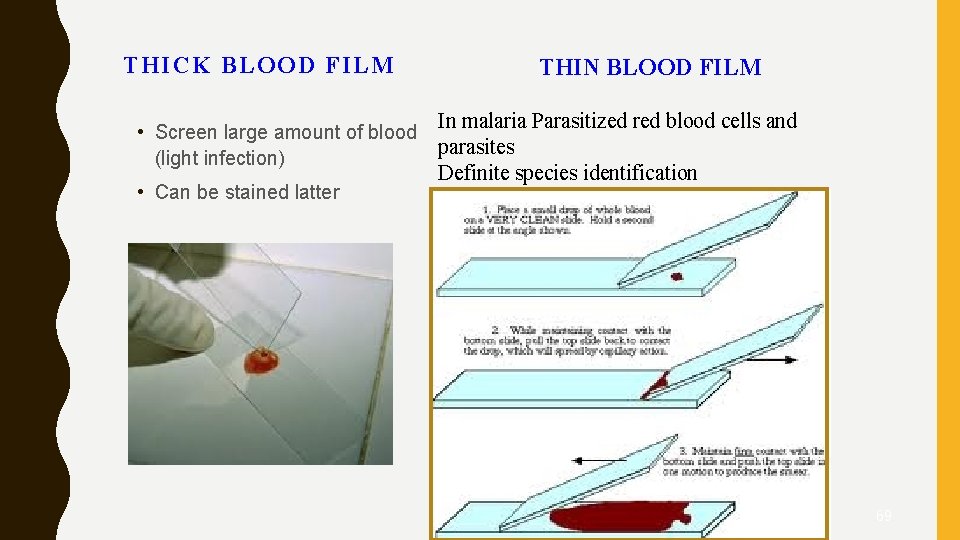
THICK BLOOD FILM • Screen large amount of blood (light infection) • Can be stained latter THIN BLOOD FILM In malaria Parasitized red blood cells and parasites Definite species identification 69
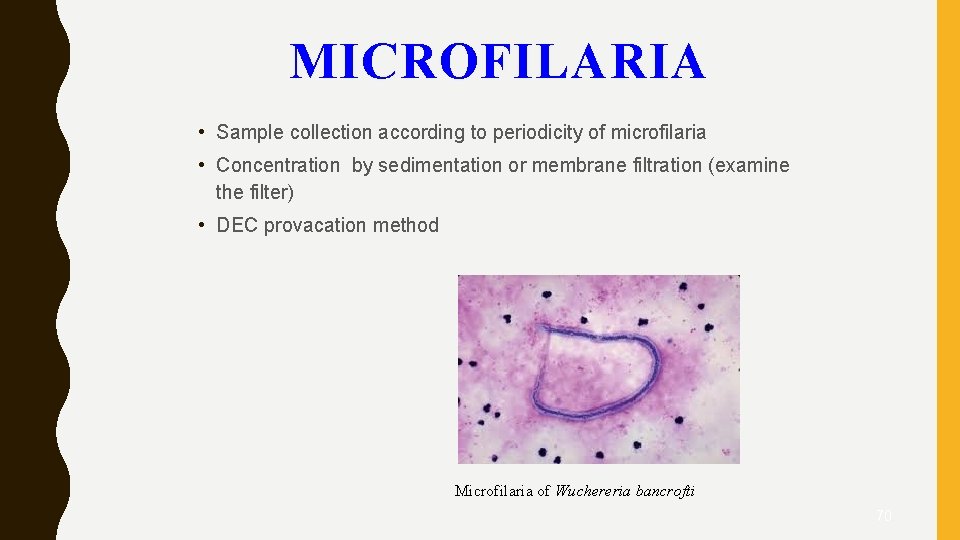
MICROFILARIA • Sample collection according to periodicity of microfilaria • Concentration by sedimentation or membrane filtration (examine the filter) • DEC provacation method Microfilaria of Wuchereria bancrofti 70

MOLECULAR DIAGNOSIS (USING BLOOD SAMPLE) • Collect a 1 -5 ml blood sample in tube with EDTA. • Blood can be collected on filter papers (e. g Whatman) • DNA is extracted using DNA extraction kits Species-specific diagnosis of malaria Detection and speciation of Plasmodium is done with a two step nested PCR using the primers of Snounou et al 1993. 71

DETECTION OF PARASITE ANTIGENS (IN BLOOD SAMPLE) Rapid diagnostic tests for malaria employing immunochromatographic methods based on the detection of malarial antigens present in peripheral blood 72

Isolation of Organisms (from blood) q. The diagnosis of Leishmania spp. is made by microscopic identification of the nonmotile, intracellular form (amastigote) in stained sections from lesions, and by culture of the motile, extracellular form (promastigote) on suitable media. q Slides should be fixed and stained before they are sent unless reagents are not available. q. Serologic tests are also available to detect for anti-leishmanial antibodies; however, these tests are often not sensitive, particularly for diagnosing cutaneous leishmaniasis. 73

Serum, plasma and others Specimen Requirements o. Serum/plasma is required for all parasitic disease immunodiagnostic tests. o. A single sample is sufficient; acute and convalescent specimens are not necessary. o. CSF and eye fluids (vitreous or aqueous) are acceptable for selected diseases) but MUST be accompanied by a serum specimen. Serum for all tests: 0. 5 ml serum/plasma separated from RBCs. CSF: 0. 5 ml. Acceptable only for cysticercosis and baylisascariasis testing. Eye fluids: 0. 1 ml neat fluid (no washings). Acceptable only for toxocariasis 74

• Serology – All tests available – IHA – ELISA • Skin Tests – CIEP – IF Specificity low, cross reactions common – CFT • More useful in Examples: – Amoebiasis – Leishmaniasis – Malaria – Toxoplasmosis – Trichinosis – Filariasis – Echinococcosis – Casoni’s test – Leishmanin test 75

CULTIVATION OF PARASITES Culture methods are used for : – Amoeba – Leishmania – Trypanosoma – Malarial parasite 76
ANIMAL INOCULATION • Leishmania (young hamster) • Trypanosomes (rat, mouse) • Toxoplasma (all lab animals) Xenodiagnosis: In Chagas’ disease Vector infected experimentally 77
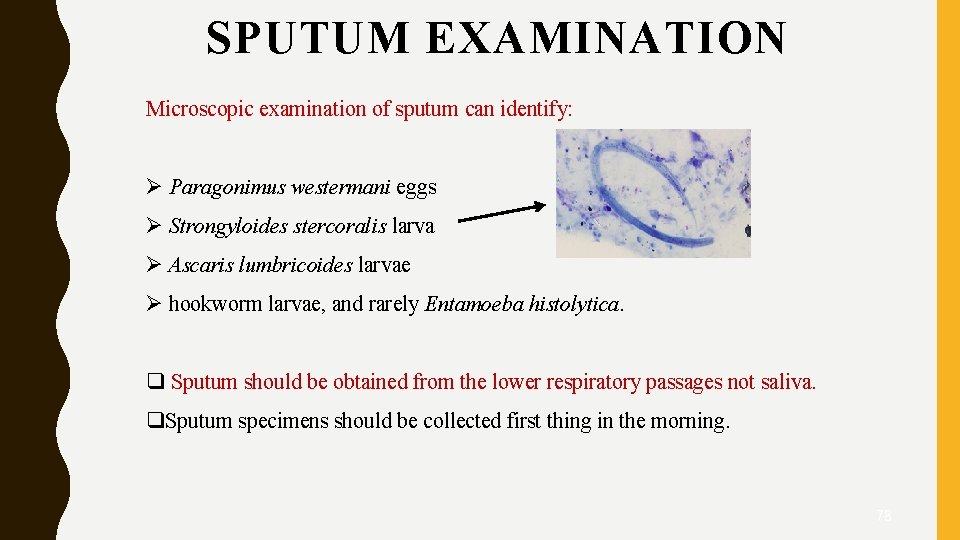
SPUTUM EXAMINATION Microscopic examination of sputum can identify: Ø Paragonimus westermani eggs Ø Strongyloides stercoralis larva Ø Ascaris lumbricoides larvae Ø hookworm larvae, and rarely Entamoeba histolytica. q Sputum should be obtained from the lower respiratory passages not saliva. q. Sputum specimens should be collected first thing in the morning. 78

A sputum sample can be examined in several ways: q. The unfixed specimen may be centrifuged and then the sediment examined as a direct wet mount. q. If the sputum is too viscous, an equal volume of 3% sodium hydroxide may be added, then centrifuge, and examine the sediment. q. The specimen can be preserved in 10% formalin and a formalin-ethyl acetate q The specimen can be preserved in PVA if protozoa are suspected and stained with trichrome stain. 79

VAGINAL SWABS §Demonstration of Trichomonas vaginalis trophozoites is usually done by preparing wet mounts made from vaginal swabs or scrapings. §If the specimen cannot be examined immediately, it should be preserved in PVA and stained smears examined later. 80

Tissue Specimens for Free-living Amebae (FLAs) Tissue specimens, including biopsy, surgical or necropsy specimens, may be collected for the detection of free-living amoebae (Naegleria, Balamuthia, and Acanthamoeba). The desired specimens include: • Tissue slides stained with hematoxylin and eosin (H&E). • Unstained slides (for indirect Immunofluorescence, or IIF). Acanthamoeba cyst • Unfixed brain tissue or CSF for PCR. • Unfixed corneal scrapings (for Acanthamoeba). • Paraffin-embedded tissue block. 81

Cellulose Tape or Swube Tube Procedure for Demonstration of Pinworm Eggs • The most reliable and widely used technique for demonstrating pinworm eggs (Enterobius vermicularis) is the cellulose tape or swube tube procedure. • The adhesive part of the swube tube or tape is applied to the perianal area first thing in the morning. • Specimens should be collected on three consecutive mornings prior to bathing. • If an infection is present, eggs and sometimes adult worms of Enterobius vermicularis will be present on the tape and can be seen under the microscope 82 E. Vermicularis egg

Urine Specimens Urinary schistosomiasis • Presence of S. haematobium eggs in urine is diagnostic for • Eggs usually shed in the urine around midday, so an optimum urine specimen for diagnosis should be collected at noon. • The specimen should be immediately centrifuged at 400 × g and the sediment examined by wet mount. Trichomonas vaginalis q. Motile trophozoites may also be found in the urine, especially in infected male patients. q. Urine specimen should be centrifuged at 400 × g, the sediment mixed with a drop or two of saline, and examined by wet mount. q. Temporary stains, such as methylene blue is helpful to see T. Vaginalis 83

Thank you 84
- Slides: 84