Introduction to Organic Compounds An organisms body is

Introduction to Organic Compounds • An organism’s body is composed of large compounds – Four Groups • • Carbohydrates (sugars, starches) lipids (fats) Proteins Nucleic acids (DNA, RNA) • These compounds contain six key elements • Carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), and sulfur (S) • Organic compounds: chemical compounds that make up tissues of living organisms © 2011 Pearson Education, Inc.

Carbon Chemistry • Carbon is a versatile atom – It has four electrons in an outer shell but can hold eight – Carbon wants to form bonds (covalent bonds), to make it more stable – Molecules that contain many carbon atoms can assume complex shapes • Simple molecule • Chains • Branches • Rings © 2011 Pearson Education, Inc.
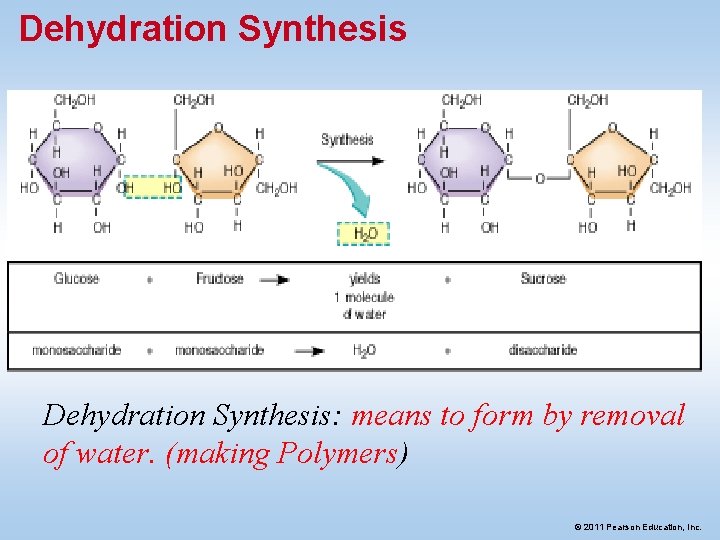
Dehydration Synthesis: means to form by removal of water. (making Polymers) © 2011 Pearson Education, Inc.
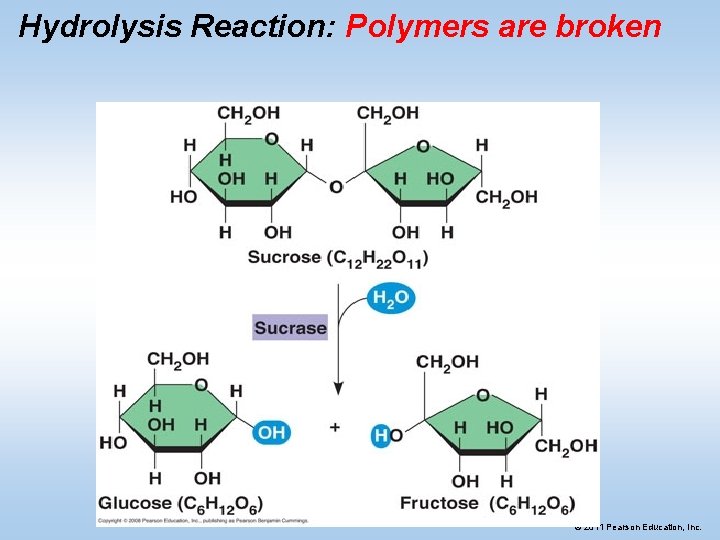
Hydrolysis Reaction: Polymers are broken © 2011 Pearson Education, Inc.

Large Molecules (polymers) are formed from Smaller Molecules (monomers) Horn – Examples: proteins and DNA are polymers Spiderweb Hair © 2011 Pearson Education, Inc.

I. Carbohydrates: Starch and Sugars – Function for quick and short term energy storage in all organisms. – Play a structural role in woody plants, fungi and animals © 2011 Pearson Education, Inc.
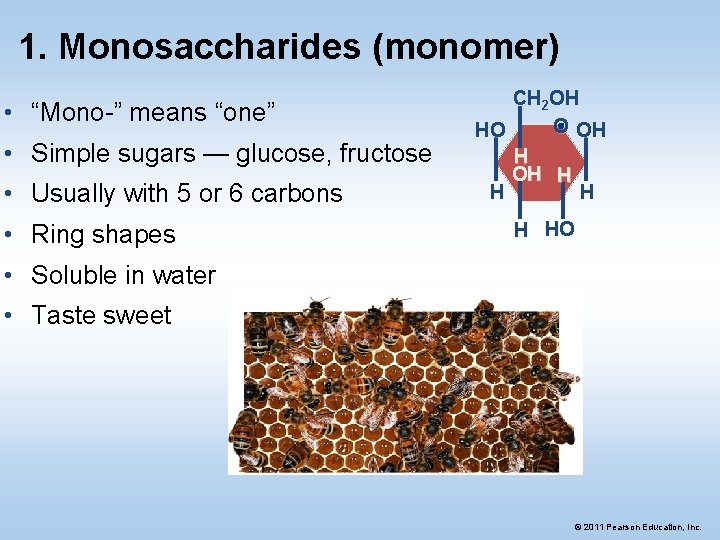
1. Monosaccharides (monomer) • “Mono-” means “one” • Simple sugars — glucose, fructose • Usually with 5 or 6 carbons • Ring shapes CH 2 OH O OH HO H OH H H HO • Soluble in water • Taste sweet © 2011 Pearson Education, Inc.

1. Monosaccharides (All have the chemical formula - C 6 H 12 O 6) They are isomers. • Glucose – Blood sugar • Fructose – Fruit sugar • Galactose – Part of milk sugar © 2011 Pearson Education, Inc.

2. Disaccharides (Dimer) • A disaccharide is a double sugar – Consists of two monosaccharides and dehydrtion synthesis. • Examples: • Sucrose (table sugar) -glucose + fructose = sucrose • Lactose (milk sugar) - glucose + galactose = lactose • Maltose (forms in digestive tract)glucose +glucose = maltose © 2011 Pearson Education, Inc.
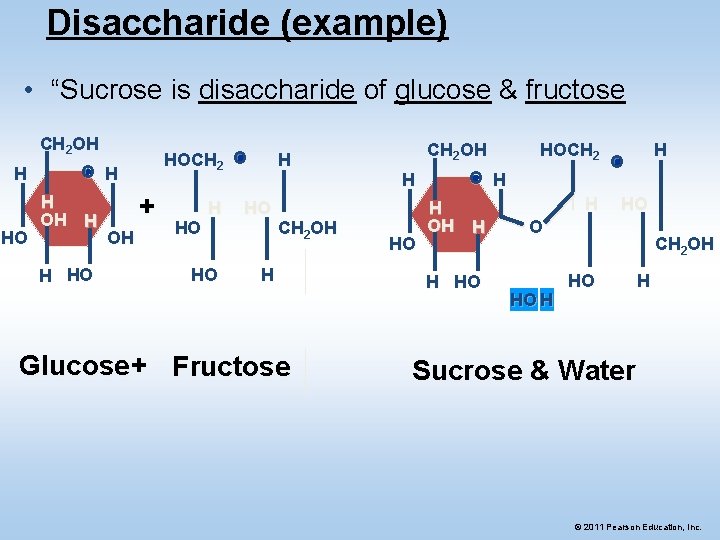
Disaccharide (example) • “Sucrose is disaccharide of glucose & fructose CH 2 OH H HO HOCH 2 O O H H OH H H HO + OH HO CH 2 OH H Glucose + Fructose CH 2 OH H HO HOCH 2 O H H OH H H HO H H O HO H CH 2 OH HO H Sucrose & Water © 2011 Pearson Education, Inc.

3. Polysaccharides (polymers) • Complex carbohydrates are called polysaccharides – They are long chains of sugar units • Examples: • Starch: energy storage in plants • Glycogen: long term energy storage in animals • Cellulose: structure of plant cell walls • Chitin: structure of fungi cell walls/ insect bodies © 2011 Pearson Education, Inc.
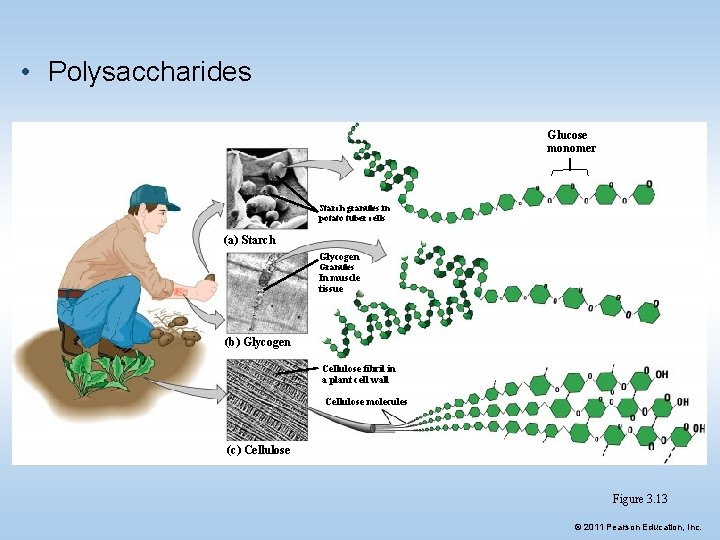
• Polysaccharides Glucose monomer Starch granules in potato tuber cells (a) Starch Glycogen Granules In muscle tissue (b) Glycogen Cellulose fibril in a plant cell wall Cellulose molecules (c) Cellulose Figure 3. 13 © 2011 Pearson Education, Inc.
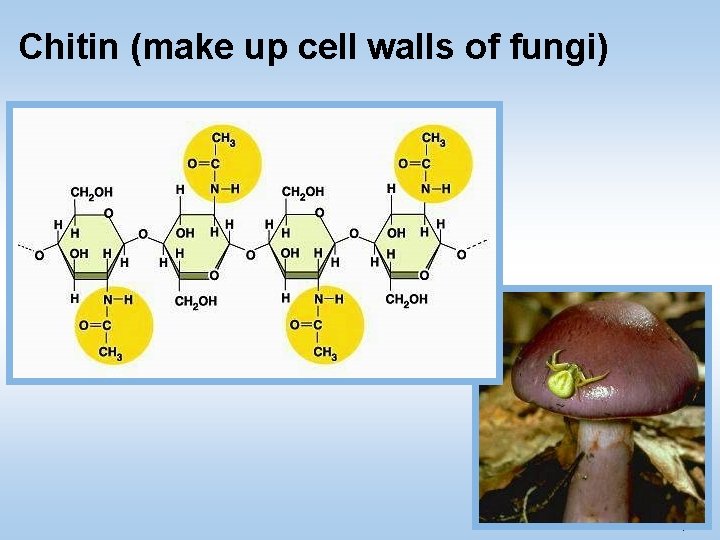
Chitin (make up cell walls of fungi) © 2011 Pearson Education, Inc.

II. Lipids • Lipids are hydrophobic (water fearing) – They do not mix with water – Examples: fats, oils, waxes and steroids – Functions: energy storage, insulation, hormones, and membrane structure © 2011 Pearson Education, Inc.
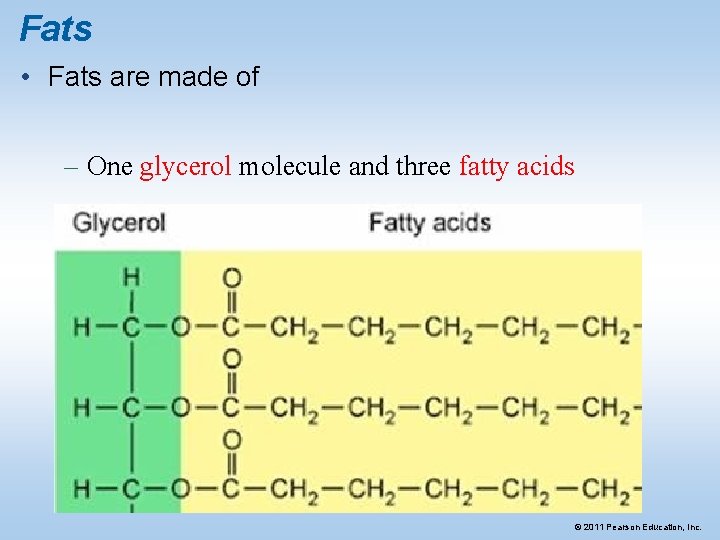
Fats • Fats are made of – One glycerol molecule and three fatty acids © 2011 Pearson Education, Inc.

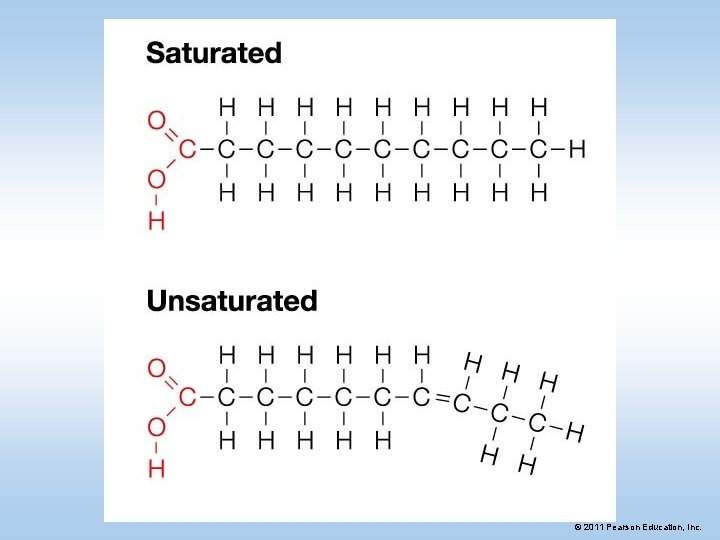
Types of Fats • Saturated Fats -have all single bonds, so saturated with hydrogens. • (butter, lard) • Animal source • Unsaturated Fats – at least one double bond • (olive oil) • Vegetable source • Polyunsaturated – many double bonds • (vegetable oil-vegetable source) © 2011 Pearson Education, Inc.
© 2011 Pearson Education, Inc.

III. Proteins • Molecules composed of one or more chains of amino acids (monomer). • Amino acid + Amino acid = protein. • 20 amino acids make up all the proteins. • Proteins (polymer) perform most of life’s functions – Enzymes: guide chemical reactions – Structural: keratin – hair, horns, claws – Energy: albumin – egg – Transport: hemoglobin – Hormones: insulin – Antibodies: fight disease – Poisons: venomous snakes © 2011 Pearson Education, Inc.

• Types of proteins (d) Transport proteins (b) Storage proteins (a) Structural proteins (c) Contractile proteins Figure 3. 18 © 2011 Pearson Education, Inc.

IV. Nucleic Acids • Nucleic acids are long chains of subunits called nucleotides – Transmit hereditary information – Determine what proteins a cell makes • There are two types of nucleic acids • DNA: Deoxyribonucleic Acid • RNA: Ribonucleic Acid © 2011 Pearson Education, Inc.
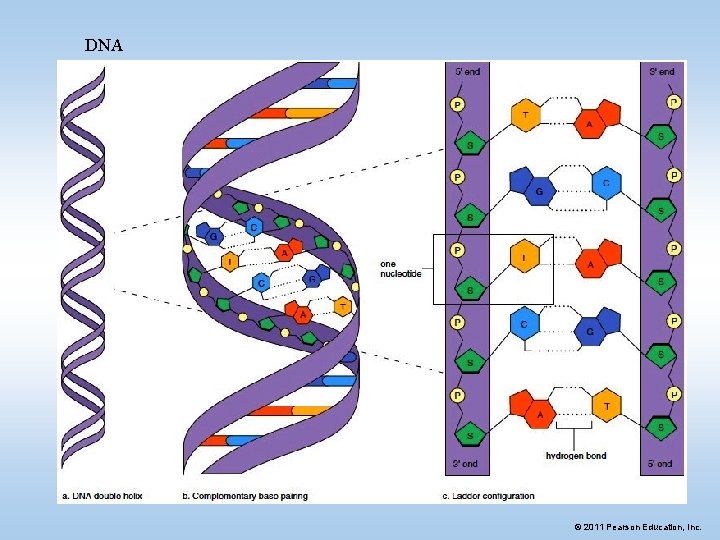
DNA © 2011 Pearson Education, Inc.
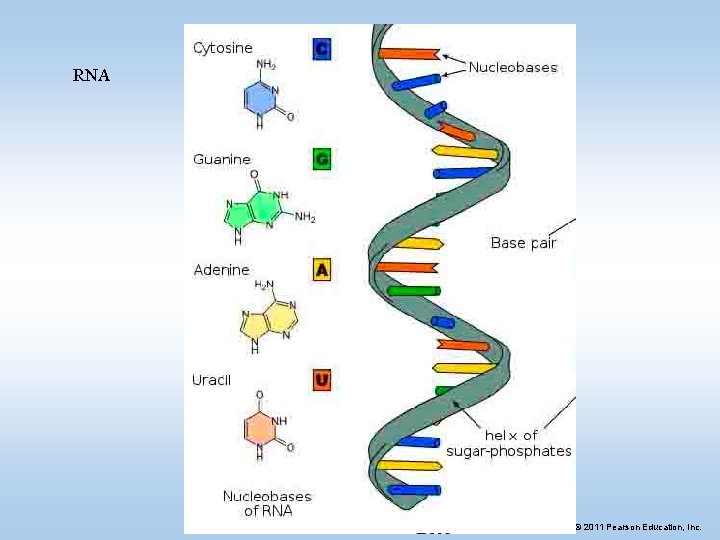
RNA © 2011 Pearson Education, Inc.
- Slides: 22