INTRODUCTION TO ORGANIC CHEMISTRY ELEMENTS IN ORGANIC MOLECULES











- Slides: 20

INTRODUCTION TO ORGANIC CHEMISTRY

ELEMENTS IN ORGANIC MOLECULES § There are six elements that are most common in organic molecules: § Carbon (C) § Hydrogen (H) § Oxygen (O) § Nitrogen (N) § Phosphorus (P) § Sulfur (S) CHONPS (Like CHOMPS, but with an N!)

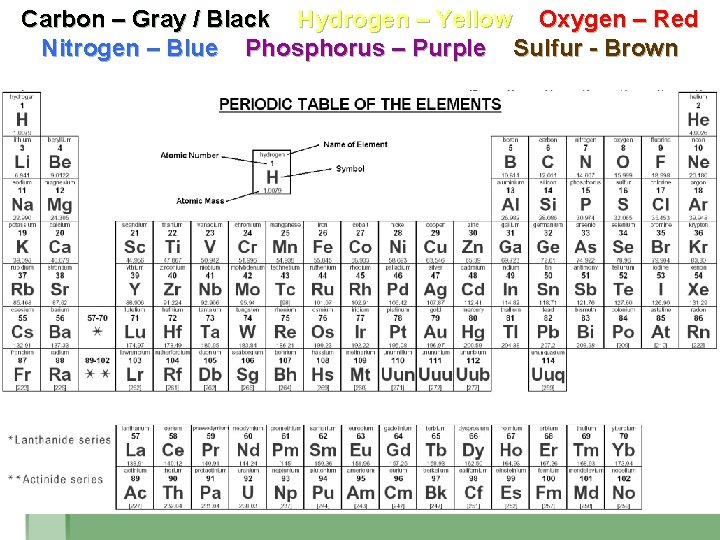
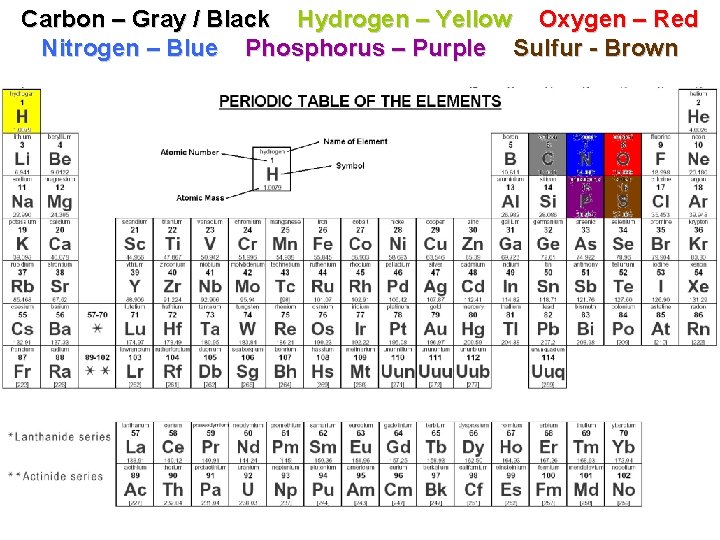
COLOR IN THE ELEMENTS § Use colored pencils or markers to color in the following: § Carbon – Gray / Black § Hydrogen – Yellow § Oxygen - Red § Nitrogen – Blue § Phosphorus – Purple § Sulfur - Brown

Carbon – Gray / Black Hydrogen – Yellow Oxygen – Red Nitrogen – Blue Phosphorus – Purple Sulfur - Brown

Carbon – Gray / Black Hydrogen – Yellow Oxygen – Red Nitrogen – Blue Phosphorus – Purple Sulfur - Brown

ORGANIC MOLECULES § Organic means: “contains carbon” § 90% of all known compounds are organic

CARBON § Carbon can form covalent bonds with 4 other atoms. § Because Carbon can bond with four other atoms, Carbon can help make many different structures. This helps to ensure diversity of life on earth! H H C H H

COVALENT BONDS § In covalent bonds, atoms share electrons § They are the strongest type of chemical bond § Atoms can share one, two or three electrons.

TYPES OF COVALENT BONDS § Share one electron: Single bond C-C § Share two electrons: Double bond C=C § Share three electrons: Triple bond CΞ C

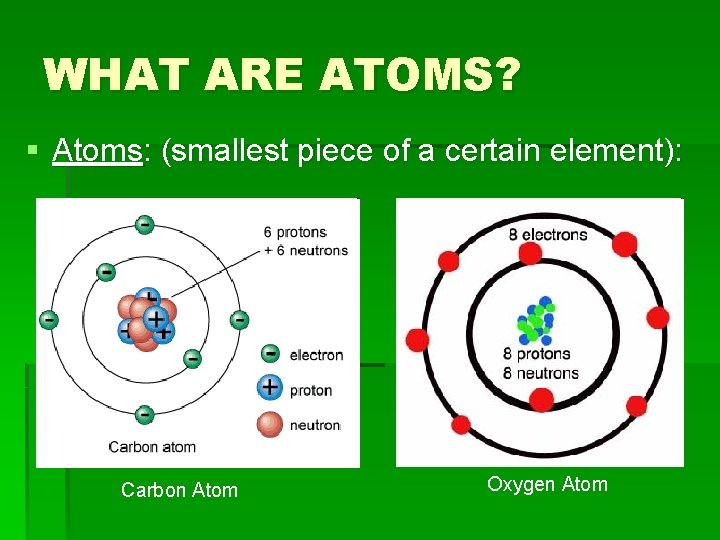
WHAT ARE ATOMS? § Atoms: (smallest piece of a certain element): Carbon Atom Oxygen Atom

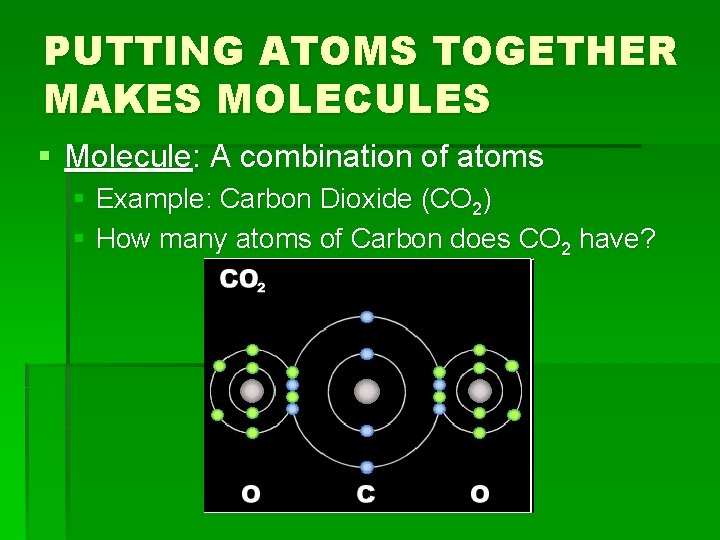
PUTTING ATOMS TOGETHER MAKES MOLECULES § Molecule: A combination of atoms § Example: Carbon Dioxide (CO 2) § How many atoms of Carbon does CO 2 have?

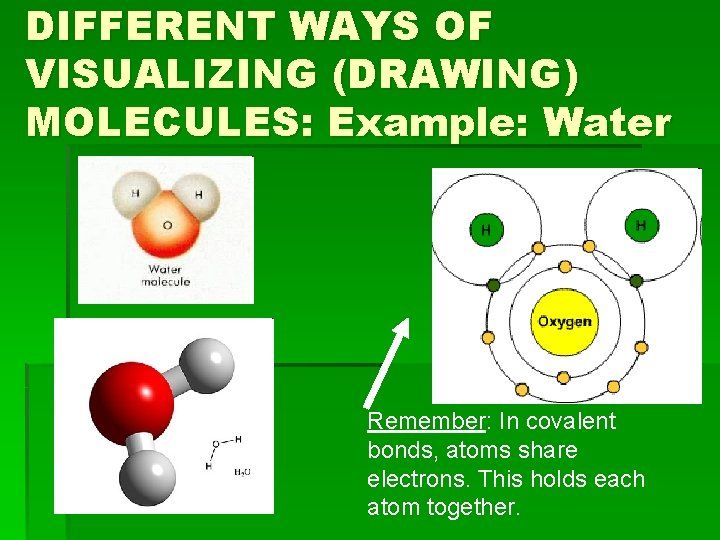
DIFFERENT WAYS OF VISUALIZING (DRAWING) MOLECULES: Example: Water Remember: In covalent bonds, atoms share electrons. This holds each atom together.

MONOMER VS. POLYMER: § Monomer: The smallest piece of a type of molecule § Also called a “subunit” § Polymer: many monomers put together to make a larger molecule

THE FOUR MOST COMMON TYPES OF ORGANIC MOLECULES § Carbohydrates: such as sugar and pasta § Lipids: such as butter and olive oil § Proteins: such as meat, nuts and soy § Nucleic Acids: such as DNA

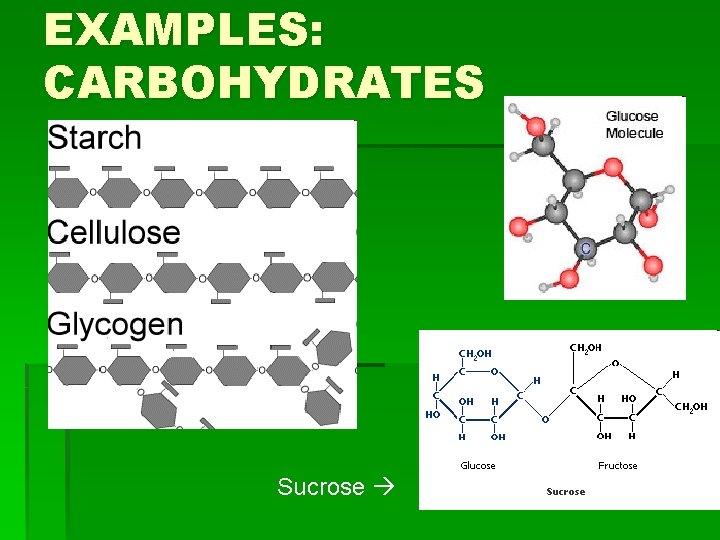
EXAMPLES: CARBOHYDRATES Sucrose

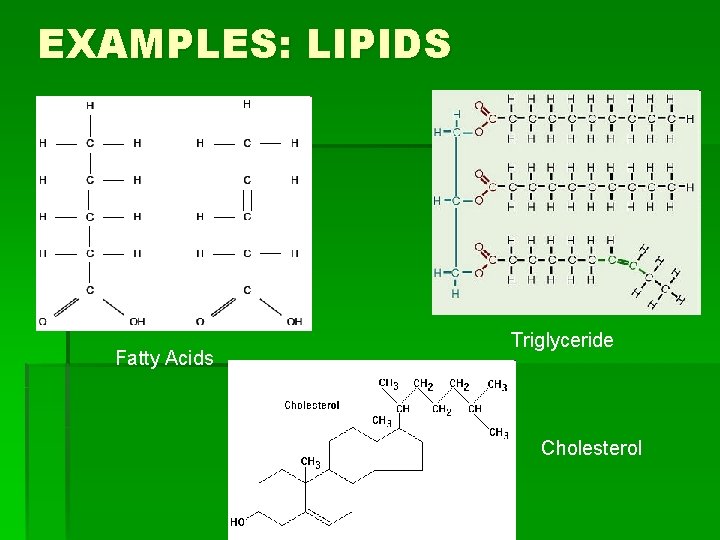
EXAMPLES: LIPIDS Fatty Acids Triglyceride Cholesterol

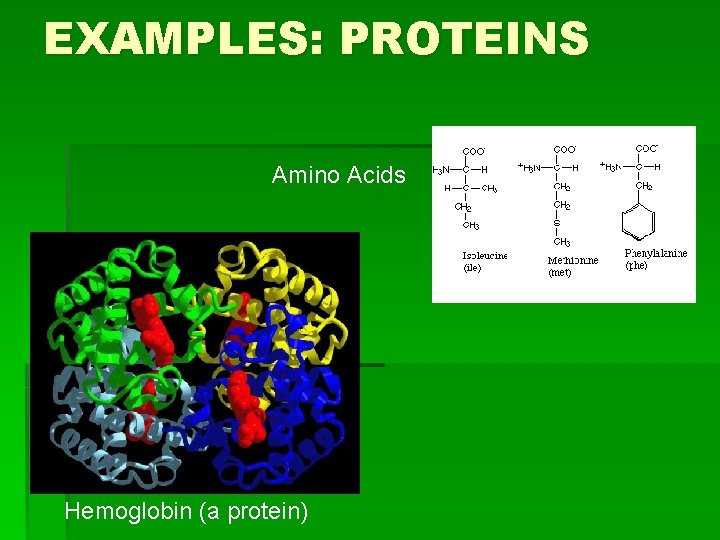
EXAMPLES: PROTEINS Amino Acids Hemoglobin (a protein)

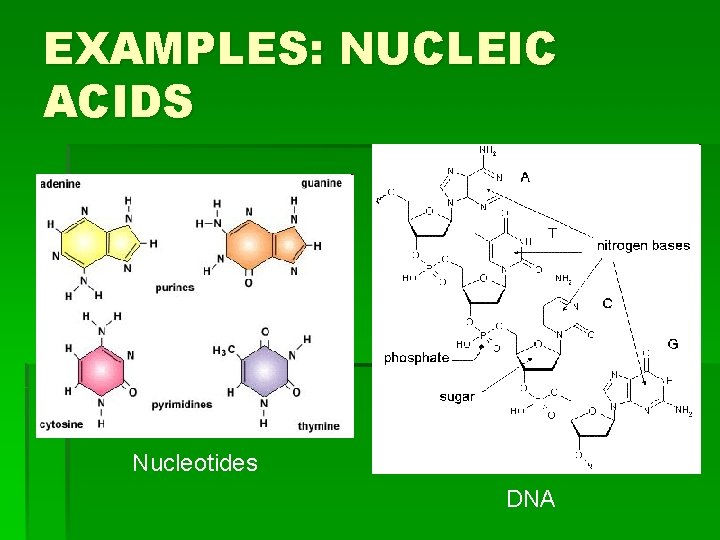
EXAMPLES: NUCLEIC ACIDS Nucleotides DNA

HOW TO REMEMBER STRUCTURES § Carbohydrates: § Have a 5 -Carbon ring form, or often have many rings joined in a line (or branching from each other) by an oxygen molecule § Lipids: § Have long hydrocarbon chains (chains of hydrogen and carbon atoms), and steroids have rings joined at the side.

HOW TO REMEMBER STRUCTURES…continued § Proteins: Carbon atom with four “groups” coming off of it: § 1) COO- 2) H 3)H 3 N 4) random § Or, the molecule is made of many of these Carbon atom groups joined together and looks “globular” § Nucleic Acids: single 6 -Carbon ring structures, or a 6 -C ring fused to a 5 -C ring. Many of these joined together