Introduction to Organic Chemistry Carbon the Building Blocks

Introduction to Organic Chemistry Carbon & the Building Blocks of Life 2007 -2008

Why study Carbon? All of life is built on carbon Cells q q ~72% H 2 O ~25% carbon compounds q carbohydrates lipids proteins nucleic acids ~3% salts Na, Cl, K…
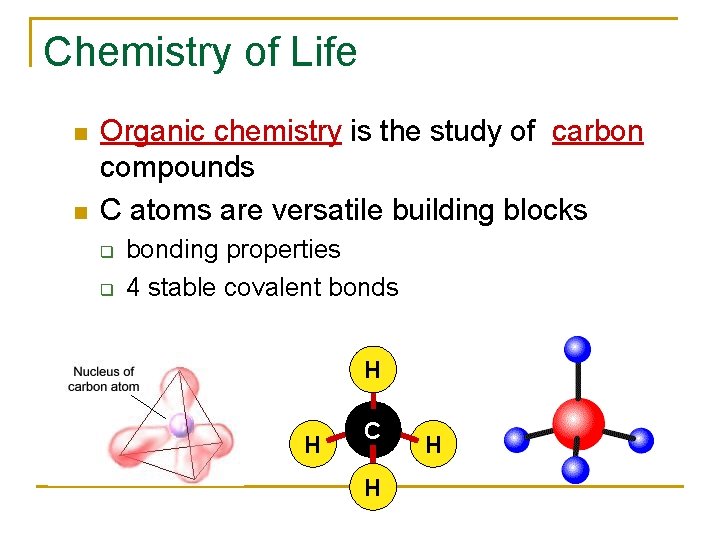
Chemistry of Life Organic chemistry is the study of carbon compounds C atoms are versatile building blocks q q bonding properties 4 stable covalent bonds H H C H H
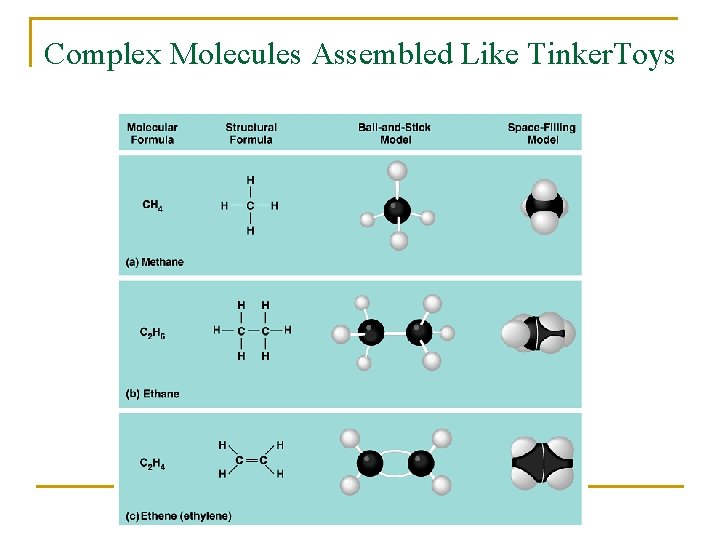
Complex Molecules Assembled Like Tinker. Toys
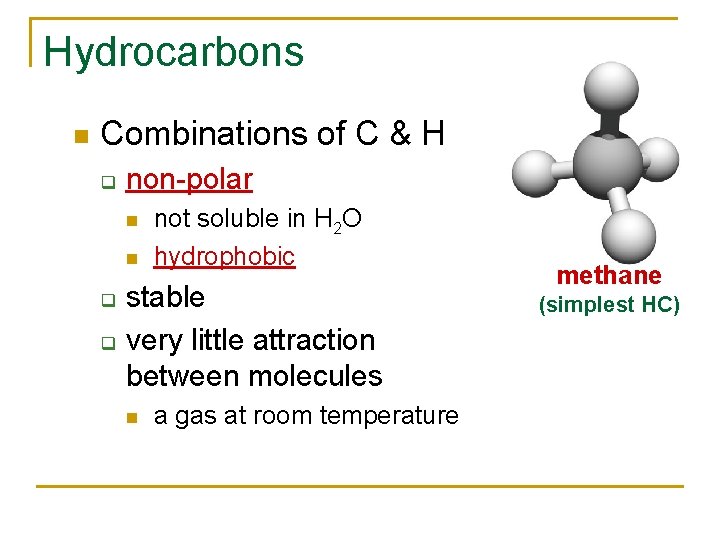
Hydrocarbons Combinations of C & H q non-polar q q not soluble in H 2 O hydrophobic stable very little attraction between molecules a gas at room temperature methane (simplest HC)
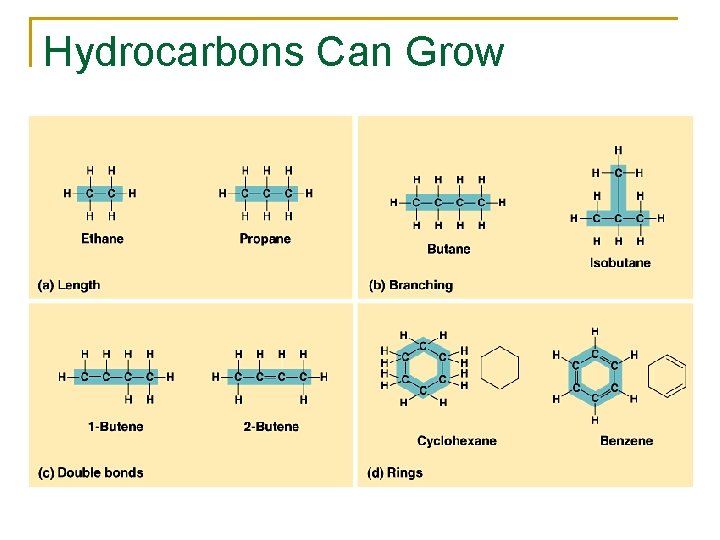
Hydrocarbons Can Grow
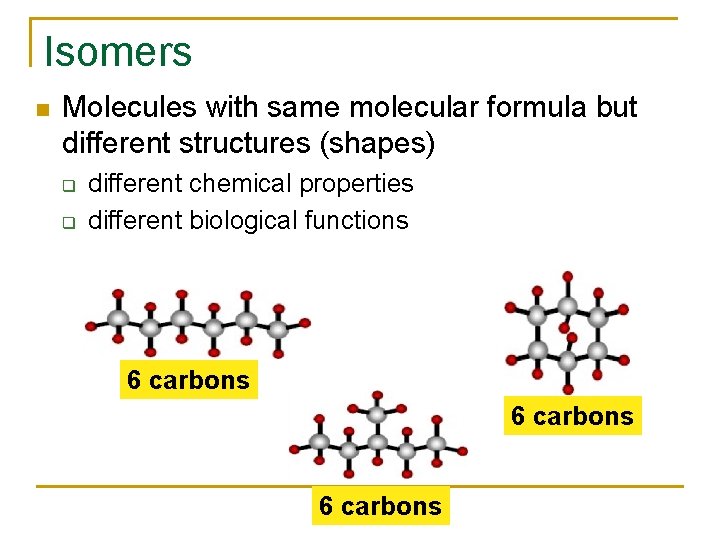
Isomers Molecules with same molecular formula but different structures (shapes) q q different chemical properties different biological functions 6 carbons
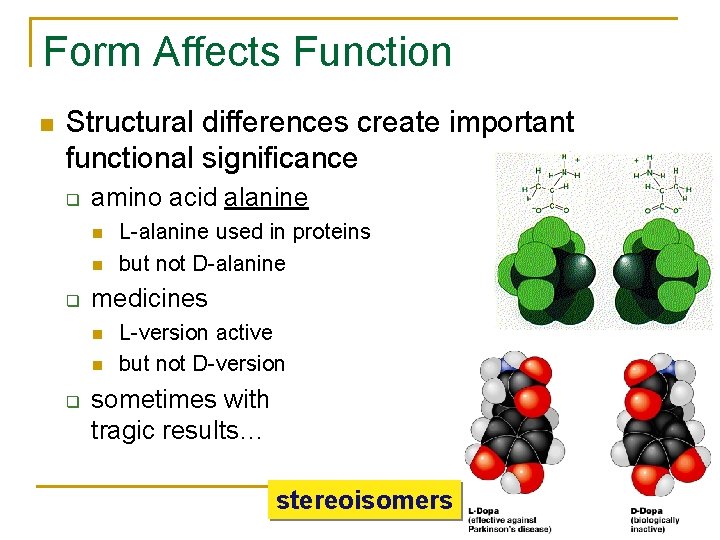
Form Affects Function Structural differences create important functional significance q amino acid alanine q medicines q L-alanine used in proteins but not D-alanine L-version active but not D-version sometimes with tragic results… stereoisomers

Form Affects Function Thalidomide q q q prescribed to pregnant women in 50 s & 60 s reduced morning sickness, but… stereoisomer caused severe birth defects
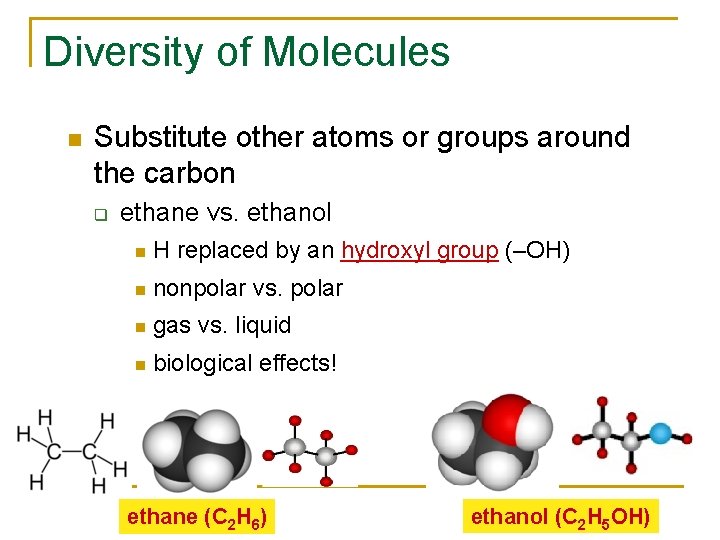
Diversity of Molecules Substitute other atoms or groups around the carbon q ethane vs. ethanol H replaced by an hydroxyl group (–OH) nonpolar vs. polar gas vs. liquid biological effects! ethane (C 2 H 6) ethanol (C 2 H 5 OH)

Functional Groups Parts of organic molecules that are involved in chemical reactions q give organic molecules distinctive properties hydroxyl carbonyl carboxyl amino sulfhydryl phosphate Affect reactivity q q makes hydrocarbons hydrophilic increase solubility in water (alter polarity)
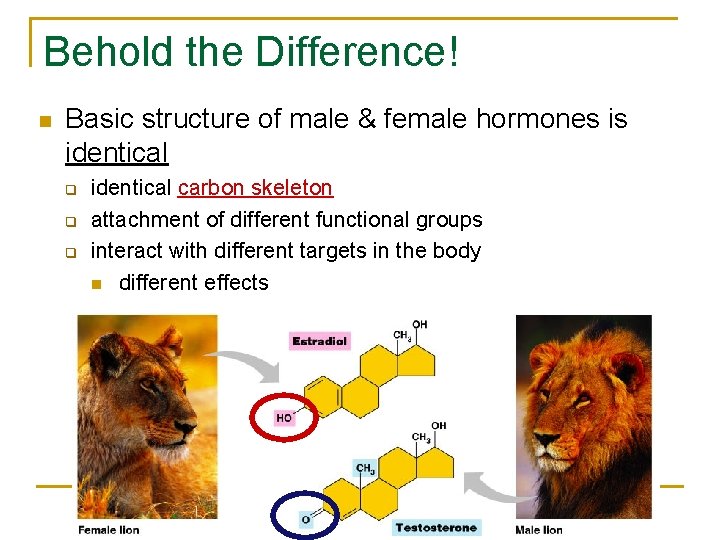
Behold the Difference! Basic structure of male & female hormones is identical q q q identical carbon skeleton attachment of different functional groups interact with different targets in the body different effects

Hydroxyl –OH q q organic compounds with OH = alcohols names typically end in -ol Ethanol Isopropanol

Carbonyl C=O q O double bonded to C if C=O at end molecule = aldehyde if C=O in middle of molecule = ketone

Carboxyl –COOH q C double bonded to O & single bonded to OH group compounds with COOH = acids q fatty acids q amino acids
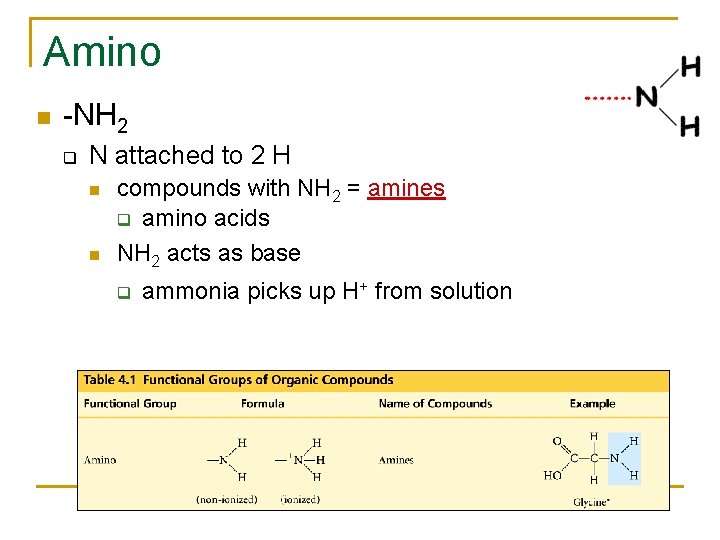
Amino -NH 2 q N attached to 2 H compounds with NH 2 = amines q amino acids NH 2 acts as base q ammonia picks up H+ from solution
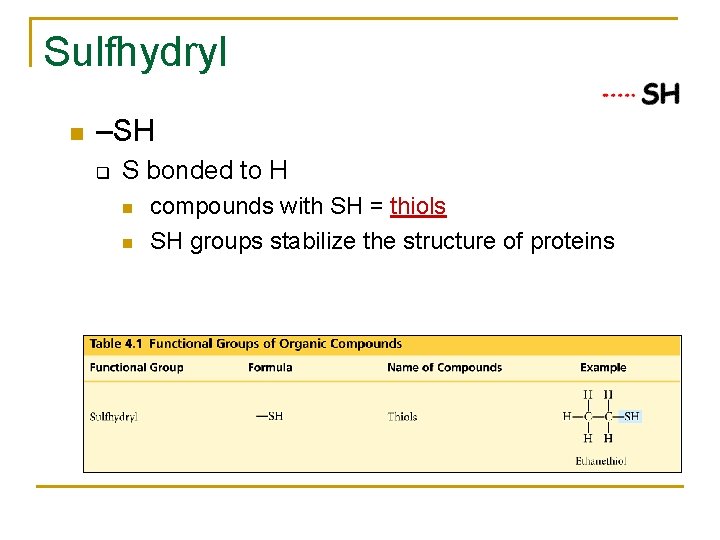
Sulfhydryl –SH q S bonded to H compounds with SH = thiols SH groups stabilize the structure of proteins
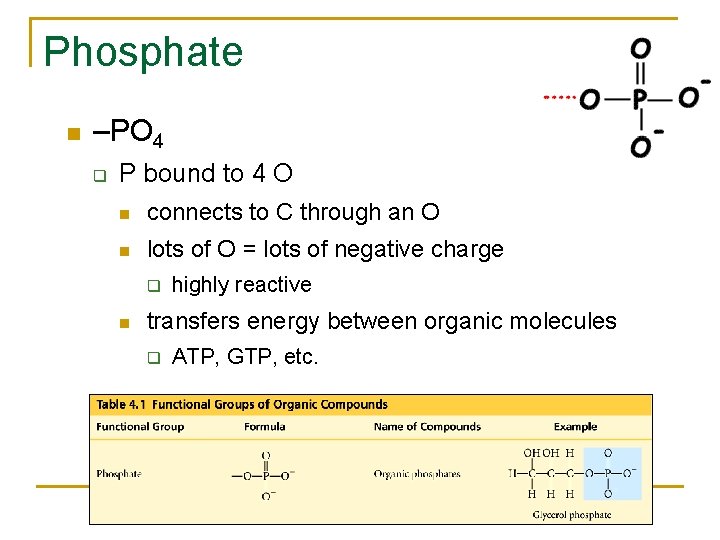
Phosphate –PO 4 q P bound to 4 O connects to C through an O lots of O = lots of negative charge q highly reactive transfers energy between organic molecules q ATP, GTP, etc.
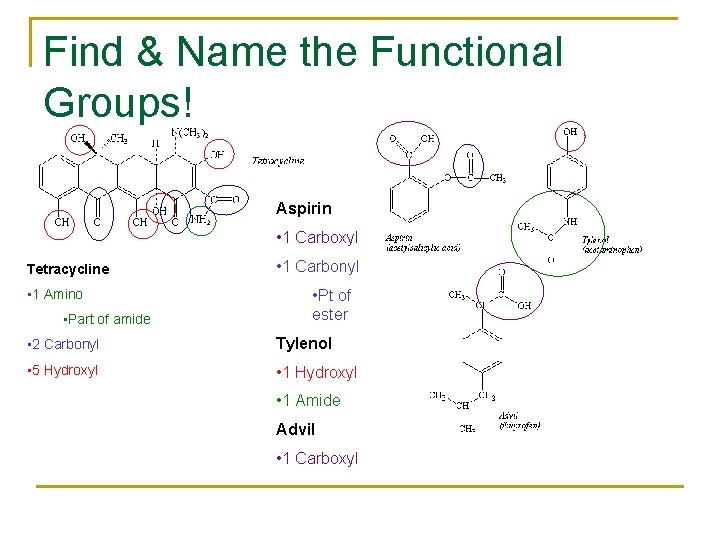
Find & Name the Functional Groups! Aspirin • 1 Carboxyl Tetracycline • 1 Amino • Part of amide • 1 Carbonyl • Pt of ester • 2 Carbonyl Tylenol • 5 Hydroxyl • 1 Hydroxyl • 1 Amide Advil • 1 Carboxyl

Macromolecules Building Blocks of Life 2007 -2008

Macromolecules Smaller organic molecules join together to form larger molecules q macromolecules 4 major classes of macromolecules: q q carbohydrates lipids proteins nucleic acids
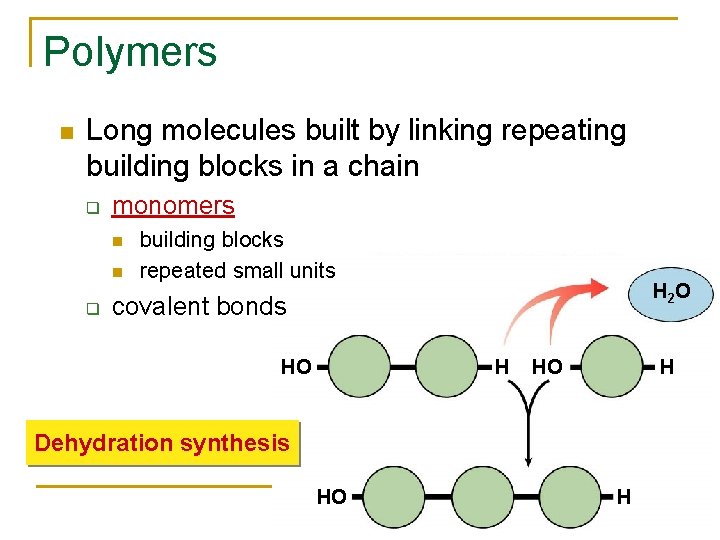
Polymers Long molecules built by linking repeating building blocks in a chain q monomers q building blocks repeated small units H 2 O covalent bonds HO H Dehydration synthesis HO H
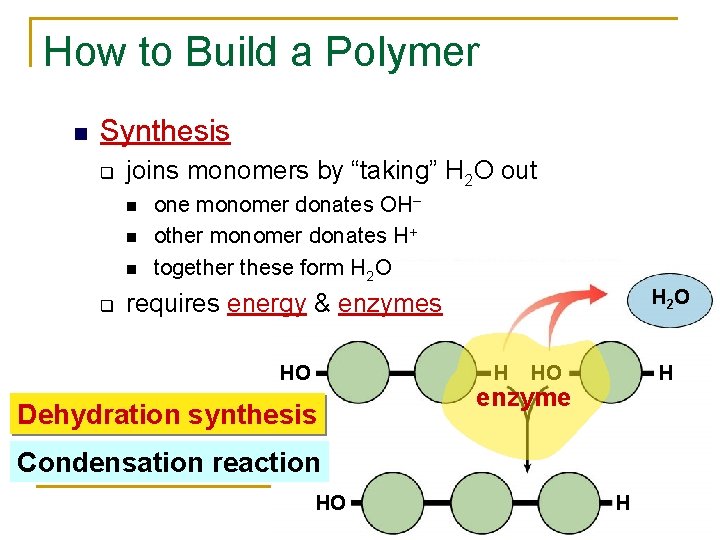
How to Build a Polymer Synthesis q joins monomers by “taking” H 2 O out q one monomer donates OH– other monomer donates H+ together these form H 2 O H 2 O requires energy & enzymes HO H Dehydration synthesis HO H enzyme Condensation reaction HO H
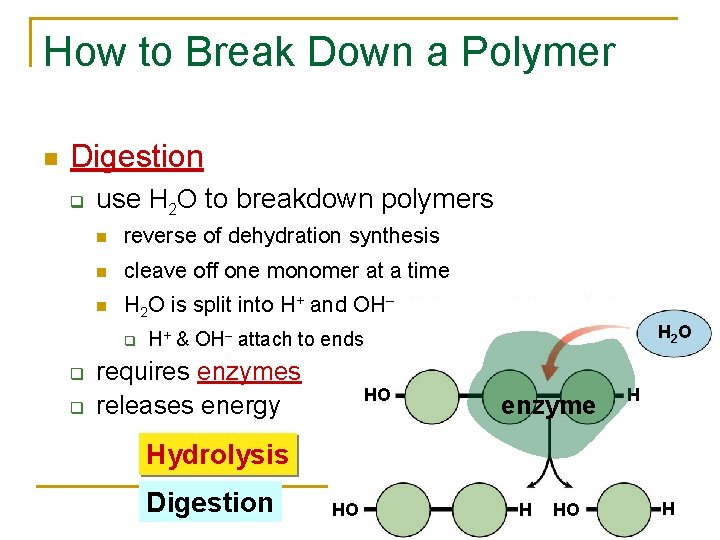
How to Break Down a Polymer Digestion q use H 2 O to breakdown polymers reverse of dehydration synthesis cleave off one monomer at a time H 2 O is split into H+ and OH– q q q H 2 O H+ & OH– attach to ends requires enzymes releases energy HO enzyme H Hydrolysis Digestion HO H
- Slides: 24