Introduction to Nuclear Chemistry The Nucleus Remember that

Introduction to Nuclear Chemistry

The Nucleus • Remember that the nucleus is comprised of the two nucleons, protons and neutrons. • The number of protons is the atomic number. • The number of protons and neutrons together is effectively the mass of the atom. © 2009, Prentice-Hall, Inc.

Radioactive Decay & Decay Products • What makes a nucleus unstable and why do some isotopes undergo spontaneous decay, emitting nuclear decay products? • Nuclear chemistry addresses the transformations and reactions that affect the nucleus of an atom, resulting in the change in identity of the element. • Types of radioactive decay: – Alpha decay – Beta decay – Positron emission – Electron capture – Gamma rays

Natural Radioactivity • -particles can be stopped by paper. • -particles require at least a cm of lead (Pb). • -particles require at least 10 cm of lead (Pb). Energy: > >
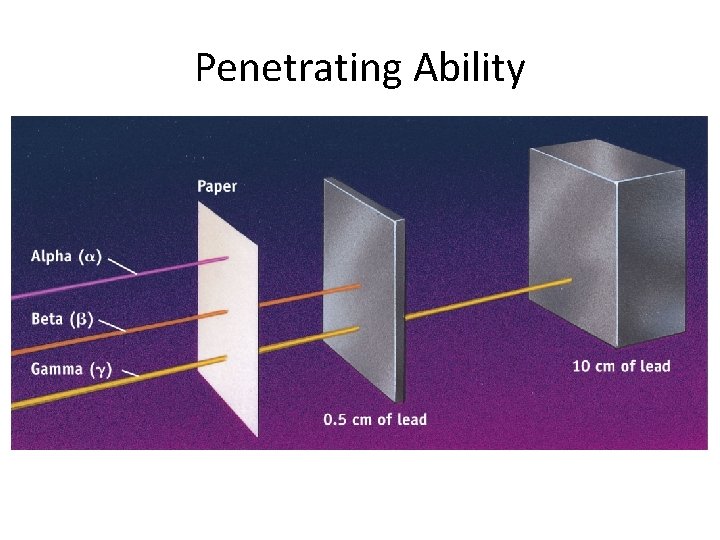
Penetrating Ability
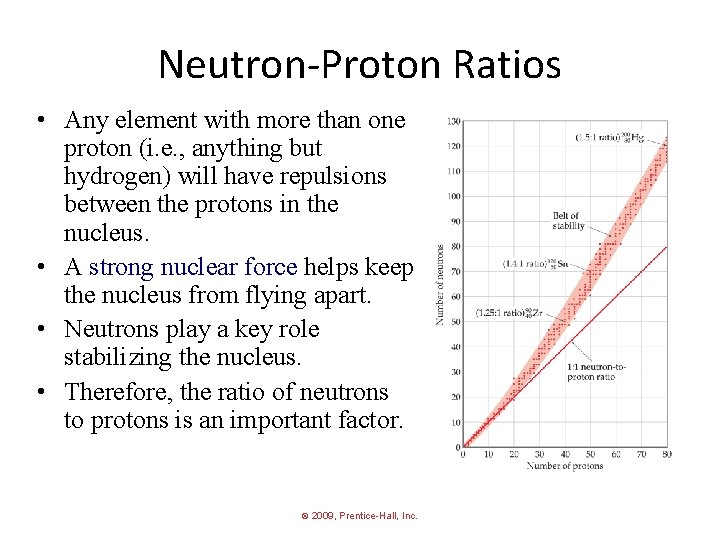
Neutron-Proton Ratios • Any element with more than one proton (i. e. , anything but hydrogen) will have repulsions between the protons in the nucleus. • A strong nuclear force helps keep the nucleus from flying apart. • Neutrons play a key role stabilizing the nucleus. • Therefore, the ratio of neutrons to protons is an important factor. © 2009, Prentice-Hall, Inc.
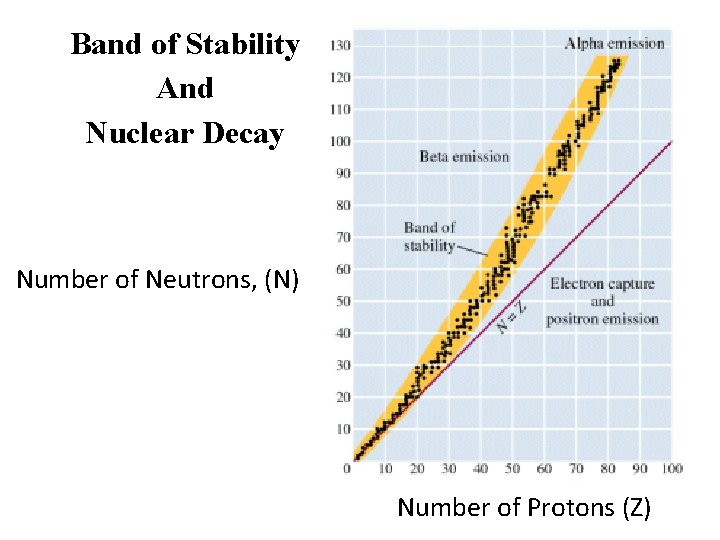
Band of Stability And Nuclear Decay Number of Neutrons, (N) Number of Protons (Z)
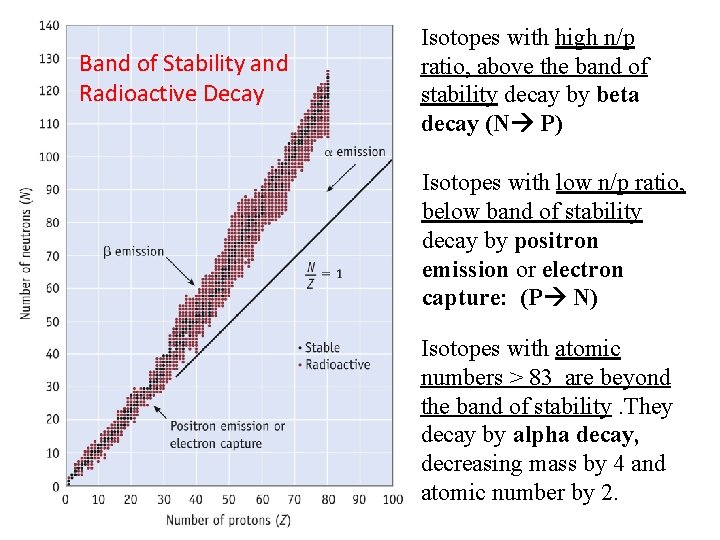
Band of Stability and Radioactive Decay Isotopes with high n/p ratio, above the band of stability decay by beta decay (N P) Isotopes with low n/p ratio, below band of stability decay by positron emission or electron capture: (P N) Isotopes with atomic numbers > 83 are beyond the band of stability. They decay by alpha decay, decreasing mass by 4 and atomic number by 2.
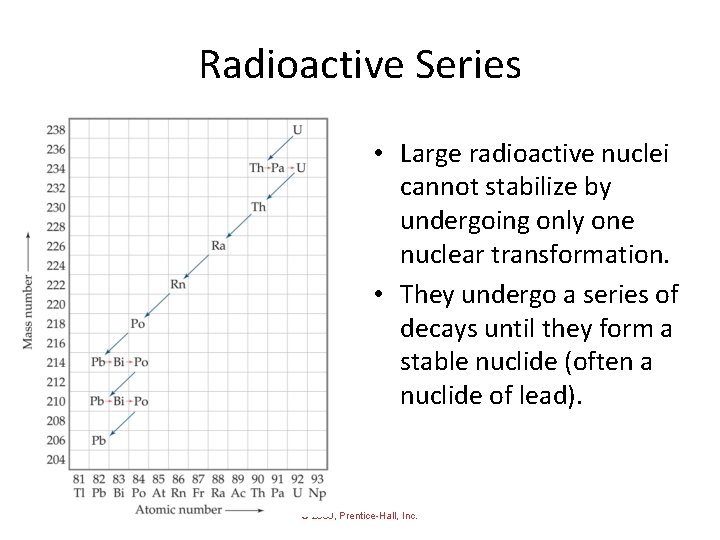
Radioactive Series • Large radioactive nuclei cannot stabilize by undergoing only one nuclear transformation. • They undergo a series of decays until they form a stable nuclide (often a nuclide of lead). © 2009, Prentice-Hall, Inc.

Half-Life • The Half-Life of an isotope is the time it takes for 1/2 a sample to decay from its initial amount. • The rate of a nuclear transformation depends only on the “reactant” concentration. • The decay and half-life for a nuclear reaction follows first order kinetics.
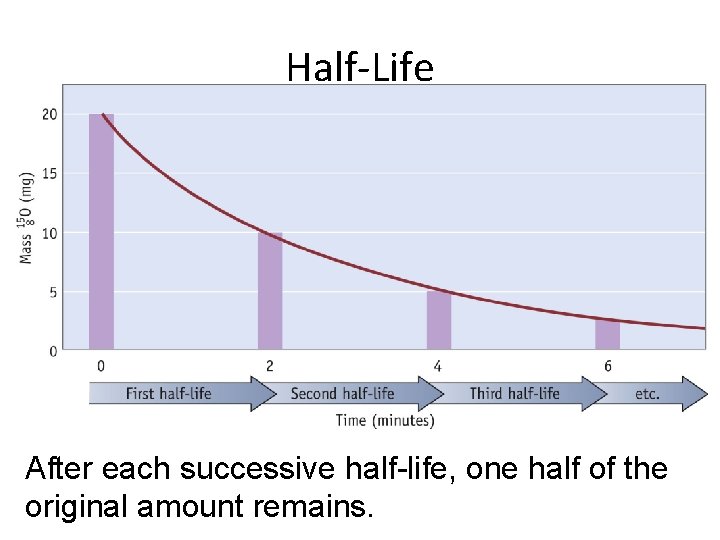
Half-Life After each successive half-life, one half of the original amount remains.

Kinetics of Radioactive Decay follows first order kinetics: The half-life of radioactive decay is t 1/2 = 0. 693/k

Half- Life Calculations • A sample of Radon-222 has an initial α-particle activity (A 0) of 7. 0 x 104 dps (disintegrations per second). • After 6. 6 days, its activity (At) is 2. 1 x 104 dps. • What is the half-life of Radon-222? 1. solve for “k” 2. plug into half- life equation Answer: 3. 8 days

Binding Energy • Binding energy- the energy required to separate a nucleus into individual protons and neutrons. • Mass is converted to energy in the formation of the nucleus based on Einstein’s equitation: E= mc 2 • Binding energy increases with atomic number.
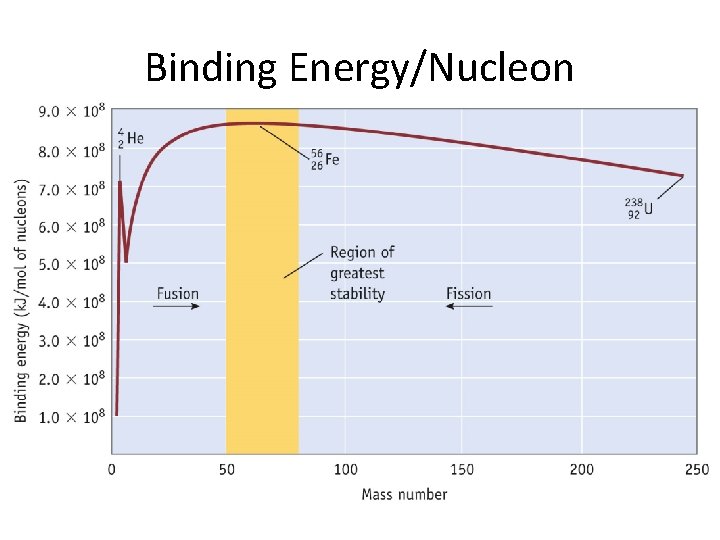
Binding Energy/Nucleon

Fission/Fusion • Discussion of binding energy • Discussion of fission & fusion for nuclear for energy source: https: //www. youtube. com/watch? v=VEVsut Wtvv. Y
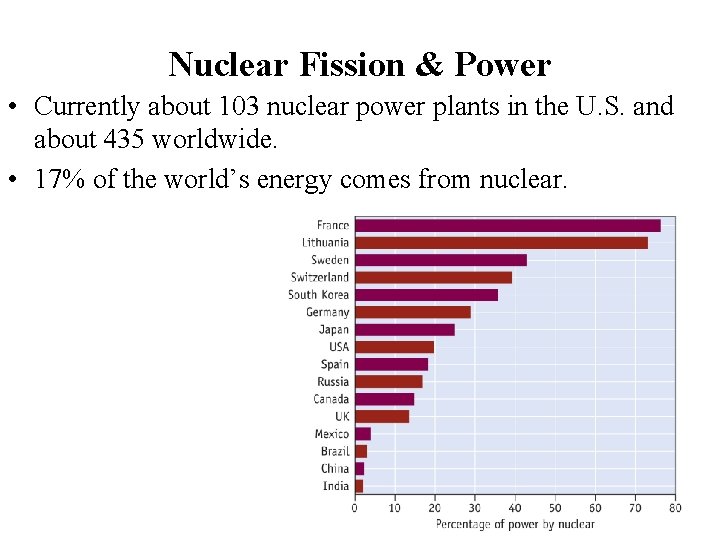
Nuclear Fission & Power • Currently about 103 nuclear power plants in the U. S. and about 435 worldwide. • 17% of the world’s energy comes from nuclear.
- Slides: 17