Introduction to Metabolism Metabolism is the totality of
Introduction to Metabolism

Metabolism is the totality of an organism’s chemical reactions – Manage the materials and energy resources of a cell
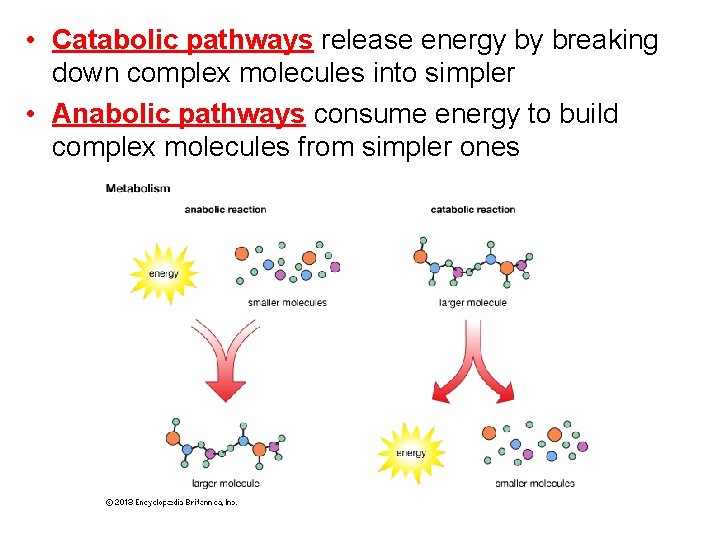
• Catabolic pathways release energy by breaking down complex molecules into simpler • Anabolic pathways consume energy to build complex molecules from simpler ones

Energy = capacity to do work Kinetic energy (KE): energy associated with motion Heat (thermal energy) is KE associated with random movement of atoms or molecules Potential energy (PE): stored energy as a result of its position or structure Chemical energy is PE available for release in a chemical reaction Energy can be converted from one form to another Eg. chemical mechanical electrical


Thermodynamics is the study of energy transformations that occur in nature In an open system, energy and matter can be transferred between the system and its surroundings Organisms are open systems

The First Law of Thermodynamics Ø The energy of the universe is constant Energy can be transferred and transformed Energy cannot be created or destroyed Also called the principle of Conservation of Energy

The Second Law of Thermodynamics ØEvery energy transfer or transformation increases the entropy (disorder) of the universe as a whole During every energy transfer or transformation, some energy is unusable, often lost as heat
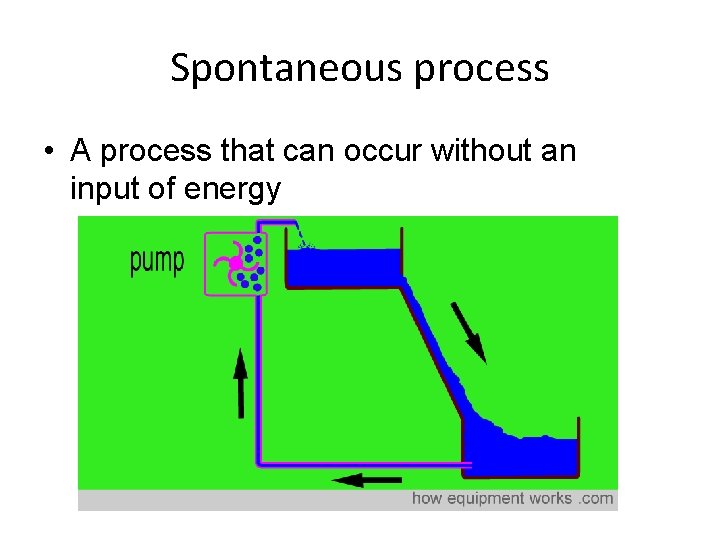
Spontaneous process • A process that can occur without an input of energy

Gibbs Free energy • Free energy: energy part of a system’s energy available to perform work – G = change in free energy • Exergonic reaction: reaction energy is released – Spontaneous reaction – G < 0 • Endergonic reaction: reaction energy is required – Absorb free energy – G > 0
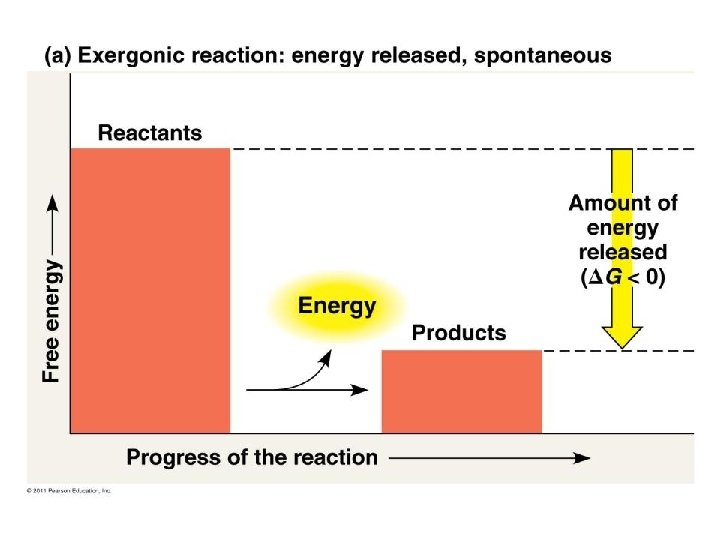
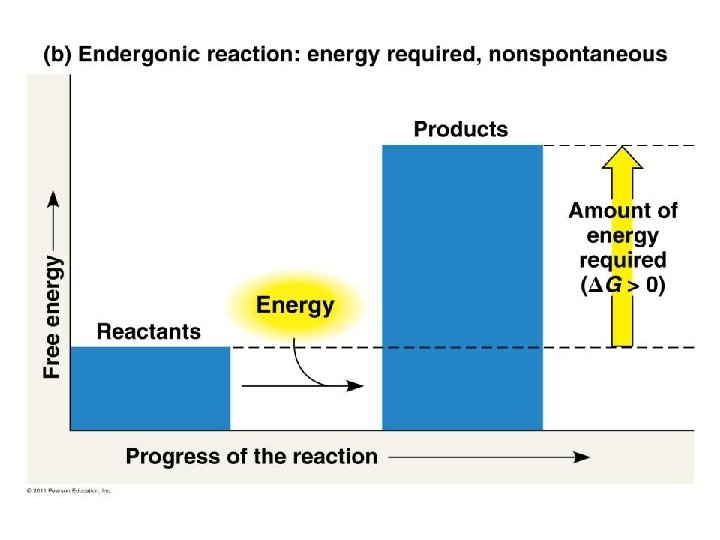

Gibbs Free Energy Gibbs free energy tells us if a reaction can happen spontaneously

Question • If you place teaspoon of sugar in the bottom of a glass of water, it will dissolve completely over time. Left longer, eventually the water will disappear and the sugar crystal will reappear. Explain these observation in terms of entropy.
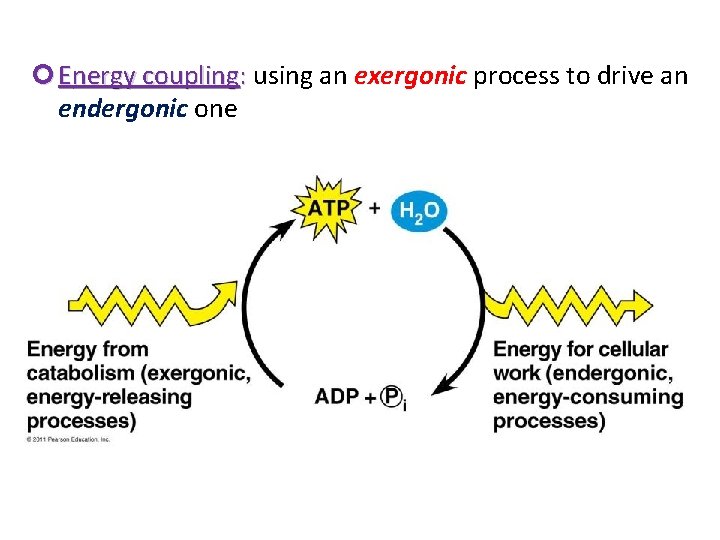
Energy coupling: using an exergonic process to drive an endergonic one
- Slides: 15