INTRODUCTION TO MATTER IS MADE OF ATOMS Atoms














































- Slides: 52

INTRODUCTION TO MATTER IS MADE OF ATOMS.

Atoms • An ______is the smallest basic unit of ________. – Atoms are too small to be seen, but we can see large collections of atoms, such as people. – Atoms are so small that they cannot even be seen with a strong optical microscope.

Molecules • When two or more atoms ______together, they make a particle called a ________. • A molecule can be made of _____numbers and types of atoms. • A molecule is the smallest amount of a _____made of combined atoms that is considered to be that substance.

Atoms are always in motion. • Regardless of the state of matter, atoms are always ______. • Atoms move the most freely in the _____state and the least freely in the _____state.

Atoms in the Solid State Atoms are very close together and vibrating off each other

Atoms in the liquid state are free flowing

Atoms in the Gaseous State are fast moving

INTRODUCTION TO MATTER COMBINES TO FORM DIFFERENT SUBSTANCES.

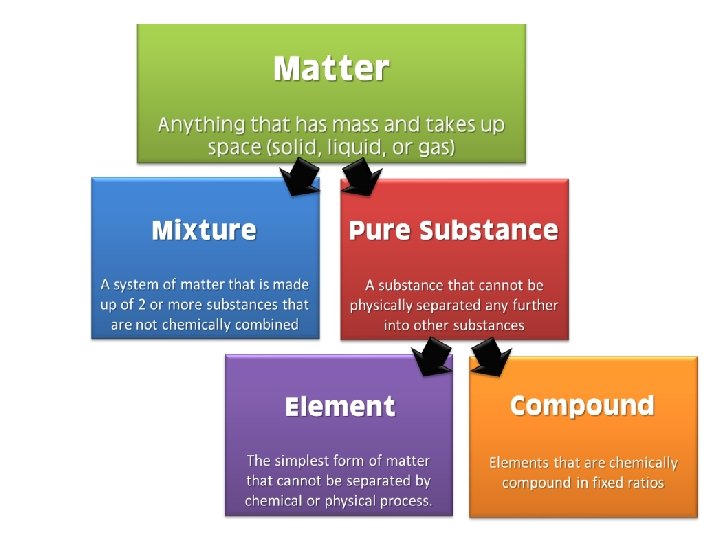
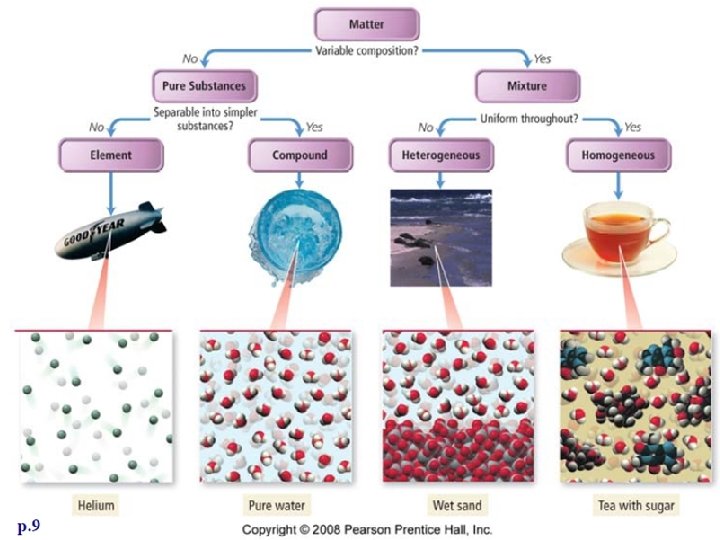
Matter can be pure or mixed. • Most of the substances around you are _______. – Examples: paper, lemonade • A ____substance has only one type of component. A substance is considered pure if it contains only _______type of atom or single combination of atoms.

Matter makes up substances. • _______ are mixtures of a particular type. Elements, compounds, and mixtures are all substances.

Elements • An ______is a substance that contains only a singe type of ____. • The _____of atoms does not matter as long as they are all the same type. • Elements ______be separated into other substances. – Examples: gold, silver, helium, oxygen

Compounds • A ________is a substance that consists of two or more different types of atoms ______together. – Example: Table salt is a compound because it is made of sodium and chlorine.

Mixtures • A _____is a combination of different substances that remain the same individual substances and can be separated by ______means. – Example: A bowl of apples, oranges, and bananas can be separated by physically removing them from the bowl. Pictures from Chem 4 kids

Comparing Mixtures and Compounds Mixtures Compounds


Parts of a Mixture • Homogenous Mixtures – when substances are _______spread throughout a mixture. – Examples: mixing sugar into a cup of water, homogenized milk • Heterogeneous Mixtures – when substances are _______evenly spread throughout a mixture – Examples: soil, salsa

Homogeneous Mixture • “Homo” means the same • has the same uniform appearance and composition throughout; maintain one phase (solid, liquid, gas) • Commonly referred to as solutions • Example: Salt Water Notice the uniform appearance

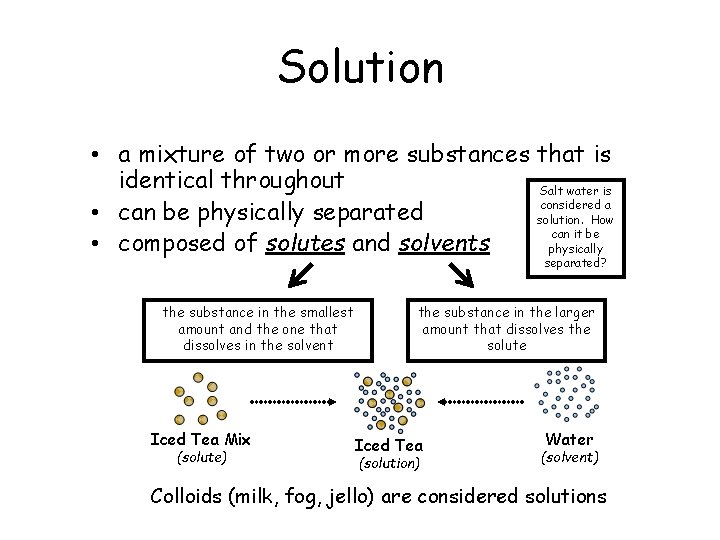
Solution • a mixture of two or more substances that is identical throughout Salt water is considered a • can be physically separated solution. How can it be • composed of solutes and solvents physically separated? the substance in the smallest amount and the one that dissolves in the solvent Iced Tea Mix (solute) the substance in the larger amount that dissolves the solute Iced Tea (solution) Water (solvent) Colloids (milk, fog, jello) are considered solutions

Heterogeneous Mixture • “Hetero” means different • consists of visibly different substances or phases (solid, liquid, gas) • a suspension is a special type of heterogeneous mixture of larger particles that eventually settle • Example: Trail Mix Notice the visibly different substances

Suspensions Properties • Suspensions separate into layers over time • Particles are larger than those in a solutions • Examples: Sand in water, muddy water, salad

Homogenous or Heterogeneous?

Homogenous or Heterogeneous?

Homogenous or Heterogeneous?


Element, Compound, or Mixture? Copper

Element, Compound, or Mixture? Copper

Element, Compound, or Mixture? Rocks

Element, Compound, or Mixture? Rocks

Element, Compound, or Mixture? Jelly Beans

Element, Compound, or Mixture? Jelly Beans

Element, Compound, or Mixture? Table Sugar

Element, Compound, or Mixture? Table Sugar

Element, Compound, or Mixture? Diamond

Element, Compound, or Mixture? Diamond

Element, Compound, or Mixture? Tea

Element, Compound, or Mixture? Tea

Element, Compound, or Mixture? Salt

Element, Compound, or Mixture? Salt

Element, Compound, or Mixture? Neon Gas

Element, Compound, or Mixture? Neon Gas

Element, Compound, or Mixture? Salad

Element, Compound, or Mixture? Salad

Element, Compound, or Mixture? Pure Water

Element, Compound, or Mixture? Pure Water

Element, Compound, or Mixture? Aluminum

Element, Compound, or Mixture? Aluminum

Element, Compound, or Mixture? Lemonade

Element, Compound, or Mixture? Lemonade

Element, Compound, or Mixture? Silver

Element, Compound, or Mixture? Silver

Element, Compound, or Mixture? Sand

Element, Compound, or Mixture? Sand