INTRODUCTION TO MANUFACTURING Ferrous Alloys l u 10110


















- Slides: 23

INTRODUCTION TO MANUFACTURING Ferrous Alloys (l. u. 10/1/10)

Ferrous Metals and Alloys Contain IRON as their base metal. second most abundant element in Earth’s crust (5%). Relatively inexpensive. Relative cost per unit Volume (Kalpakjian & Schmid, 2006) Carbon Steel Copper Nickel Silver Gold 1 5 -6 X 35 X 600 X 60, 000 X Engr 241 1

Three materials used in Steel: 1. Iron ore - pelletized metal 2. Coke - for heat & producing carbon monoxide which reduces iron-oxide to iron (removes oxygen) 3. Limestone - combines with impurities which floats to surface (slag) Engr 241 2

Common Terms Pig Iron- hot metal, molten metal used in making iron and steels Ingot- molten metal to solid form – ready for rolling/forging = Inefficient! Continuous Casting become most popular steelmaking technique Engr 241 3

Three Types of Steel Ingots: 1) Killed Steel- steel is fully deoxidized: oxygen is removed and porosity is eliminated (consistent mechanical and chemical properties) 2) Semi-Killed Steel- partially deoxidized steel: contains some porosity (economical) 3) Rimmed Steel- low carbon content, porosity (blowholes), lower quality steel (requires inspection) Engr 241 4

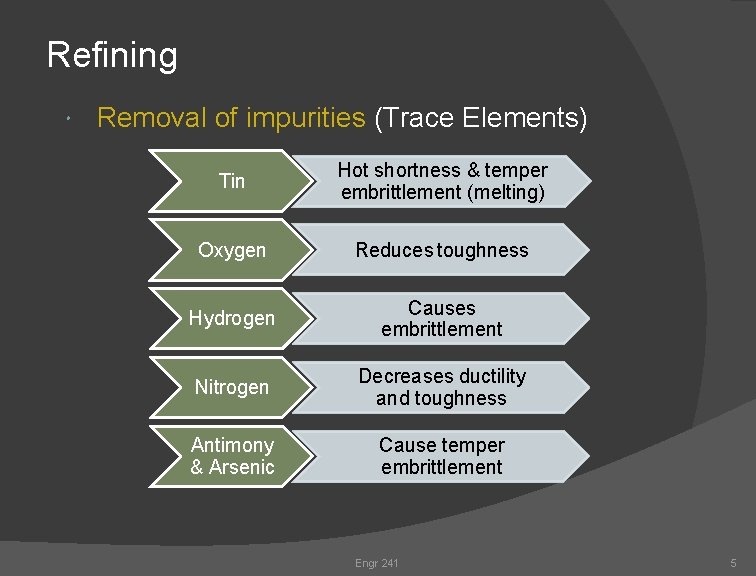
Refining Removal of impurities (Trace Elements) Tin Hot shortness & temper embrittlement (melting) Oxygen Reduces toughness Hydrogen Causes embrittlement Nitrogen Decreases ductility and toughness Antimony & Arsenic Cause temper embrittlement Engr 241 5

Refining: Used to create higher quality steels Improves uniformity and consistency in composition Removing impurities, inclusions, other elements Adding various elements Engr 241 6

Inclusions? Good or bad? “Free-machining steels are basically carbon steels that have been modified by an addition of sulfur, lead, bismuth, selenium, tellurium, or phosphorous plus sulfur to enhance machinability. Sulfur combines with manganese to form soft manganese sulfide inclusions. These, in turn, serve as chipbreaking discontinuities within the structure. The inclusions also provide a build-in lubricant that prevents formation of a build-up edge on the cutting tool and imparts an improved geometry” (Black & Kohser, 2008, p. 130). Engr 241 7

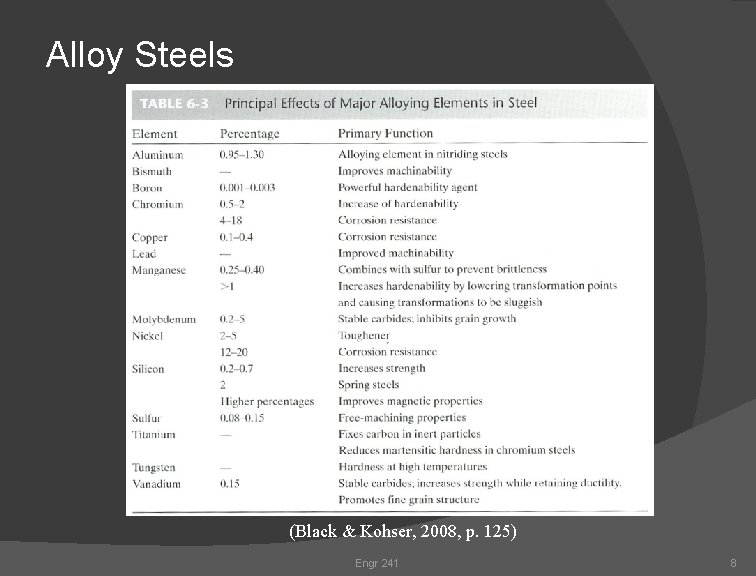
Alloy Steels (Black & Kohser, 2008, p. 125) Engr 241 8

Iron - Carbon Alloys Pure Iron - less than 0. 008% C Steel - up to 2. 11% C Cast Iron - up to 6. 67% C* typically less than 4. 5% C Engr 241 9

Carbon Steels Classification Low-Carbon (mild steel): 0. 30% or less Bolts, nuts, sheet, tubes, plate, low strength machine components Medium-Carbon: 0. 30 -0. 60% Machinery and automotive parts, gears, axles, connecting rods, etc. High-Carbon: more than 0. 60% Cutting tools, cables, springs, cutlery Engr 241 10

Carbon Effects (Black & Kohser, 2008, p. 124) Engr 241 11

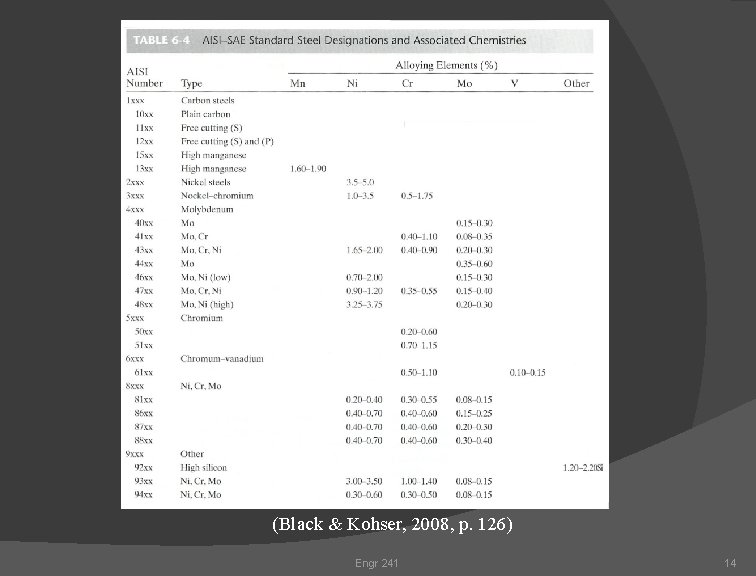
Common Designations for Steel: AISI - The American Iron and Steel Institute SAE - Society of Automotive Engineers ASTM - American Society for Testing Materials Engr 241 12

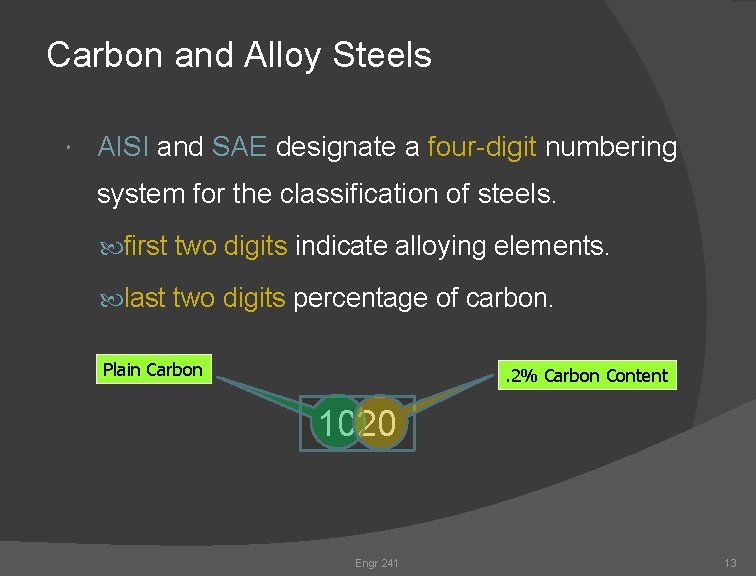
Carbon and Alloy Steels AISI and SAE designate a four-digit numbering system for the classification of steels. first two digits indicate alloying elements. last two digits percentage of carbon. Plain Carbon . 2% Carbon Content 1020 Engr 241 13

(Black & Kohser, 2008, p. 126) Engr 241 14

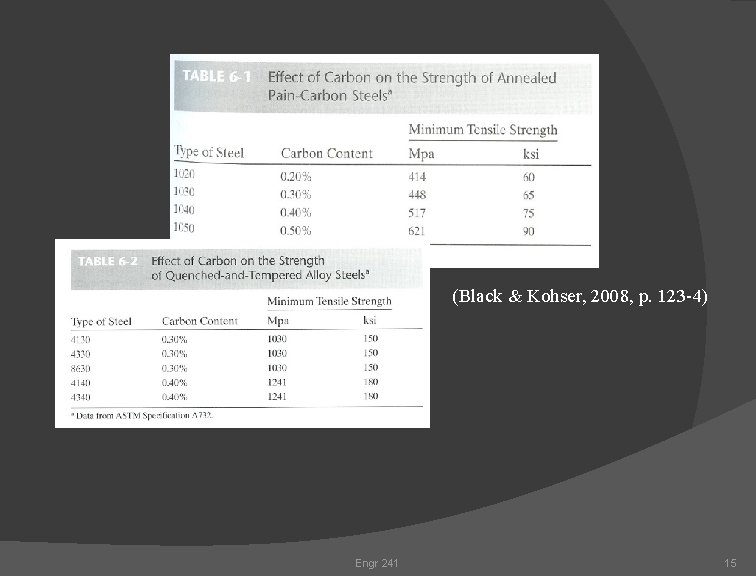
(Black & Kohser, 2008, p. 123 -4) Engr 241 15

Sample Question What makes up a “forty-three forty” 4340 steel? 43 = Mo, Cr, Ni 40 =. 40% C Engr 241 16

Sample Question Why is aircraft landing gear made of 4140 or 4340 steel? Mo = imparts temperature strength, toughness, hardness, dimensional stability C = imparts hardness, wear resistance, reduces ductility Ni/Cr = hardness and oxidation resistance Engr 241 17

Stainless Steels Characterized by their corrosion resistance, high strength and ductility, and high chromium content. In the presence of air (oxygen) they develop a thin and hard adherent film of chromium oxide which protects the metal from corrosion Passivation Engr 241 18

(Black & Kohser, 2008, p. 132) (Black & Kohser, 2008, p. 134) Engr 241 19

Stainless Steels Classifications Austenitic (200 -300 series) Ferritic (400 series) Kitchenware, fittings, welded construction, • Nonmagnetic resistant • Most. Heat/chemical ductile of all stainless steels environments • Susceptible to stress-corrosion cracking Non-structural applications, • High chromium content automotive trim, kitchenware • Lower ductility • Not heat-treatable (400 and 500 series) • Magnetic. Cutlery, surgical tools, springs, • Moderate corrosion resistance. • High strength, hardness, fatigue resistance, good ductility Precipitation Hardening (PH) Aircraft aerospace applications • Chromium and nickel (along&with copper, aluminum, titanium, or molybdenum. • Good corrosion resistance, ductility, and high strength at elevated temperatures Martensitic valves Heat exchangers Duplex Structure • Austenite and ferrite. • Higher resistance to corrosion and stress corrosion than 300 series Engr 241 20

Tool and Die Steels Special alloys designed for high strength, impact toughness, and wear resistance at room or elevated temperatures. For forming and machining of metals High-Speed Steels ○ maintain strengths at elevated temperatures. ○ molybdenum (95% of all HSS) and tungsten series. Engr 241 21

Tool and Die Steels (Cont. ) – (See text table) Hot Work Steels ○ designed for use at elevated temperatures ○ high toughness, resistance to wear and cracking. Cold Work Steels ○ designed for cold working operations. Shock-resistant ○ designed for impact toughness. ○ For dies, punches, chisels Engr 241 22