INTRODUCTION TO MAMMALIAN CELL CULTURE Lecturer Assist Prof


























- Slides: 29

INTRODUCTION TO MAMMALIAN CELL CULTURE Lecturer: Assist. Prof. Dr. Mirza Suljagić Assistant: Jasmin Sutkovic March, 16 th 2018

Chapter 1 – Introduction Textbook: „Introduction to Mammalian Cell Culture“ (D. Ler, U. Glamočlija, M. Suljagić)

Introduction • Cell Culture – removal of cells from an organism and their subsequent growth in a favorable, artificially controlled environment • Primary Cell Culture – removal from tissue • Can be sub-cultured • Cell line – due to proliferation • Finite – limited life span • Immortal – due to transformation (Continous cell line)

Primary Cell Culture • Contains heterogenous cell population • Sub-culturing leads to cell line generation • Different purification methods • Tissue sample: Dissection, Rinsing, Mechanical Dissagregation (Enz. Digestion) in trypsin • Different separation techniques: • Density gradient separation • MACS • FACS Figure – Cell line

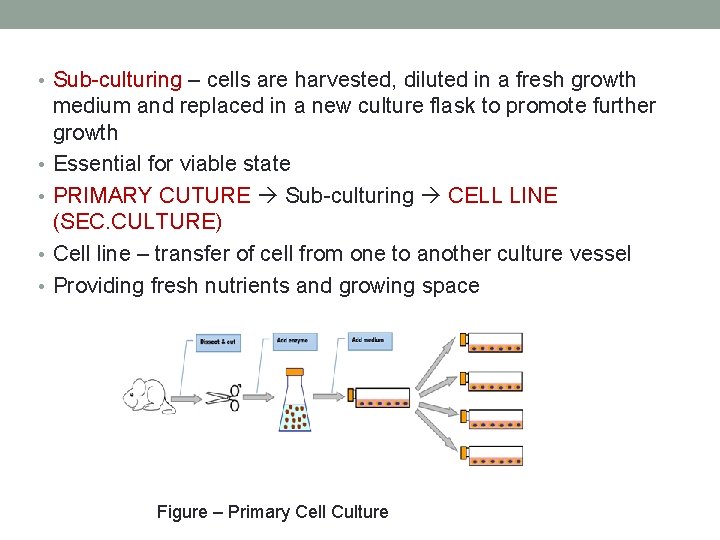
• Sub-culturing – cells are harvested, diluted in a fresh growth • • medium and replaced in a new culture flask to promote further growth Essential for viable state PRIMARY CUTURE Sub-culturing CELL LINE (SEC. CULTURE) Cell line – transfer of cell from one to another culture vessel Providing fresh nutrients and growing space Figure – Primary Cell Culture

Types of cell lines (on the basis of life span of culture): 1. Finite cell lines - limited life span - limited number of cell generations (40 -60 doublings) - slow growth rate - doubling time 24 -96 h 2. Continuous cell lines - transformed under laboratory conditions - grow in a monolayer or in suspension - rapid growth rate - doubling time 12 -24 h • Cell strain – positive selection of cells either from primary culture or cell line by cell cloning • Limited division potential, lose ability to proliferate (Hayflick limit)

CHAPTER 2 BASICS OF CELL CULTURE

Cell Culture Requirements and Equipment • Differ depending on a cell culture (e. g. Cancer & GMO cells) • Microbiologically free • Equipment common to most cell culture labs: 1. Essential Equipment - cell culture hood, humid CO 2 incubator, centrifuge, refrigerator (2 -8 C) and freezer (-20 C) 2. Automated Cell Counter - inverted microscope, pipettor, micropipettes, cultureware 3. Outside culture environment - liquid nitrogen container, sterilizer (autoclave), waterbath

Work Surfaces • Bench tops, wallsm flooring – smooth and resistant to temperature and cracking (for liquid nitorgen) • Major requirement – to maintain aseptic work area • Use cell culture hood (for aseptic conditions)

Basic Equipment 1. Clean Benches – horizontal or vertical laminar flow - provide product protection - dust-free assembly of sterile equipment - never used for handling of cell cultures 2. Cell Culture Hood – laminar flow cabinet or tissue culture hood - HEPA filter

Figure – Laminar Flow Hood Figure – Cell Culture Hood

Types of Cell Culture Hood • Class I – significant protection levels to lab personnel - no culture protection from contamination • Class II – designed for work involving BSL-1, 2, 3 materials - provide aseptic environment - for handling potentially hazardous materials (viruses, toxic/carcinogenic reagents, etc. ) • Class III – highest level of protection - working with human pathogens and BSL-4 material

3. Incubators – for growth conditions (temperature, CO 2, humidity) • Mammalian cell cultures – at 37 C, 5 -10% CO 2 • Large, forced air circulation, have temperature control ± 0. 2 C, made of stainless steel • Mammalian cells – kept at humid CO 2 incubators • Humid CO 2 incubators – incubation of cells in sterile flasks • Dry incubators – water dish controles humidity

4. Centrifuge – for maintanance of most cell lines and cyropreservation • Balance the tubes • Centrifuge rate – 80 -150 xg • Higher forces may promote cell damage • Tubes 15 -50 ml Storage • Storage areas for liquids (media, antibiotics) and consumables ( disposable pipettes, gloves, media bottles) • Follow the instructions

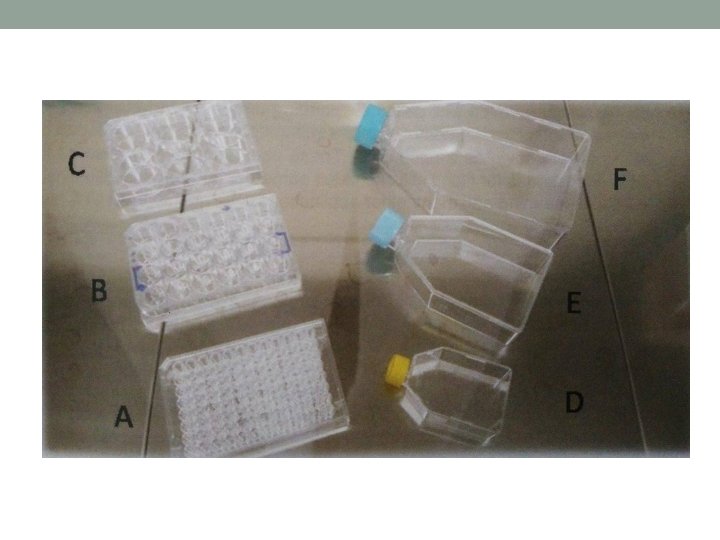
5. Refrigerators and Freezers - for small cell culture lab (domestic refrigerators) • Storing at -5 to -20 C • Ultra-deep freezer - -80 C storing 6. Plastic-ware for Cell Culture • Includes Petri-dishes, multi-well plates (6, 12, 24, 48, 96 wells per plate) • Screwcap flasks – classified acc. to surface areas (T-25, T -75, T-175, T-225 cm 2) • Provide hydrophilic surface to facilitate attachment of anchorage dependent cells


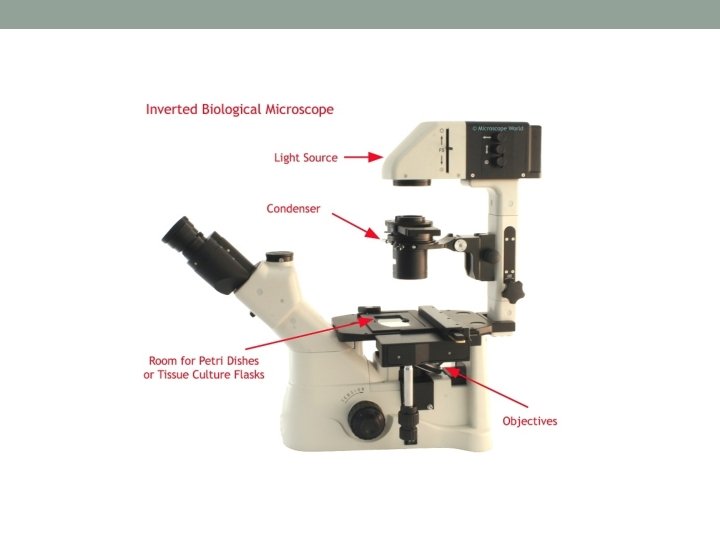
7. Inverted Microscope – used to visualize cells in culture • Easy to operate • Useful for observing living cells or organisms at the bottom of a dish • Provide information about cell morphology and cell state • Cell culture can be viewed in flasks, dishes, multi-well plates • Liquid Nitrogen – cellular maintenance for prolonged period


Biosafety • Main aim – prevent injury, protect property, avoid harm to individuals and environment • Common hazards: accidental punctures with syringe needles, spills, splashes onto skin and mucous membrane, inhalation, ingestion • Strict adherence to standard microbiological practices and techniques • Type of risks: a) Low risk – non-human cont. cell lines (well characterized) b) Medium risk – poorly characterized cell lines (mammalian) c) High risk – primary cells derived from human/primate tissue

Biohazards • Biological substances that represent a threat to human health • Medical waste, toxins, viruses, m. o. • Most common biohazards presented by cell culture – human pathogenic viruses • Cell products, biomolecules with modulations Figure – Symbol of biohazard materials

Biosafety levels • „Biosafety in Microbiological and Biomedical Laboratories“ by NIH • Describe safety equipment, microbiological practices 1. BSL-1 • Basic level of protection • Common to most labs • For agents that are not known to cause disease to humans (Bacillus subtilis, Canine hepatitis, E. coli)

2. BSL-2 • For moderate-risk agents • Cause mild human disease (by ingestion) • Work with lentiviral vectors (Zika, Hepatitis A, B, C, HIV, Salmonella) 3. BSL-3 • For agents with known potential for aerosol transmission • Agents may cause lethal infection (for which vaccines exist) • West Nile Virus, Typhus, TBC, Plasmodium falciparum

4. BSL-4 • For agents that pose high individual risk of life threatening disease • No treatment • Involves special equipment • Contains UV light, showers, vacuum room, showers, autonomous detection system

Disinfection and Waste Disposal • To minimize risk of contamination of cell culture • Personal protective equipment should be used • Main disinfectant: a) Hypochlorite (Sodium hypochlorite) - for general purpose - cannot be used on metal surfaces - should be made fresh daily - active against viruses b) Alcohol (Ethanol, Isopropanol) - effective against bacteria, viruses (only ethanol)

c) Aldehyde (Formaldehyde) - irritants, use should be limited due to sensitization problems - Used only in well ventilated areas • Different waste forms (different treatments) • Tissue culture waste • Contaminated pipettes • Solid waste

Aseptic Manipulations • Keep the cells free from m. o. contamination • Wear basic protective equipment • Aseptic manipulation – barrier between m. o and environment • Elements of aseptic technique a) Sterile Work Area - use gloves, lab coat, cell culture hood - disinfect surrounding areas before and after use - Bunsen burner – not recomended

b) Personal Hygiene - Wash hands to remove adherent m. o that would contaminate cell cultures - Keep places clean - Avoid talking, sneezing, coughing - One pipette – cannot be used between two different bottles of media

Cell Culture Contamination • Contaminants: a) Chemical – media, sera, detergents, water, endotoxin impurities b) Biological – bacteria, yeast, viruses, etc. • Bacteria – contamination is easily detected (p. H drop, cloudly appearance, thin films on a surface) • Yeast – little p. H change until the contamination is heavy - appear as individual ovoid or spherical particles • Mycoplasmas – Difficult to detect until they achieve extremely high densities - decrease proliferation rate, not cell death - fluorescent staining

• Molds – grow as multicellular filaments - appear as thin filaments or dense clumps of spores • Viruses – take over cell machinery to reproduce - hard to detect - detected by ELISA, PCR, electron microscopy