Introduction to Equilibrium Equilibrium Chemical equilibrium occurs when

Introduction to Equilibrium
![Equilibrium Chemical equilibrium – occurs when the [reactants] & [products] stop changing and the Equilibrium Chemical equilibrium – occurs when the [reactants] & [products] stop changing and the](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-2.jpg)
Equilibrium Chemical equilibrium – occurs when the [reactants] & [products] stop changing and the rates of the forward and reverse reactions are equal. Equilibrium by ↔ reactions are always denoted
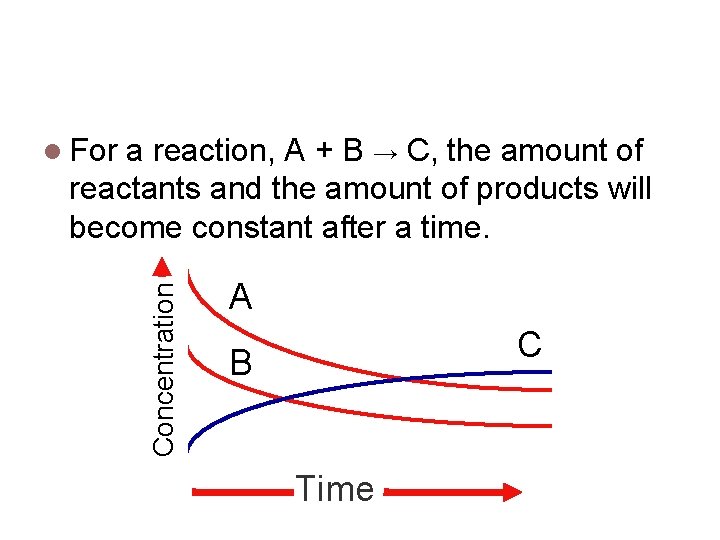
A matter of time. a reaction, A + B → C, the amount of reactants and the amount of products will become constant after a time. Concentration For A C B Time

Reversible Reactions A reaction goes to completion if it converts all reactants to products. But most reactions don’t go to completion, they appear to stop before all of the reactants are used up. In these reactions some of the product returns to reactants, reversible reactions.
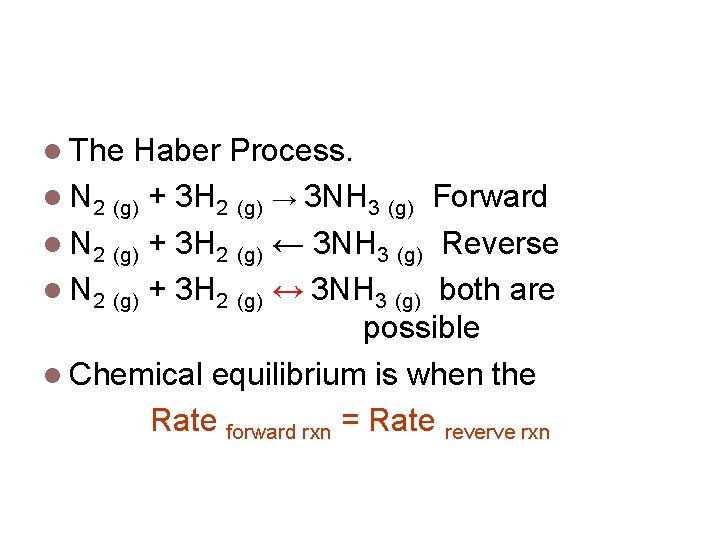
A famous reversible reaction. The Haber Process. N 2 (g) + 3 H 2 (g) → 3 NH 3 (g) Forward N 2 (g) + 3 H 2 (g) ← 3 NH 3 (g) Reverse N 2 (g) + 3 H 2 (g) ↔ 3 NH 3 (g) both are possible Chemical equilibrium is when the Rate forward rxn = Rate reverve rxn

Law of Chemical Equilibrium Gulberg and Waage proposed that at a given set of conditions the ratio of reactant and product concentrations is constant. For the reaction a. A + b. B ↔ c. C + d. D Keq = [C]c [D]d [A]a [B]b The equlibrium constant Keq is the ratio for the reaction at a temperature.

Homogeneous vs Heterogeneous Homogeneous-all reactants and products in same phase. Use equation on previous slide. When all of the reactants aren’t homogeneous you must evaluate the reaction. Pure liquids and solids don’t change concentrations, so ignore them. This is the heterogeneous equilibrium.
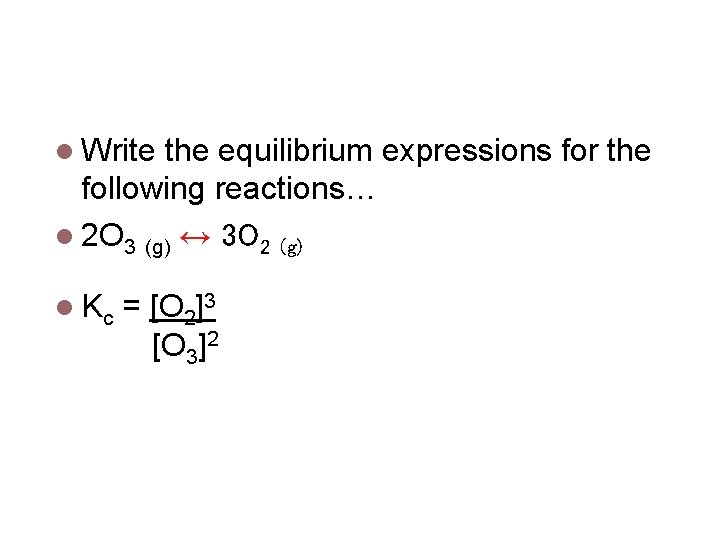
Equilibrium Expressions Write the equilibrium expressions for the following reactions… 2 O 3 (g) ↔ 3 O 2 (g) Kc = [O 2]3 [O 3]2

Equilibrium Expressions Write the equilibrium expressions for the following reactions… H 2 (g) + I 2 (g) ↔ 2 HI (g) Kc = [HI]2 [H 2][I 2]

Equilibrium Expressions Write the equilibrium expressions for the following reactions… Fe. O (s) + H 2 (g) ↔ Fe (s) + H 2 O (g) Kc = [H 2 O ] [H 2]

Look at the last 3 reactions Tell if they are heterogeneous or homogeneous 2 O 3 (g) ↔ 3 O 2 (g) Homogeneous H 2 (g) + I 2 (g) ↔ 2 HI (g) Homogeneous Fe. O (s) + H 2 (g) ↔ Fe (s) + H 2 O (g) Heterogeneous

Determining Keq In order to calculate Keq you must be given the concentrations of the products and the reactants. These come from experiments or are given. Plug the numbers into your Keq equation and solve.

Equilibrium Constant in Terms of Pressure When the reactants & products are gases the keq will be in partial pressures not molarity Kp when using pressure a. A + b. B ↔ c. C + d. D Kc = [PC]c[PD]d [PA]a[PB]b

Switching between Kc & Kp = Kc(RT) n R = 0. 0821 T = temperature in Kelvin n = change in moles (# moles products - # moles reactants) Kp

Switching between kc & kp N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) Calculate Kp at 300°C if Kc Kp = Kc(RT) n Kp = 9. 60 (0. 0821 x 573) -2 4. 34 x 10 -3 = 9. 60

Magnitude of Keq Will either be big or small Value of Kc will determine if the products or reactants are favored CO + Cl 2 ↔ COCl 2 Kc = [COCl 2] = 4. 57 x 109 [CO][Cl 2]
![Magnitude of Keq Kc is greater that 1 Therefore the [products] is greater than Magnitude of Keq Kc is greater that 1 Therefore the [products] is greater than](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-17.jpg)
Magnitude of Keq Kc is greater that 1 Therefore the [products] is greater than the [reactants] So products are favored Kc 1 reactants are favored
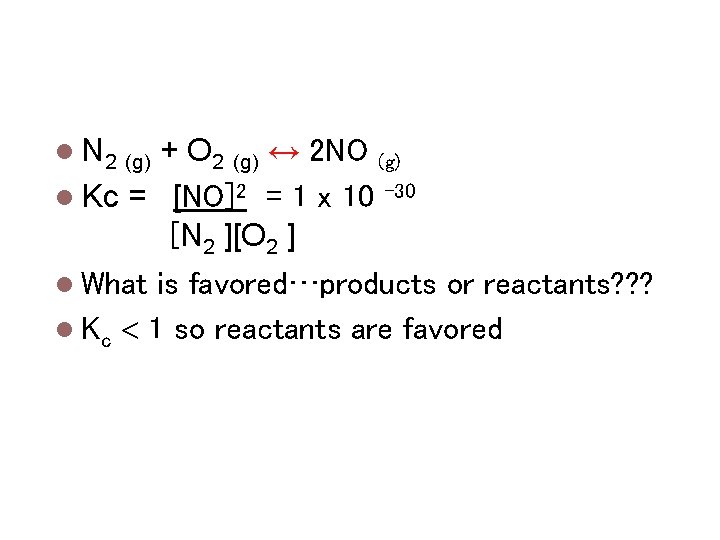
Magnitude of Keq N 2 (g) Kc = What + O 2 (g) ↔ 2 NO (g) [NO]2 = 1 x 10 -30 [N 2 ][O 2 ] is favored…products or reactants? ? ? Kc 1 so reactants are favored

Direction of Equilibrium & k Equilibrium reactions occur in both directions You could have… N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) Kc = [NH 3]2 / [N 2][H 2]3 Or 2 NH 3 (g) ↔ N 2 (g) + 3 H 2 (g) Kc = [N 2][H 2]3 / [NH 3]2

Direction of Equilibrium & k The equilibrium constant in one direction is the reciprocal of the one in the reverse reaction N 2 O 4 ↔ 2 NO 2 kc = 0. 212 What is the kc of … 2 NO 2 ↔ N 2 O 4 Kc = 1/0. 212 = 4. 72
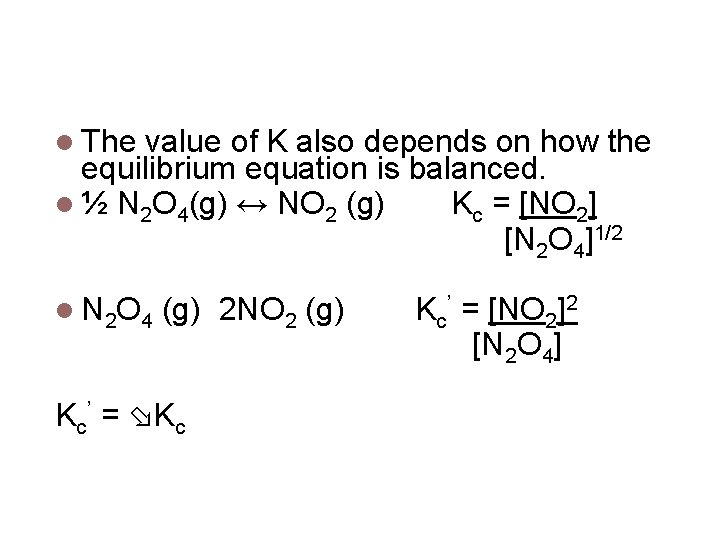
Relationship between coefficients and K The value of K also depends on how the equilibrium equation is balanced. ½ N 2 O 4(g) ↔ NO 2 (g) Kc = [NO 2] [N 2 O 4]1/2 N 2 O 4 (g) 2 NO 2 (g) Kc’ = Kc Kc’ = [NO 2]2 [N 2 O 4]

Calculating Equilibrium Constants A mixture of N 2 gas and H 2 gas produce NH 3 gas and are allowed to come to equilibrium at 472°C. The equilibrium mixture was analyzed and found to contain 0. 1207 M H 2, 0. 0402 M N 2, & 0. 00272 M NH 3. Calculate Kc.
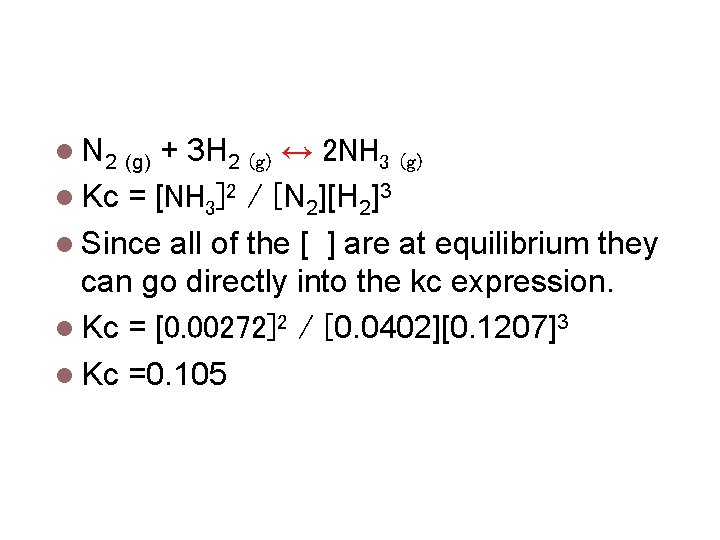
Calculating Equilibrium Constants N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) Kc = [NH 3]2 / [N 2][H 2]3 Since all of the [ ] are at equilibrium they can go directly into the kc expression. Kc = [0. 00272]2 / [0. 0402][0. 1207]3 Kc =0. 105

Calculating Equilibrium Constants 2 NO 2 Cl ↔ 2 NO 2 + Cl 2 At equilibrium the [NO 2 Cl] = 0. 00106 M [NO 2] = 0. 0108 M [Cl 2] = 0. 00538 M Calculate kc
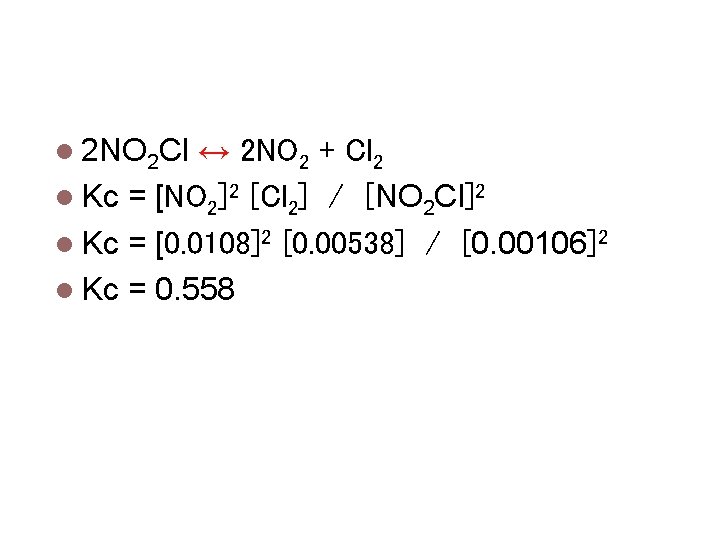
Calculating Equilibrium Constants 2 NO 2 Cl ↔ 2 NO 2 + Cl 2 Kc = [NO 2]2 [Cl 2] / [NO 2 Cl]2 Kc = [0. 0108]2 [0. 00538] / [0. 00106]2 Kc = 0. 558
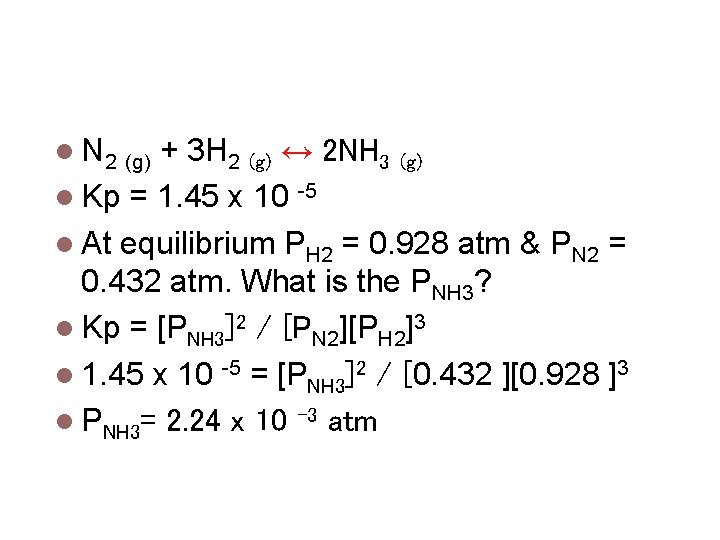
Calculating Equilibrium Constants N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) Kp = 1. 45 x 10 -5 At equilibrium PH 2 = 0. 928 atm & PN 2 = 0. 432 atm. What is the PNH 3? Kp = [PNH 3]2 / [PN 2][PH 2]3 1. 45 x 10 -5 = [PNH 3]2 / [0. 432 ][0. 928 ]3 PNH 3= 2. 24 x 10 -3 atm

Calculating Equilibrium Constants PCl 5 (g) ↔ PCl 3 (g) + At equilibrium P PCl 5 Cl 2 (g) Kp = 0. 497 = 0. 860 atm, P PCl 3 = 0. 350 atm. Calculate P Cl 2 Kp = [P PCl 3][P Cl 2] / [P PCl 5] 0. 497 = [0. 350 ][P Cl 2] / [0. 860 ] [P Cl 2] = 1. 22 atm

Multiple Equilibria Suppose the products of one equilibrium reaction are the reactants in another A+B↔C+D Kc’ = [C][D] [A][B] C+D↔E+F Kc’’ = [E][F] [C][D] The overall reaction is A+B↔E+F Kc = [E][F] [A][B] The relationship between K , K, and K is Kc = Kc’’ If a reaction can be expressed as the sum of two or more reactions, the equilibrium constant for the overall reaction is given by the product of the equilibrium constants for the individual reactions. • •

Multiple Equilibria Example The following equilibrium constants have been determined for carbonic acid at 25 C. H 2 CO 3(aq) ↔ H+(aq) + HCO 3 -(aq) K = [H+][HCO 3 -] = 4. 2 x 10 [H 2 CO 3] • HCO (aq) ↔ H (aq) + CO • K = [H ][CO ] = 4. 8 x 10 3 + + [HCO 3 3 -] - 3 -(aq) • The overall reaction is • H CO (aq) ↔ 2 H (aq) + CO (aq) • K = [H ] [CO ] [H CO ] • What is the value of the equilibrium constant for the overall reaction? 2 + 3 + 2 2 3 3 -

Equilibrium Part 2
![Calculating keq If you don’t know all of the [equilibrium], you must make an Calculating keq If you don’t know all of the [equilibrium], you must make an](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-31.jpg)
Calculating keq If you don’t know all of the [equilibrium], you must make an ICE table I = initial concentration C= change in concentration E = equilibrium concentration Fill in what you know & solve for all unknowns

Reaction Quotient It is also useful to know whether a mixture is at equilibrium or, if not, the direction in which the system must shift to reach equilibrium. We use the reaction quotient (Q) to determine this For the synthesis of ammonia, Q = [NH 3 ] 2 [N 2] [H 2] 3

Reaction Quotient, cont'd Q is equal to K- the system is at equilibrium Q is greater than K- the system will shift to the left Q is less than K- the system will shift to the right.

Solving Equilibrium Problems Write the balanced equation for the reaction. Write the equilibrium expression using the law of mass action. List the initial concentrations. Calculate Q and determine the direction of the shift to equilibrium. Define the change needed to reach equilibrium, and define equilibrium concentrations by applying the change. Substitute the equilibrium concentrations into the equilibrium expression and solve for the unknown. Check your calculated equilibrium concentrations by making sure they give the correct value of K.

Calculating keq A mixture of 5. 00 x 10 -3 mol of H 2 and 1. 000 x 10 -2 mol of I 2 are placed into a 5. 000 L container at 448°C and allowed to come to equilibrium. Analysis of the equilibrium showed that [HI] was 1. 87 x 10 -3 M. Calculate kc at 448°C.
![Calculating keq H 2 + Kc = [HI]2 [H 2] [I 2] I 2 Calculating keq H 2 + Kc = [HI]2 [H 2] [I 2] I 2](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-36.jpg)
Calculating keq H 2 + Kc = [HI]2 [H 2] [I 2] I 2 ↔ 2 HI
![Calculating keq Initial Concentrations [H 2] initial = 5. 00 x 10 -3 mol Calculating keq Initial Concentrations [H 2] initial = 5. 00 x 10 -3 mol](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-37.jpg)
Calculating keq Initial Concentrations [H 2] initial = 5. 00 x 10 -3 mol of H 2 / 5. 000 L = 1. 000 x 10 -3 M H 2 [I 2] initial = 1. 00 x 10 -3 mol of H 2 / 5. 000 L = 2. 000 x 10 -3 M I 2 [HI] initial = 0 M • Equilibrium Concentrations • [HI] eq = 1. 87 x 10 -3 M

Calculating keq Do not need to calculate Q as the initial concentration of product is 0, the system must shift to the right. Knowing the change in 1, you can find the change in all 1. 87 x 10 -3 mol HI x 1 mol H 2 = 9. 35 x 10 -4 M H 2 1 L 2 mol HI 1. 87 x 10 -3 mol HI x 1 mol I 2 = 9. 35 x 10 -4 M I 2 1 L 2 mol HI
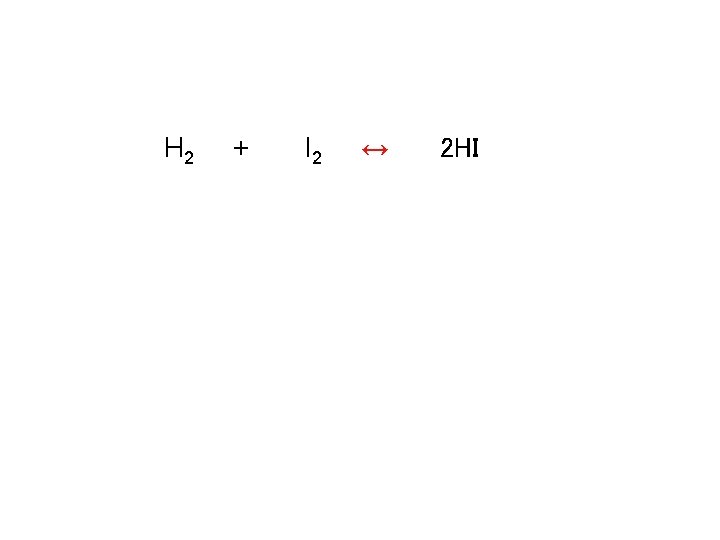
Calculating keq H 2 + I 2 ↔ 2 HI
![Calculating keq H 2 + I 2 ↔ 2 HI Kc = [HI]2 / Calculating keq H 2 + I 2 ↔ 2 HI Kc = [HI]2 /](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-40.jpg)
Calculating keq H 2 + I 2 ↔ 2 HI Kc = [HI]2 / [H 2][I 2] Kc = [1. 87 x 10 -3]2 / [6. 5 x 10 -5][1. 06 x 10 -3] Kc = 51
![Calculating keq 2 SO 3 ↔ 2 SO 2 + O 2 [SO 3] Calculating keq 2 SO 3 ↔ 2 SO 2 + O 2 [SO 3]](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-41.jpg)
Calculating keq 2 SO 3 ↔ 2 SO 2 + O 2 [SO 3] initial = 6. 09 x 10 -3 M [SO 3] eq = 2. 44 x 10 -3 M Calculate Kc
![Calculating keq 2 SO 3 ↔ 2 SO 2 + Kc = [SO 2]2 Calculating keq 2 SO 3 ↔ 2 SO 2 + Kc = [SO 2]2](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-42.jpg)
Calculating keq 2 SO 3 ↔ 2 SO 2 + Kc = [SO 2]2 [O 2] [SO 3]2 O 2
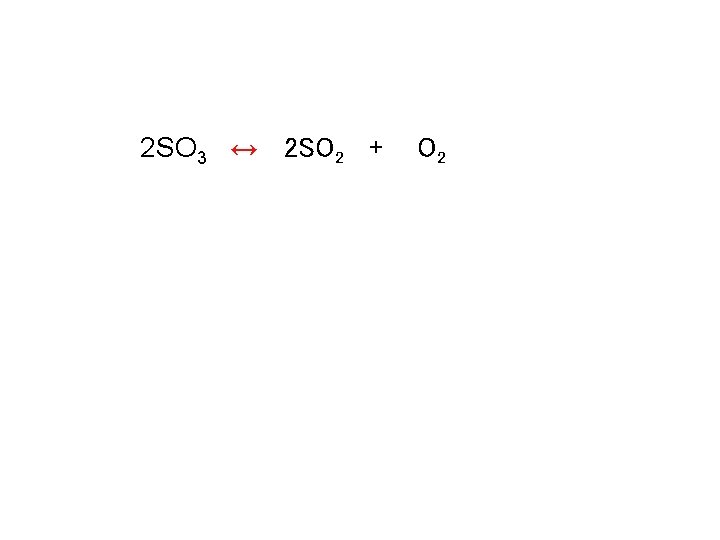
Calculating keq 2 SO 3 ↔ 2 SO 2 + O 2
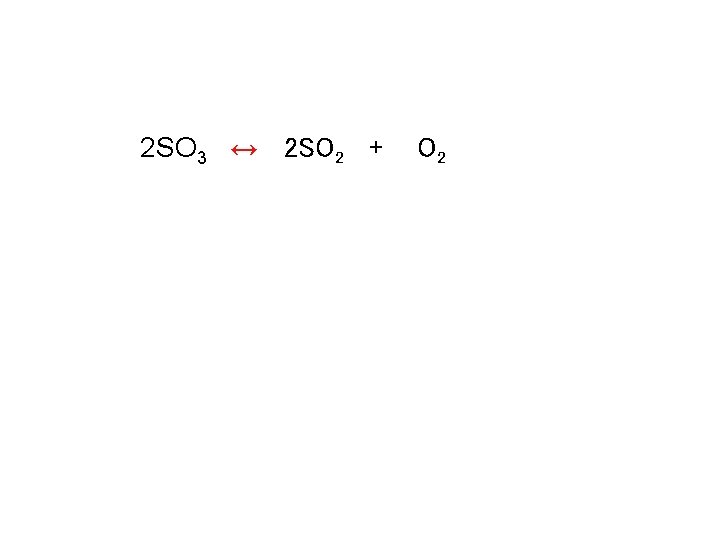
Calculating keq 2 SO 3 ↔ 2 SO 2 + O 2
![Calculating keq Kc = [SO 2]2[O 2] / [SO 3]2 Kc = [3. 65 Calculating keq Kc = [SO 2]2[O 2] / [SO 3]2 Kc = [3. 65](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-45.jpg)
Calculating keq Kc = [SO 2]2[O 2] / [SO 3]2 Kc = [3. 65 x 10 -3 ]2[1. 83 x 10 -3] / [2. 44 x 10 -3]2 Kc = 4. 10 x 10 -3

Calculating keq 3. 000 mol of H 2 and 6. 000 mol F 2 are placed in a 3. 000 L container. The kc for this reaction is 1. 15 x 10 2. Calculate the equilibrium concentrations for H 2, F 2, and HF
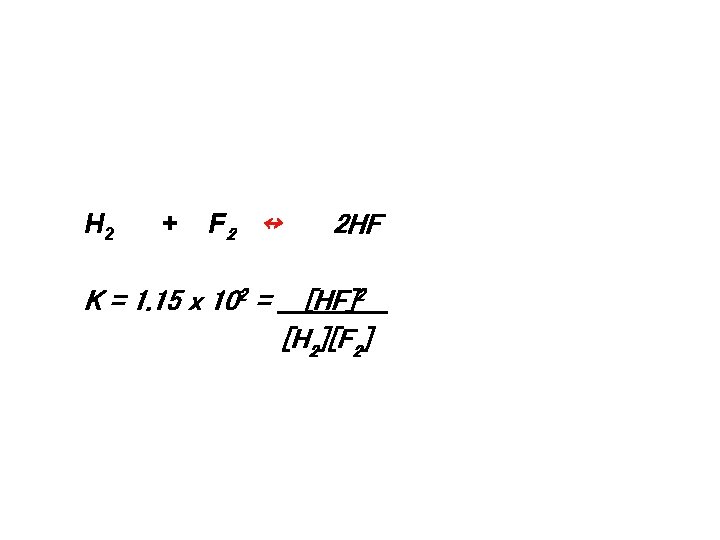
Calculating keq H 2 + F 2 ↔ K = 1. 15 x 102 = 2 HF [HF]2 [H 2][F 2]
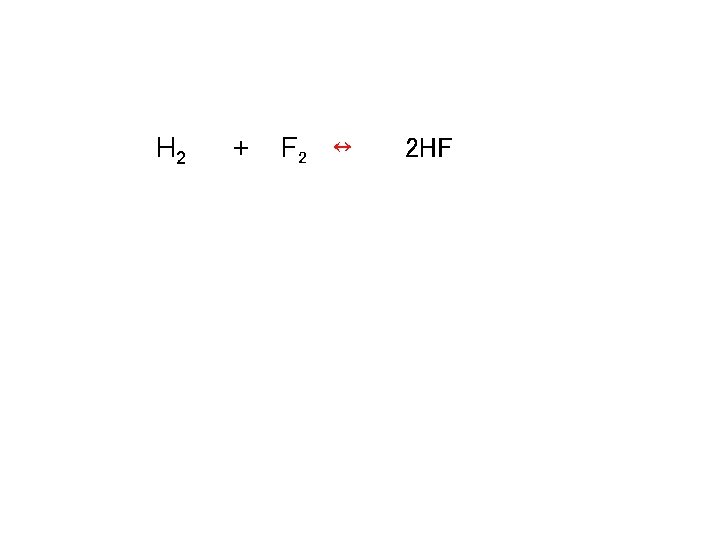
Calculating keq H 2 + F 2 ↔ 2 HF

Calculating keq H 2 + F 2 ↔ 2 HF
![Calculating keq H 2 + F 2 ↔ 2 HF Kc = [HF]2 / Calculating keq H 2 + F 2 ↔ 2 HF Kc = [HF]2 /](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-50.jpg)
Calculating keq H 2 + F 2 ↔ 2 HF Kc = [HF]2 / [H 2][F 2] Kc = 1. 15 x 102 = [2 x]2 / [1. 000 -x][2. 000 -x] (1. 000 -x)(2. 000 -x)(1. 15 x 102) = 4 x 2 (1. 11 x 102)x 2 – (3. 45 x 102)x + 2. 30 x 102 = 0 Use the quadratic equation to solve for x -b +/-√(b 2 -4 ac) 2 a X = 2. 14 or 0. 968
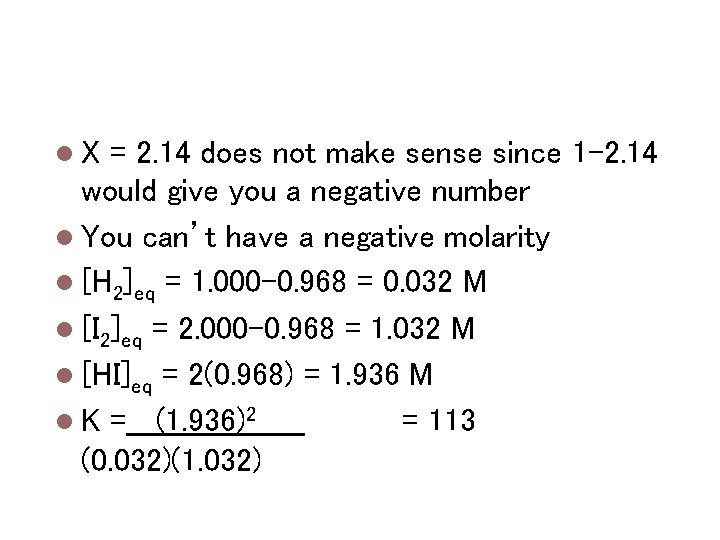
Calculating keq X = 2. 14 does not make sense since 1 -2. 14 would give you a negative number You can’t have a negative molarity [H 2]eq = 1. 000 -0. 968 = 0. 032 M [I 2]eq = 2. 000 -0. 968 = 1. 032 M [HI]eq = 2(0. 968) = 1. 936 M K =_ _(1. 936)2 = 113 (0. 032)(1. 032)

Calculating keq PCl 5 ↔ PCl 3 + Cl 2 Kp = 0. 497 (P PCl 5) initial = 1. 66 atm Calculate all equilibrium partial pressures
![Calculating keq PCl 5 ↔ K = [PCl 3][Cl 2] [PCl 5] PCl 3 Calculating keq PCl 5 ↔ K = [PCl 3][Cl 2] [PCl 5] PCl 3](http://slidetodoc.com/presentation_image_h2/27cec07b2fd075007689075d41b395a5/image-53.jpg)
Calculating keq PCl 5 ↔ K = [PCl 3][Cl 2] [PCl 5] PCl 3 + Cl 2
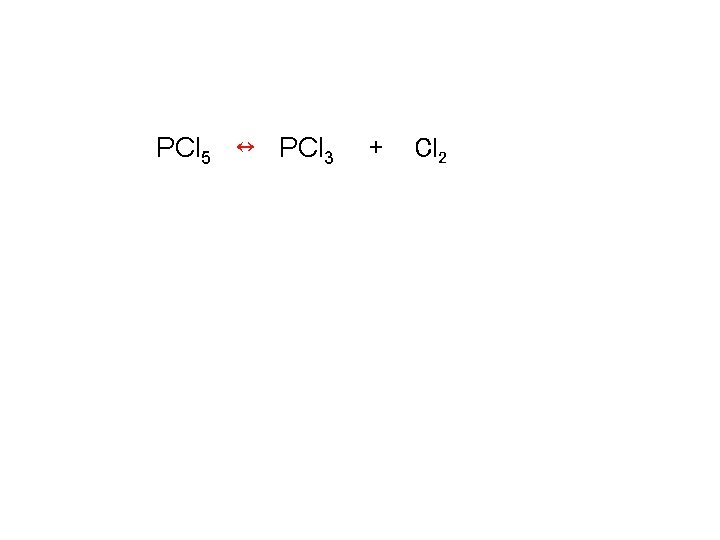
Calculating keq PCl 5 ↔ PCl 3 + Cl 2

Calculating keq Kc = (P PCl 3)(P Cl 2) / (P PCl 5) Kc = (x)(x) / (1. 66 - x) X = 0. 689 or -1. 18 Eliminate -1. 18 (P PCl 5) = 1. 66 – 0. 689 = 0. 971 atm (P PCl 3) = 0. 689 atm (P Cl 2) = 0. 689 atm K =(0. 693) = 0. 495 0. 97

Le. Châtelier’s Principle Le Châtelier’s principle states that if a system at equilibrium is subjected to a stress, the equilibrium is shifted in the direction that tends to relieve the stress. Stresses include: concentration, temperature, volume & pressure Stresses DO NOT include: adding a catalyst & increasing surface area

Concentration CO + 3 H 2 ↔ CH 4 + H 2 O Which way does the equilibrium shift if [CO] is increased Products Which way does the equilibrium shift if [CH 4] is increased Reactants Which way does the equilibrium shift if [H 2] is increased products Which way does the equilibrium shift if [H 2 O] is increased reactants

Concentration Add N 2 Products Add H 2 Products Add NH 3 Reactants Remove N 2 Reactants Remove H 2 Reactants Remove NH 3 Products N 2 + 3 H 2 ↔ 2 NH 3
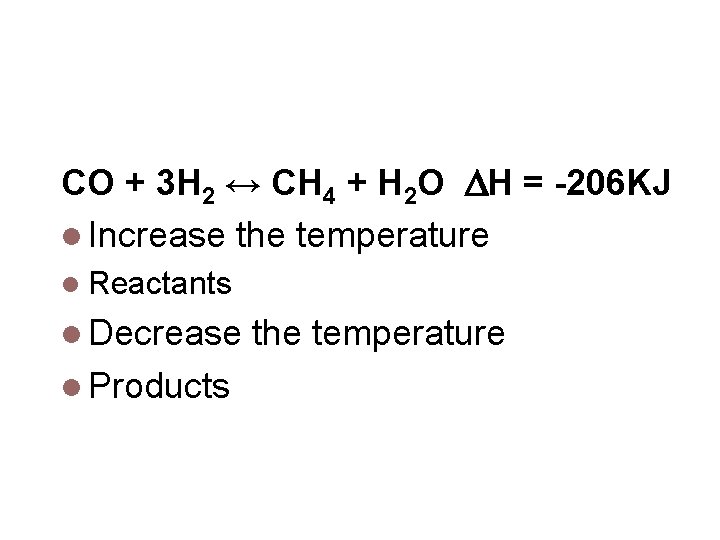
Temperature CO + 3 H 2 ↔ CH 4 + H 2 O H = -206 KJ Increase the temperature Reactants Decrease Products the temperature

Volume & Pressure When doing volume & Pressure…you look at the number of moles CO + 3 H 2 ↔ CH 4 + H 2 O 4 moles 2 moles If you decrease the volume (which is increasing the pressure) you are getting cramped & the molecules will want to go to the side with the fewest # of moles) If you increase the volume (which is decreasing the pressure) you have room will want to go to the side with the most # of moles)

Volume & Pressure CO + 3 H 2 ↔ CH 4 + H 2 O Increase the pressure Products Increase the volume Reactants Decrease the volume Products Decrease the pressure Reactants Added He to the system No change

Volume & Pressure If the number of moles are the same…there will be no change when to the system when volume & pressure are changed. For example… H 2 + I 2 ↔ 2 HI Volume & pressure no change
- Slides: 62