INTRODUCTION TO ELECTRONIC INSTRUMENTATION Chapter 6 INTC 1305

































- Slides: 34

INTRODUCTION TO ELECTRONIC INSTRUMENTATION Chapter 6 INTC 1305 – 01 ANALYTICAL INSTRUMENTS Vern Wilson

Super Hadron Project Switzerland � http: //www. youtube. com/watch? v=j 50 Zss. Eoj t. M

Review of Chapter 5 – FLOW � Fluid in motion � Always from hi to lo p � Molecules continually change � Laminar - streamline � Turbulent � Obstructions mess up smooth flow � Usually turbulent flow is consistent

MEASUREMENT � FLOW MEASURED IN: �GPM �POUNDS PER MINUTE

GPM EXERCISE � An injection program requires 900 barrels of injection fluid per day. � How many GPM is equal to 900 barrels per day? � First: There are 42 gallons per barrel � Second: Convert to GPM

POUNDS PER MINUTE EXERCISE � We are pumping mud that weighs 10. 8 pounds per gallon at 15 barrels per minute. � How many pounds per minute are we pumping? � First: Determine how many pounds are we pumping = 42 gals/barrel � Second: Determine how many pounds per minute.

ANALYTICAL INSTRUMENTS Chapter 6

LEARNING THE TERMS � DCS = Distributed Control System – Man/Machine interface � ESD = Emergency Shut Down � SP = Set Point – the desired process value � Live Zero = Instead of reading zero the instrument reads 3 (psig) or 4 (ma) � PLC = Programmable Logic Controller � ΔP = Differential Pressure � SCADA = Supervisory Control and Data Acquisition

Objectives � Define ANALYTICAL – Using logical techniques � Review instruments � Discuss the purposes

Intro to Analytical Instruments � Instruments designed to monitor chemical and physical properties of a process stream � These are instruments that can continuously monitor a process stream � The analyzer system catches a sample, sends it to an analyzer, conditions the sample, analyzes the sample and returns the sample to the system

ANALYZERS � Designed to catch a physical sample � That sample is analyzed and put back in system � They get plugged, contaminated, bent and broken

Controllers and Control Theory � http: //ipims. com/scorm/IPIMS_BL_Module/m odule. FS. asp? Student. ID=34609&First. Name=& Last. Name=&Email=&Launch. URL=%2 Fwelcom e%5 F%2 Fportal%2 Easp&Launch. Frame=%5 Ftop &Course. Entry=1&Max. Attempt=&Passing. Grad e=&Reload. Opener=Y&Show. Logout. Link=N&Fir st. Page. URL=%2 Fwelcome%5 F%2 Fportal%2 Easp &Course. ID=142

p. H Measurements � Measurement of hydrogen ion concentration � The instrument measures ions after they have been isolated into Positive and Negative ions � Measures a millivolt potential caused and compares it to a known p. H

p. H Meter

ORP � Oxidation Reduction Potential – �OXIDATION means the loss of electrons �REDUCTION means the gain of electrons � Measures the specific free electron potential to let you see into the chemical reaction of a process

ORP - Oxidation. Reduction. Potential

CONDUCTIVITY � Measures �C ability to conduct electricity =1/R � Conductivity is the reciprocal of resistivity � When Na. Cl goes into water the Na and Cl separate making the water into an electrolyte � Can help analyze the composition

Conductivity Meter

OPTICAL MEASUREMENT � Uses reflection, refraction, or absorption light � Color – can indicate process problems � Turbidity – measures cloudiness of liquid � Opacity – measures particles in gas

Turbidity Meter

COMBUSTION � Rapid oxidation of a substance

Analytical Sensing Instruments � EPA � Mechanical Integrity – can be affected by acidity or alkalinity � Economics – continual quality control � Product Quality Assurance – Strict calibration

Common Types Left off 7 18 11 � p. H / ORP – Electrochemical analyzers – a specific ion analyzer � p. H Meter – measures acidity and alkalinity � 0 -14 � ORP – Oxidation Reduction Potential Meter – measures how much free electron potential exists � Conductivity meters – Recip of R – Pure water is Non Conductive – add a little Na. Cl and it becomes conductive. Meter quantifies ions.

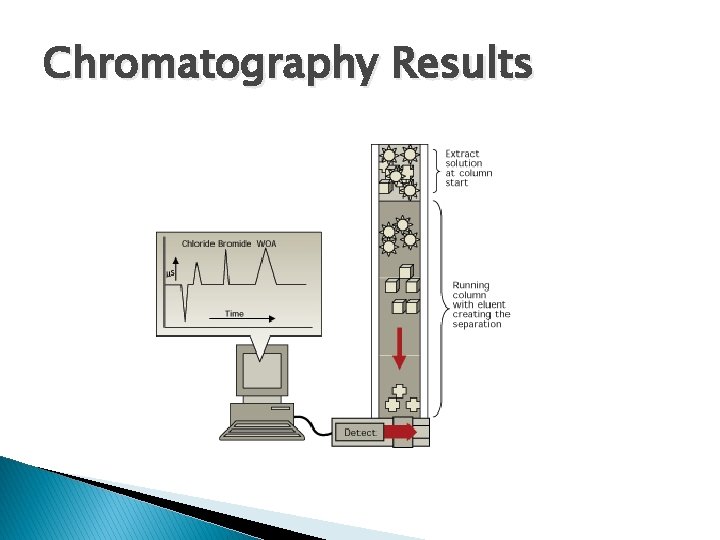
Chromatographs � Provides a molecular separation of components

Chromatograph

Results of Chromatograph

Chromatography Results

Mass Spectrometers �A device capable of separating a gaseous stream into a spectrum according to mass and charge.

Spectrometers � An analyzer to detect and or quantify chemical components in a process sample by measuring variations in transmittance (or absorption) of a spectrum of light passed through the sample.

Spectrometer

Total Carbon Analyzers � Carbon is in everything � Used to determine how much carbon is in a sample � Used to detect carbon based contaminants and steam condensate and waste water � Works for organic and inorganic matter in a sample

Total Carbon Analyzers

Other Monitors � Personnel monitors � Area monitors � Unit or Plant monitors

Watch type H 2 S detector