Introduction to Electroanalytical Chemistry Potentiometry Voltammetry Amperometry Biosensors

Introduction to Electroanalytical Chemistry Potentiometry, Voltammetry, Amperometry, Biosensors

Uses • To study Redox Chemistry – electron transfer reactions, oxidation, reduction, organics & inorganics, proteins – Adsorption of species at interfaces • Electrochemical analysis – Measure the Potential of reaction or process E = const + k log C (potentiometry) – Measure the Rate of a redox reaction; Current (I) = k C (voltammetry); C = concentration

Electrochemical Cells • Galvanic Cells and Electrolytic Cells • Galvanic Cells – power output; batteries (car) • Potentiometric cells (I=0) – measure potential for analyte to react – current = 0 (reaction is not allowed to occur) – Equil. Voltage is measured (Eeq) • Electrolytic cells, power applied, output meas. – The Nernst Equation • For a reversible process: Ox + ne- → Red • E = Eo – (2. 303 RT/n. F) Log (ared/aox) • a (activity), proportional to concentration

Voltammetry is a dynamic method Related to rate of reaction at an electrode O + ne = R, Eo in Volts I = k. A[O] k = const. A = area Faradaic current, caused by electron transfer Also a non-faradaic current forms part of background current
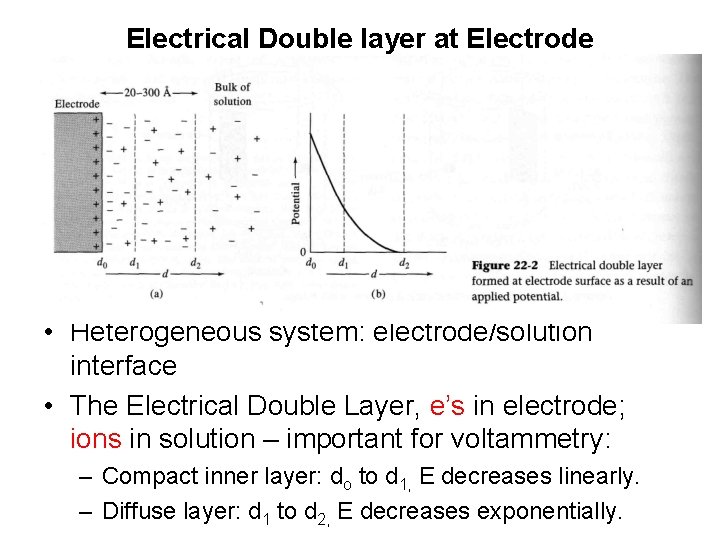
Electrical Double layer at Electrode • Heterogeneous system: electrode/solution interface • The Electrical Double Layer, e’s in electrode; ions in solution – important for voltammetry: – Compact inner layer: do to d 1, E decreases linearly. – Diffuse layer: d 1 to d 2, E decreases exponentially.

Electrolysis: Faradaic and Non-Faradaic Currents • Two types of processes at electrode/solution interface that produce current – Direct transfer of electrons (ET), oxidation, reduction • Faradaic Processes. ET reaction rate at electrode proportional to the Faradaic current. – Nonfaradaic current: due to change in double layer when E is changed; not useful for analysis • Mass Transport: continuously brings reactant from the bulk of solution to electrode surface to be oxidized or reduced (Faradaic) – Convection: stirring or flowing solution – Migration: electrostatic attraction of ion to electrode – Diffusion: due to concentration gradient.
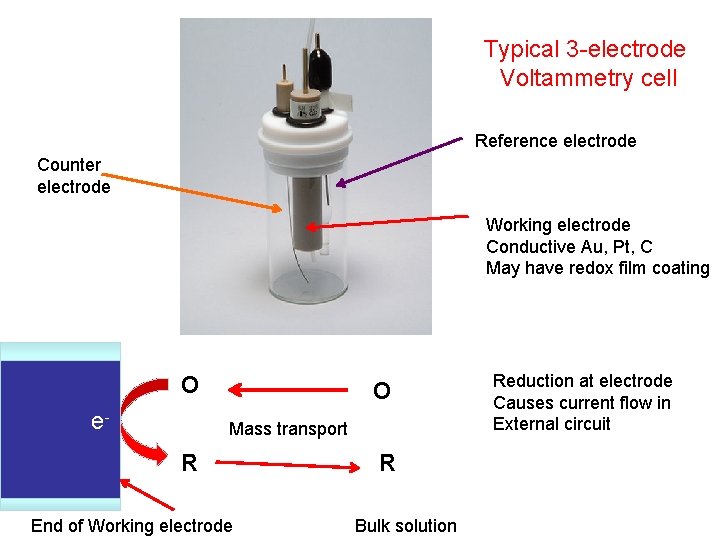
Typical 3 -electrode Voltammetry cell Reference electrode Counter electrode Working electrode Conductive Au, Pt, C May have redox film coating O e- O Mass transport R End of Working electrode R Bulk solution Reduction at electrode Causes current flow in External circuit

Analytical Electrolytic Cells • Use external potential (voltage) to drive reaction • Applied potential controls electron energy • As Eo gets more negative, need more energetic electrons in order to cause reduction. For a reversible reaction: – Eapplied is more negative than Eo, reduction will occur – if Eapplied is more positive than Eo, oxidation will occur O + ne- = R Eo, V electrode reaction
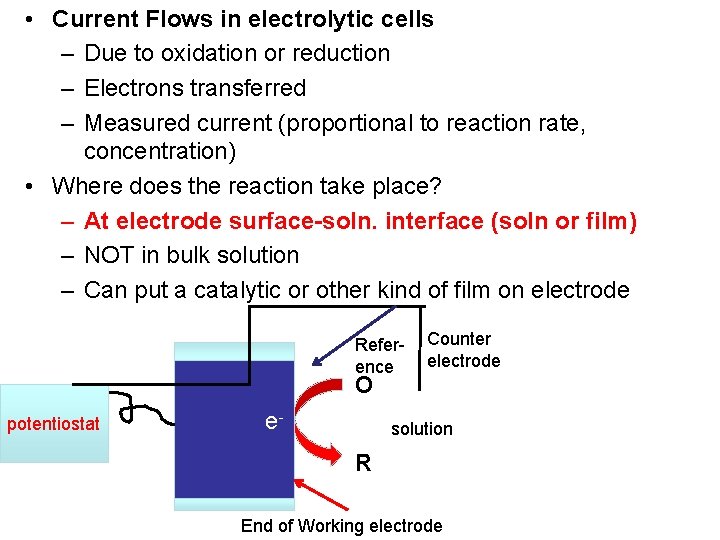
• Current Flows in electrolytic cells – Due to oxidation or reduction – Electrons transferred – Measured current (proportional to reaction rate, concentration) • Where does the reaction take place? – At electrode surface-soln. interface (soln or film) – NOT in bulk solution – Can put a catalytic or other kind of film on electrode Reference Counter electrode O potentiostat e- solution R End of Working electrode

Analytical Applications of Electrolytic Cells • Amperometry – Set Eapplied so that desired reaction occurs – Stir or flow solution – Measure Current • Voltammetry – Quiet or stirred solution; or redox film on electrode – Vary (“scan”) Eapplied – Measure Current • • Indicates reaction rate Reaction at electrode surface Mass transport brings reactive species to electrode surface For film, mass transport within and outside of film
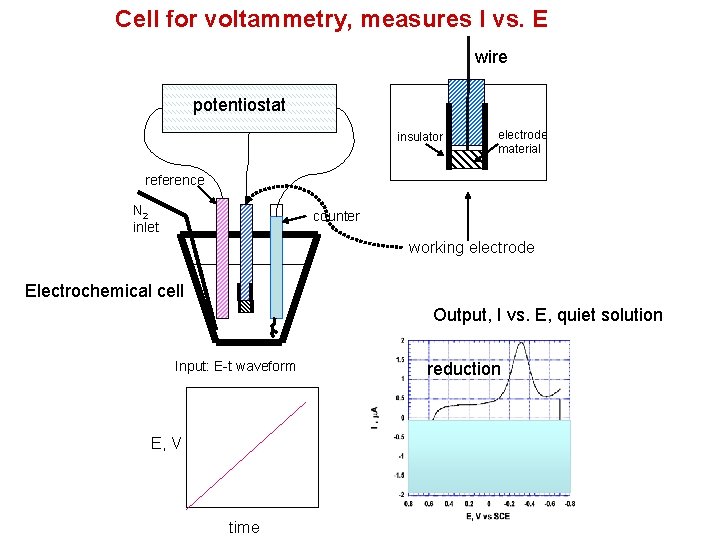
Cell for voltammetry, measures I vs. E wire potentiostat insulator electrode material reference N 2 inlet counter working electrode Electrochemical cell Output, I vs. E, quiet solution Input: E-t waveform E, V time reduction

NERNST Equation: Fundamental Equation for reversible electron transfer at electrodes O + ne- = R, Eo in Volts • E. g. , Fe 3+ + e- = Fe 2+ If in a cell, I = 0, then E = Eeq All equilibrium electrochemical reactions obey the Nernst Equation Reversibility means that O and R are at equilibrium at all times, not all Electrochemical reactions are reversible E = Eo - [RT/n. F] ln (a. R/a. O) a. R = f R CR ao = f o Co ; a = activity f = activity coefficient, depends on ionic strength Then E = Eo - [RT/n. F] ln (f. R/f. O) - [RT/n. F] ln (CR/CO) F = Faraday const. , 96, 500 coul/e, R = gas const. T = absolute temperature

Ionic strength I = Σ zi 2 mi, z = charge on ion, m = concentration of ion Debye Huckel theory says log f. R = 0. 5 zi 2 I 1/2 So f. R/f. Owill be constant at constant I. And so, below are more usable forms of Nernst Eqn. E = Eo - const. - [RT/n. F] ln (CR/CO) Or E = Eo’ - [RT/n. F] ln (CR/CO); Eo’ = formal potential of O/R At 25 o. C using base 10 logs E = Eo’ - [0. 0592/n] log (CR/CO); equil. systems
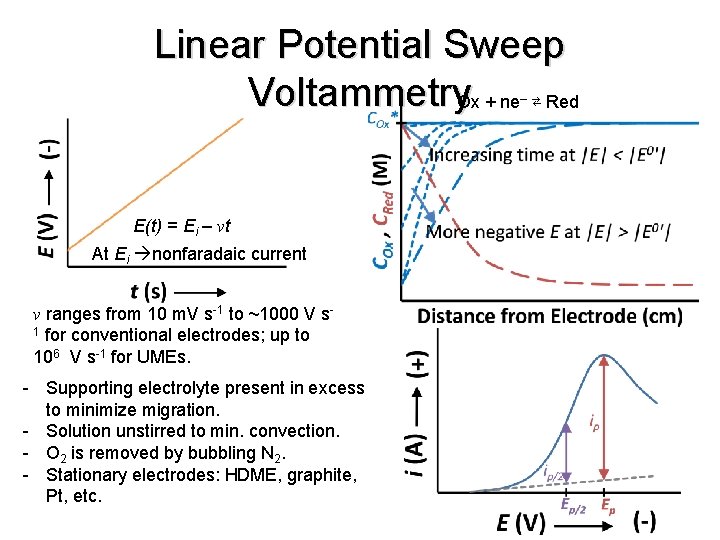
Linear Potential Sweep Voltammetry. Ox + ne ⇄ Red – E(t) = Ei – vt At Ei nonfaradaic current v ranges from 10 m. V s-1 to ~1000 V s 1 for conventional electrodes; up to 106 V s-1 for UMEs. - Supporting electrolyte present in excess to minimize migration. - Solution unstirred to min. convection. - O 2 is removed by bubbling N 2. - Stationary electrodes: HDME, graphite, Pt, etc.
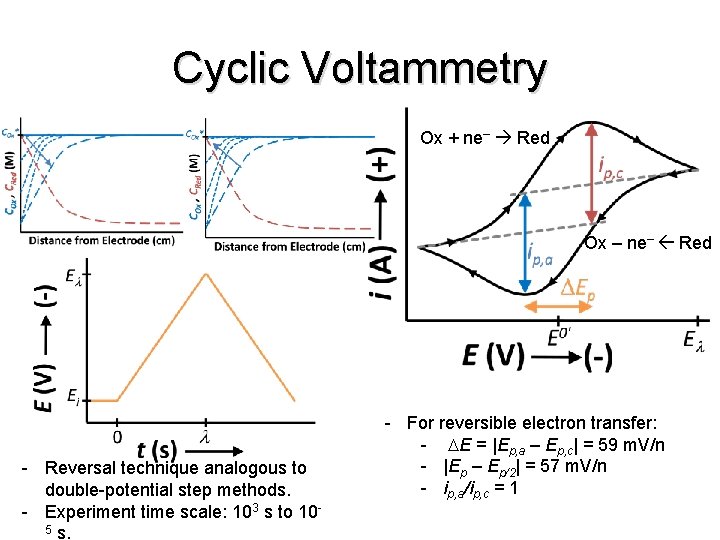
Cyclic Voltammetry Ox + ne– Red Ox – ne– Red - Reversal technique analogous to double-potential step methods. - Experiment time scale: 103 s to 105 s. - For reversible electron transfer: - DE = |Ep, a – Ep, c| = 59 m. V/n - |Ep – Ep/2| = 57 m. V/n - ip, a/ip, c = 1
- Slides: 15