Introduction to Computational Chemistry Shubin Liu Ph D

Introduction to Computational Chemistry Shubin Liu, Ph. D. Research Computing Center University of North Carolina at Chapel Hill

Outline § Introduction § Methods in Computational Chemistry • Ab Initio • Semi-Empirical • Density Functional Theory • New Developments (QM/MM) § Hands-on Exercises The PPT format of this presentation is available here: http: //its 2. unc. edu/divisions/rc/training/scientific/ /afs/isis/depts/its/public_html/divisions/rc/training/scientific/short_courses/ its. unc. edu 2

About Us § ITS – Information Technology Services • • http: //its. unc. edu http: //help. unc. edu Physical locations: w 401 West Franklin St. w 211 Manning Drive 10 Divisions/Departments w Information Security w Research Computing Center w User Support and Engagement w Communication Technologies w Enterprise Applications its. unc. edu IT Infrastructure and Operations Teaching and Learning Office of the CIO Communications Finance and Administration 3

Research Computing § Where and who are we and what do we do? • • ITS Manning: 211 Manning Drive Website http: //its. unc. edu/research-computing. html • Groups w Infrastructure -- Hardware w User Support -- Software w Engagement -- Collaboration its. unc. edu 4

About Myself § Ph. D. from Chemistry, UNC-CH § Currently Senior Computational Scientist @ Research Computing Center, UNC-CH § Responsibilities: • Support Computational Chemistry/Physics/Material Science software • Support Programming (FORTRAN/C/C++) tools, code porting, parallel computing, etc. • Offer short courses on scientific computing and computational chemistry • Conduct research and engagement projects in Computational Chemistry w Development of DFT theory and concept tools w Applications in biological and material science systems its. unc. edu 5

About You § Name, department, research interest? § Any experience before with high performance computing? § Any experience before with computational chemistry research? § Do you have any real problem to solve with computational chemistry approaches? its. unc. edu 6

Think BIG!!! § What is not chemistry? • § • • What cannot computational chemistry deal with? • From small molecules, to DNA/proteins, 3 D crystals and • • its. unc. edu From microscopic world, to nanotechnology, to daily life, to environmental problems From life science, to human disease, to drug design Only our mind limits boundary surfaces From species in vacuum, to those in solvent at room temperature, and to those under extreme conditions (high T/p) From structure, to properties, to spectra (UV, IR/Raman, NMR, VCD), to dynamics, to reactivity All experiments done in labs can be done in silico Limited only by (super)computers not big/fast enough! 7
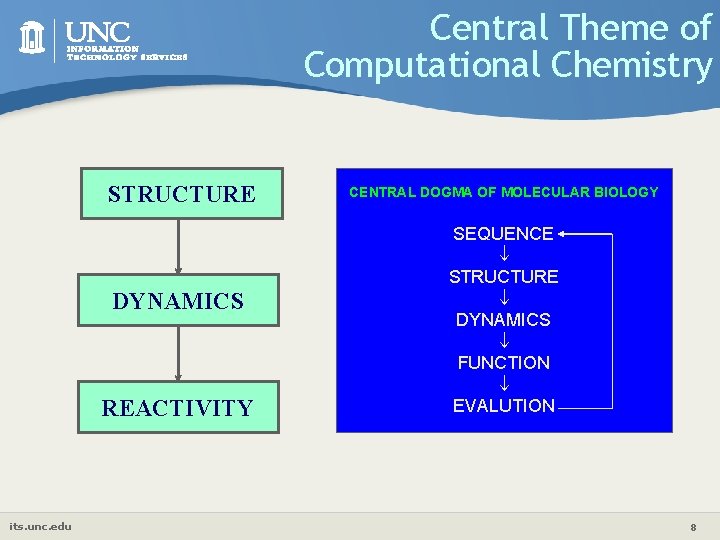
Central Theme of Computational Chemistry STRUCTURE DYNAMICS REACTIVITY its. unc. edu CENTRAL DOGMA OF MOLECULAR BIOLOGY SEQUENCE STRUCTURE DYNAMICS FUNCTION EVALUTION 8
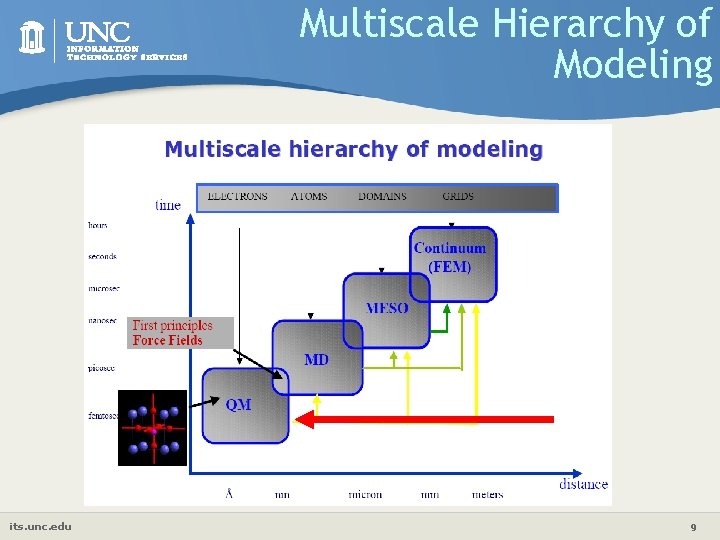
Multiscale Hierarchy of Modeling its. unc. edu 9

What is Computational Chemistry? Application of computational methods and algorithms in chemistry • Quantum Mechanical i. e. , via Schrödinger Equation also called Quantum Chemistry • Molecular Mechanical i. e. , via Newton’s law F=ma also Molecular Dynamics Focus Today • Empirical/Statistical e. g. , QSAR, etc. , widely used in clinical and medicinal chemistry its. unc. edu 10

How Big Systems Can We Deal with? Assuming typical computing setup (number of CPUs, memory, disk space, etc. ) § § its. unc. edu Ab initio method: ~100 atoms DFT method: ~1000 atoms Semi-empirical method: ~10, 000 atoms MM/MD: ~100, 000 atoms 11
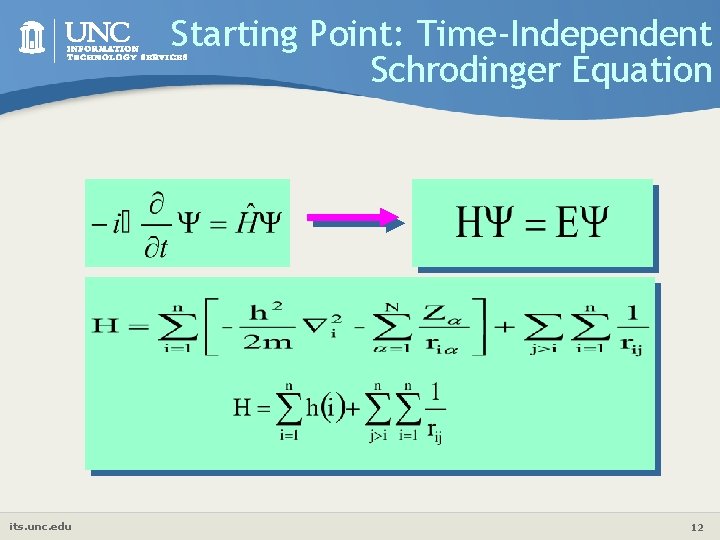
Starting Point: Time-Independent Schrodinger Equation its. unc. edu 12
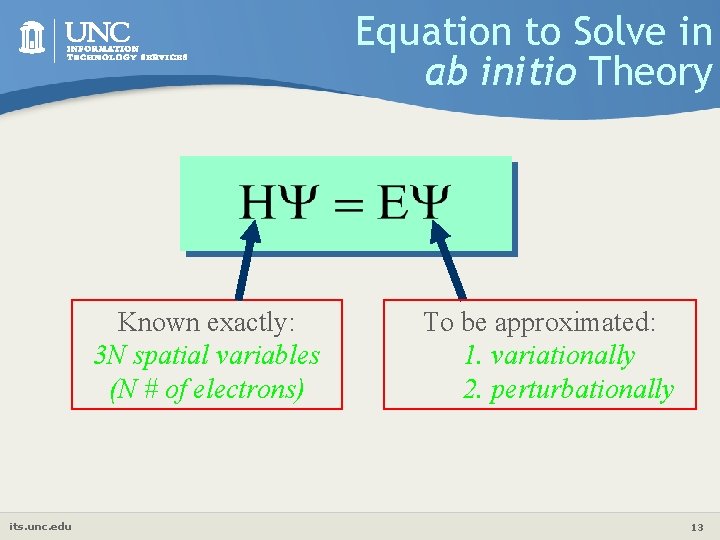
Equation to Solve in ab initio Theory Known exactly: 3 N spatial variables (N # of electrons) its. unc. edu To be approximated: 1. variationally 2. perturbationally 13

Hamiltonian for a Molecule § kinetic energy of the electrons § kinetic energy of the nuclei § electrostatic interaction between the electrons and the nuclei § electrostatic interaction between the electrons § electrostatic interaction between the nuclei its. unc. edu 14

Ab Initio Methods § Accurate treatment of the electronic distribution using the § § § its. unc. edu full Schrödinger equation Can be systematically improved to obtain chemical accuracy Does not need to be parameterized or calibrated with respect to experiment Can describe structure, properties, energetics and reactivity What does “ab intio” mean? • Start from beginning, with first principle Who invented the word of the “ab initio” method? • Bob Parr of UNC-CH in 1950 s; See Int. J. Quantum Chem. 37(4), 327(1990) for details. 15

Three Approximations § Born-Oppenheimer approximation • Electrons act separately of nuclei, electron and nuclear coordinates are independent of each other, and thus simplifying the Schrödinger equation § Independent particle approximation • Electrons experience the ‘field’ of all other electrons as a group, not individually • Give birth to the concept of “orbital”, e. g. , AO, MO, etc. § LCAO-MO approximation • Molecular orbitals (MO) can be constructed as linear combinations of atom orbitals, to form Slater determinants its. unc. edu 16

Born-Oppenheimer Approximation § the nuclei are much heavier than the electrons and move more slowly than the electrons § freeze the nuclear positions (nuclear kinetic energy is zero in the electronic Hamiltonian) § calculate the electronic wave function and energy § E depends on the nuclear positions through the nuclear-electron attraction and nuclear-nuclear repulsion terms § E = 0 corresponds to all particles at infinite separation its. unc. edu 17

Approximate Wavefunctions ¶ Construction of one-electron functions (molecular orbitals, MO’s) as linear combinations of one-electron atomic basis functions (AOs) MO-LCAO approach. · Construction of N-electron wavefunction as linear combination of anti-symmetrized products of MOs (these anti-symmetrized products are denoted as Slaterdeterminants). its. unc. edu 18

The Slater Determinant its. unc. edu 19

The Two Extreme Cases ¶ One determinant: The Hartree–Fock method. There are N MOs and each MO is a linear combination of N AOs. Thus, there are n N coefficients ukl, which are determined by making stationary the functional: The ij are Lagrangian multipliers. · All possible determinants: The full CI method. its. unc. edu 20

The Full CI Method § The full configuration interaction (full CI) method expands the wavefunction in terms of all possible Slater determinants: § There are possible ways to choose n molecular orbitals from a set of 2 N AO basis functions. § The number of determinants gets easily much too large. For example: Davidson’s method can be used to find one or a few eigenvalues of a matrix of rank 109. its. unc. edu 21

The Hartree–Fock Method Hartree–Fock equations its. unc. edu 22

The Hartree–Fock Method Overlap integral Density Matrix its. unc. edu 23

Self-Consistent-Field (SCF) 1. Choose start coefficients for MO’s 2. Construct Fock Matrix with coefficients 3. Solve Hartree-Fock-Roothaan equations 4. Repeat 2 and 3 until ingoing and outgoing coefficients are the same its. unc. edu 24
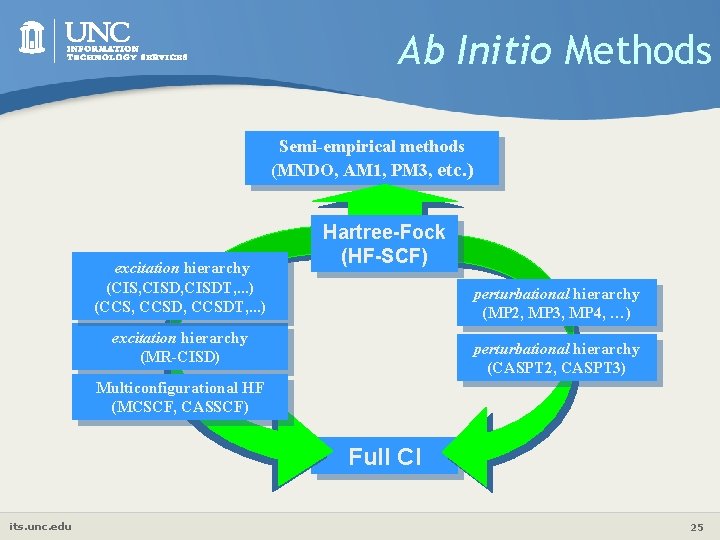
Ab Initio Methods Semi-empirical methods (MNDO, AM 1, PM 3, etc. ) excitation hierarchy (CIS, CISDT, . . . ) (CCS, CCSDT, . . . ) Hartree-Fock (HF-SCF) perturbational hierarchy (MP 2, MP 3, MP 4, …) excitation hierarchy (MR-CISD) perturbational hierarchy (CASPT 2, CASPT 3) Multiconfigurational HF (MCSCF, CASSCF) Full CI its. unc. edu 25

Who’s Who its. unc. edu 26
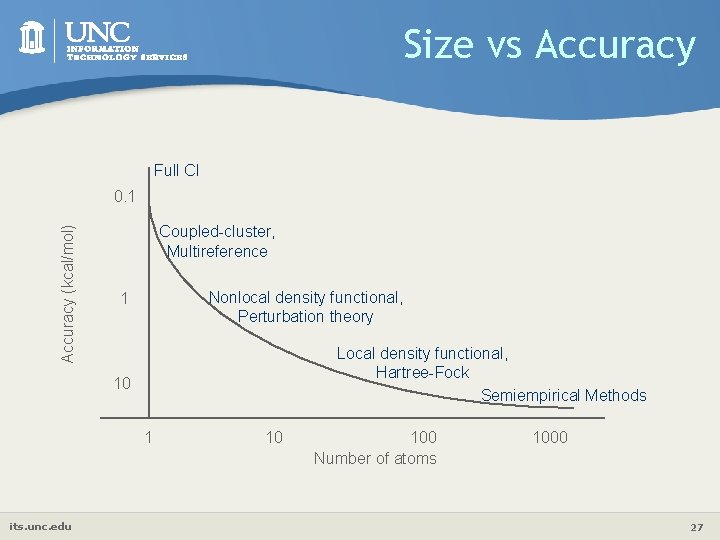
Size vs Accuracy Full CI Accuracy (kcal/mol) 0. 1 Coupled-cluster, Multireference Nonlocal density functional, Perturbation theory 1 Local density functional, Hartree-Fock Semiempirical Methods 10 1 its. unc. edu 10 100 Number of atoms 1000 27

AN EXAMPLE Equilibrium structure of (H 2 O)2 W. K. , J. G. C. M. van Duijneveldt-van de Rijdt, and F. B. van Duijneveldt, Phys. Chem. Phys. 2, 2227 (2000). 95. 7 pm symmetry: RCOO, e = 291. 2 s 96. 4 pm 95. 8 pm pm Experimental [J. A. Odutola and T. R. Dyke, J. Chem. Phys 72, 5062 (1980)]: ROO 2 ½ = 297. 6 ± 0. 4 pm SAPT-5 s potential [E. M. Mas et al. , J. Chem. Phys. 113, 6687 (2000)]: ROO 2 ½ – ROO, e= 6. 3 pm ROO, e(exptl. ) = 291. 3 pm its. unc. edu 28
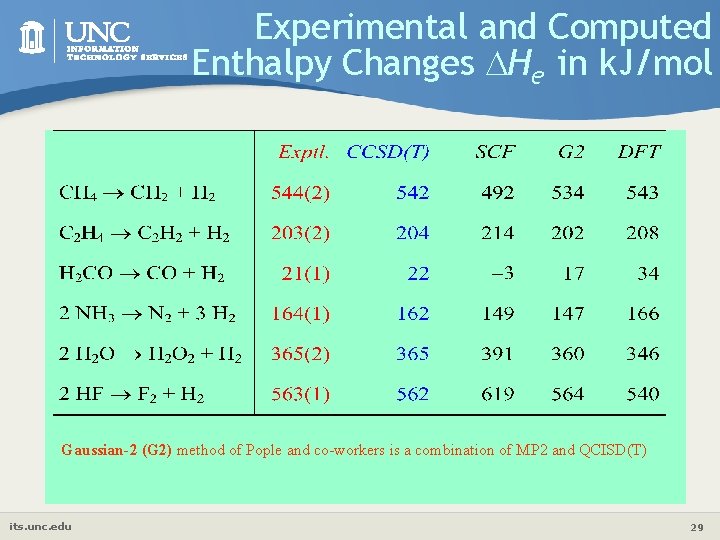
Experimental and Computed Enthalpy Changes He in k. J/mol Gaussian-2 (G 2) method of Pople and co-workers is a combination of MP 2 and QCISD(T) its. unc. edu 29

LCAO Basis Functions § ’s, which are atomic orbitals, are called basis functions § § usually centered on atoms § larger number of well chosen basis functions yields more accurate approximations to the molecular orbitals its. unc. edu can be more general and more flexible than atomic orbital functions 30

Basis Functions § Slaters (STO) § Angular part * § Better behaved than Gaussians § 2 -electron integrals hard § Gaussians (GTO) § 2 -electron integrals simpler § Wrong behavior at nucleus § Decrease too fast with r its. unc. edu 31

Contracted Gaussian Basis Set § Minimal • STO-n. G • § Split Valence: 321 G, 4 -31 G, 631 G its. unc. edu Each atom optimized STO is fit with n GTO’s Minimum number of AO’s needed • Contracted GTO’s optimized per atom • Doubling of the number of valence AO’s 32

Polarization / Diffuse Functions § Polarization: Add AO with higher angular momentum (L) to give more flexibility Example: 3 -21 G*, 6 -31 G**, etc. § Diffusion: Add AO with very small exponents for systems with very diffuse electron densities such as anions or excited states Example: 6 -31+G*, 6 -311++G** its. unc. edu 33

Correlation-Consistent Basis Functions its. unc. edu § § a family of basis sets of increasing size § § cc-p. VQZ, cc-p. V 5 Z, cc-p. V 6 Z can be used to extrapolate to the basis set limit cc-p. VDZ – DZ with d’s on heavy atoms, p’s on H cc-p. VTZ – triple split valence, with 2 sets of d’s and one set of f’s on heavy atoms, 2 sets of p’s and 1 set of d’s on hydrogen can also be augmented with diffuse functions (aug-cc-p. VXZ) 34

Pseudopotentials, Effective Core Potentials its. unc. edu § core orbitals do not change much during chemical interactions § valence orbitals feel the electrostatic potential of the nuclei and of the core electrons § can construct a pseudopotential to replace the electrostatic potential of the nuclei and of the core electrons § reduces the size of the basis set needed to represent the atom (but introduces additional approximations) § for heavy elements, pseudopotentials can also include of relativistic effects that otherwise would be costly to treat 35

Correlation Energy § HF does not include correlations anti-parallel electrons § Eexact – EHF = Ecorrelation § Post HF Methods: • Configuration Interaction (CI, MCSCF, CCSD) • Møller-Plesset Perturbation series (MP 2, MP 4) § Density Functional Theory (DFT) its. unc. edu 36

Configuration-Interaction (CI) o In Hartree-Fock theory, the n-electron wavefunction is approximated by one single Slater-determinant, denoted as: o This determinant is built from n orthonormal spin-orbitals. The spin-orbitals that form are said to be occupied. The other orthonormal spin-orbitals that follow from the Hartree-Fock calculation in a given one-electron basis set of atomic orbitals (AOs) are known as virtual orbitals. For simplicity, we assume that all spin-orbitals are real. o In electron-correlation or post-Hartree-Fock methods, the wavefunction is expanded in a many-electron basis set that consists of many determinants. Sometimes, we only use a few determinants, and sometimes, we use millions of them: In this notation, is a Slaterdeterminant that is obtained by replacing a certain number of occupied orbitals by virtual ones. o Three questions: 1. Which determinants should we include? 2. How do we determine the expansion coefficients? 3. How do we evaluate the energy (or other properties)? its. unc. edu 37

Truncated configuration interaction: CIS, CISDT, etc. § We start with a reference wavefunction, for example the Hartree. Fock determinant. § We then select determinants for the wavefunction expansion by substituting orbitals of the reference determinant by orbitals that are not occupied in the reference state (virtual orbitals). § Singles (S) indicate that 1 orbital is replaced, doubles (D) indicate 2 replacements, triples (T) indicate 3 replacements, etc. , leading to CIS, CISDT, etc. its. unc. edu 38

Truncated Configuration Interaction Number of linear variational parameters in truncated CI for n = 10 and 2 N = 40. its. unc. edu 39

Multi-Configuration Self-Consistent Field (MCSCF) o The MCSCF wavefunctions consists of a few selected determinants or CSFs. In the MCSCF method, not only the linear weights of the determinants are variationally optimized, but also the orbital coefficients. o One important selection is governed by the full CI space spanned by a number of prescribed active orbitals (complete active space, CAS). This is the CASSCF method. The CASSCF wavefunction contains all determinants that can be constructed from a given set of orbitals with the constraint that some specified pairs of - and -spinorbitals must occur in all determinants (these are the inactive doubly occupied spatial orbitals). o Multireference CI wavefunctions are obtained by applying the excitation operators to the individual CSFs or determinants of the MCSCF (or CASSCF) reference wave function. Internally-contracted MRCI: its. unc. edu 40

Coupled-Cluster Theory § System of equations is solved iteratively (the convergence is accelerated by utilizing Pulay’s method, “direct inversion in the iterative subspace”, DIIS). § CCSDT model is very expensive in terms of computer resources. Approximations are introduced for the triples: CCSD(T), CCSD[T], CCSD-T. § Brueckner coupled-cluster (e. g. , BCCD) methods use Brueckner orbitals that are optimized such that singles don’t contribute. § By omitting some of the CCSD terms, the quadratic CI method (e. g. , QCISD) is obtained. its. unc. edu 41

Møller-Plesset Perturbation Theory § The Hartree-Fock function is an eigenfunction of the n-electron operator . § We apply perturbation theory as usual after decomposing the Hamiltonian into two parts: MP 2, MP 3, MP 4, …etc. number denotes order to which energy is computed (2 n+1 rule) § More complicated with more than one reference determinant (e. g. , MR-PT, CASPT 2, CASPT 3, …) its. unc. edu 42

Semi-Empirical Methods § These methods are derived from the Hartee–Fock model, that is, § § § its. unc. edu they are MO-LCAO methods. They only consider the valence electrons. A minimal basis set is used for the valence shell. Integrals are restricted to one- and two-center integrals and subsequently parametrized by adjusting the computed results to experimental data. Very efficient computational tools, which can yield fast quantitative estimates for a number of properties. Can be used for establishing trends in classes of related molecules, and for scanning a computational poblem before proceeding with highlevel treatments. A not of elements, especially transition metals, have not be parametrized 43

Semi-Empirical Methods Number 2 -electron integrals ( u|ls) is n 4/8, n = number of basis functions Treat only valence electrons explicit Neglect large number of 2 -electron integrals Replace others by empirical parameters Models: • • • its. unc. edu Complete Neglect of Differential Overlap (CNDO) Intermediate Neglect of Differential Overlap (INDO/MINDO) Neglect of Diatomic Differential Overlap (NDDO/MNDO, AM 1, PM 3) 44

Approximations of 1 -e integrals U from atomic spectra VAB value per atom pair , u on the same atom One parameter per element its. unc. edu 45
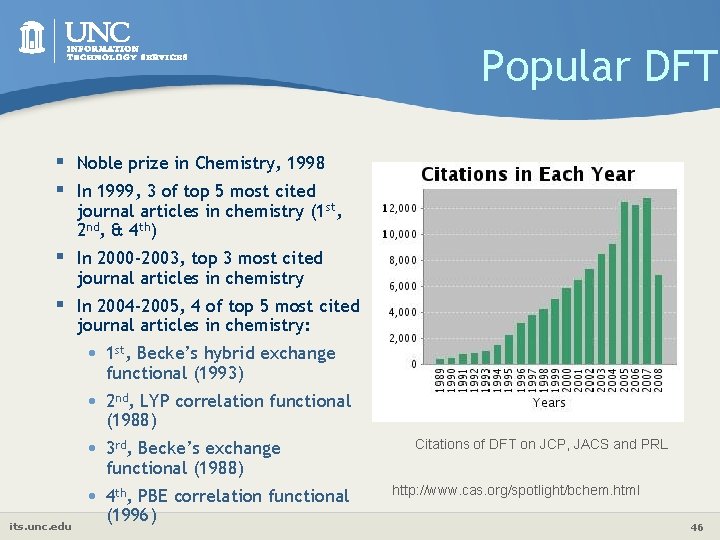
Popular DFT § Noble prize in Chemistry, 1998 § In 1999, 3 of top 5 most cited journal articles in chemistry (1 st, 2 nd, & 4 th) § In 2000 -2003, top 3 most cited journal articles in chemistry § In 2004 -2005, 4 of top 5 most cited journal articles in chemistry: its. unc. edu • 1 st, Becke’s hybrid exchange functional (1993) • 2 nd, LYP correlation functional (1988) • 3 rd, Becke’s exchange functional (1988) • 4 th, PBE correlation functional (1996) Citations of DFT on JCP, JACS and PRL http: //www. cas. org/spotlight/bchem. html 46

Brief History of DFT § First speculated 1920’ • Thomas-Fermi (kinetic energy) and Dirac (exchange energy) formulas § Officially born in 1964 with Hohenberg. Kohn’s original proof § GEA/GGA formulas available later 1980’ § Becoming popular later 1990’ § Pinnacled in 1998 with a chemistry Nobel prize its. unc. edu 47

What could expect from DFT? § § LDA, ~20 kcal/mol error in energy GGA, ~3 -5 kcal/mol error in energy G 2/G 3 level, some systems, ~1 kcal/mol Good at structure, spectra, & other properties predictions § Poor in H-containing systems, TS, spin, excited states, etc. its. unc. edu 48

Density Functional Theory § Two Hohenberg-Kohn theorems: • “Given the external potential, we know the ground-state energy of the molecule when we know the electron density ”. • The energy density functional is variational. its. unc. edu 49
![But what is E[ ]? E[ ] = TS[ ] + Vne[ ] + But what is E[ ]? E[ ] = TS[ ] + Vne[ ] +](http://slidetodoc.com/presentation_image_h/bd5f3e42f1972ae383b7f12c672c605e/image-50.jpg)
But what is E[ ]? E[ ] = TS[ ] + Vne[ ] + J[ ] + Vnn[ ] + Exc[ ] § How do we compute the energy if the density is known? w The Coulombic interactions are easy to compute: w But what about the kinetic energy TS[ ] and exchangecorrelation energy Exc[ ]? its. unc. edu 50

Kohn-Sham Scheme • Suppose, we know the exact density. • Then, we can formulate a Slater determinant that generates this exact density (= Slater determinant of system of N non-interacting electrons with same density ). The Only Unknown • We know how to compute the kinetic energy Ts exactly from a Slater determinant. • Then, the only thing unknown is to calculate Exc[ ]. its. unc. edu 51

All about Exchange-Correlation Energy Density Functional § LDA – f(r) is a function of (r) only § GGA – f(r) is a function of (r) and |∇ (r)| § Mega-GGA – f(r) is also a function of ts(r), kinetic energy density § Hybrid – f(r) is GGA functional with extra contribution from Hartree. Fock exchange energy its. unc. edu Jacob's ladder for the five generation of DFT functionals, according to the vision of John Perdew with indication of some of the most common DFT functionals within each rung. 52

LDA Functionals § Thomas-Fermi formula (Kinetic) – 1 parameter § Slater form (exchange) – 1 parameter § Wigner correlation – 2 parameters its. unc. edu 53

Popular Functional: BLYP/B 3 LYP Two most well-known functionals are the Becke exchange functional Ex[ ] with 2 extra parameters & The Lee-Yang-Parr correlation functional Ec[ ] with 4 parameters a-d Together, they constitute the BLYP functional: The B 3 LYP functional is augmented with 20% of Hartree-Fock exchange: its. unc. edu 54
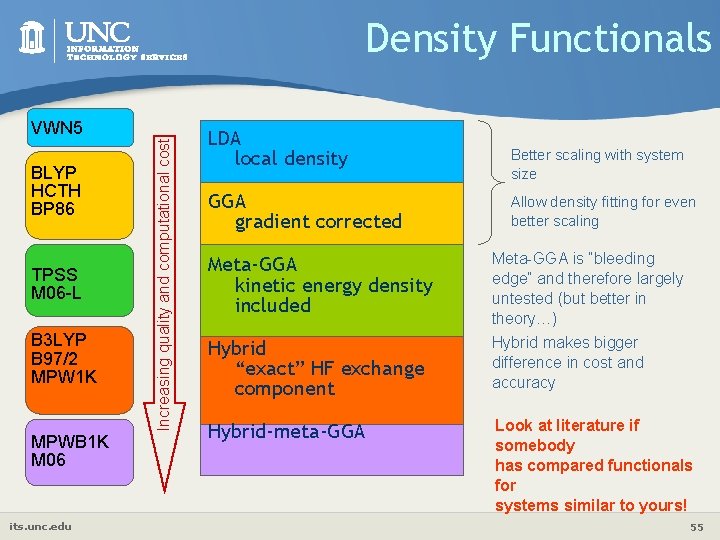
VWN 5 BLYP HCTH BP 86 TPSS M 06 -L B 3 LYP B 97/2 MPW 1 K MPWB 1 K M 06 its. unc. edu Increasing quality and computational cost Density Functionals LDA local density GGA gradient corrected Meta-GGA kinetic energy density included Hybrid “exact” HF exchange component Hybrid-meta-GGA Better scaling with system size Allow density fitting for even better scaling Meta-GGA is “bleeding edge” and therefore largely untested (but better in theory…) Hybrid makes bigger difference in cost and accuracy Look at literature if somebody has compared functionals for systems similar to yours! 55
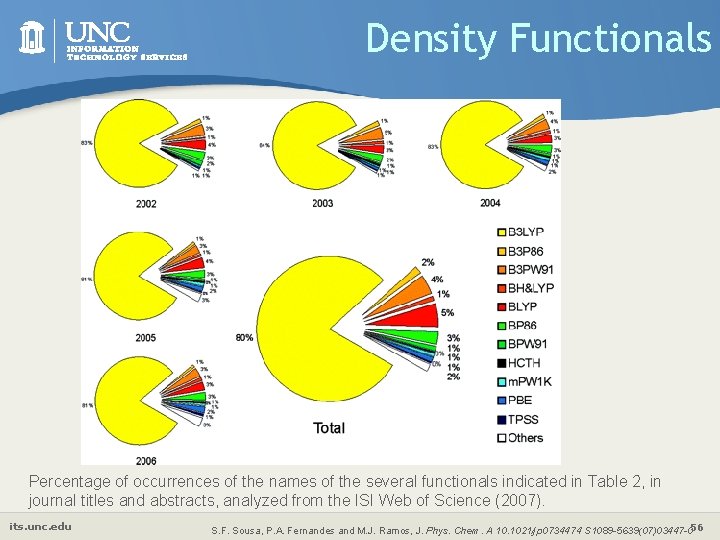
Density Functionals Percentage of occurrences of the names of the several functionals indicated in Table 2, in journal titles and abstracts, analyzed from the ISI Web of Science (2007). its. unc. edu S. F. Sousa, P. A. Fernandes and M. J. Ramos, J. Phys. Chem. A 10. 1021/jp 0734474 S 1089 -5639(07)03447 -056

Problems with DFT § ground-state theory only § universal functional still unknown § even hydrogen atom a problem: self-interaction correction § no systematic way to improve approximations like LDA, GGA, etc. § extension to excited states, spin multiplets, etc. , though proven exact in theory, is not trivial in implementation and still far from being generally accessible thus far its. unc. edu 57

DFT Developments § Theoretical • Extensions to excited states, etc. • Better functionals (mega-GGA), etc • Understanding functional properties, etc. § Conceptual • More concepts proposed, like electrophilicity, spin • philicity, surfaced-integrated Fukui fnc Dynamic behaviors, profiles, etc. § Computational • Linear scaling methods • QM/MM related issues • Applications its. unc. edu 58
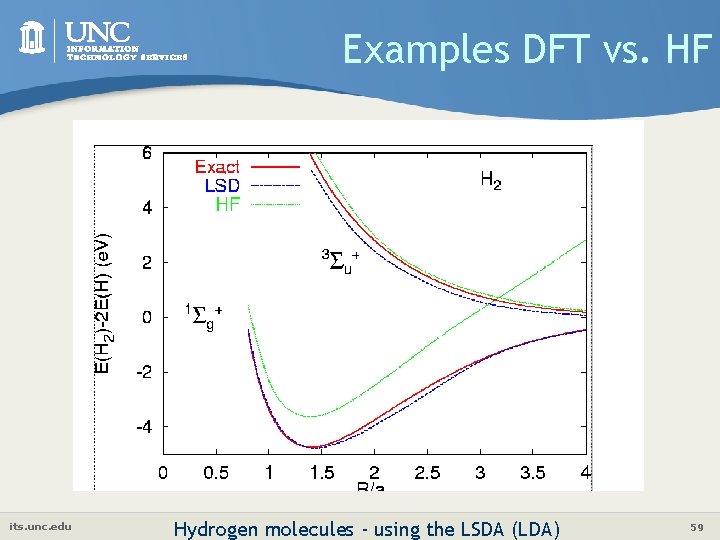
Examples DFT vs. HF its. unc. edu Hydrogen molecules - using the LSDA (LDA) 59

Chemical Reactivity Theory Chemical reactivity theory quantifies the reactive propensity of isolated species through the introduction of a set of reactivity indices or descriptors. Its roots go deep into the history of chemistry, as far back as the introduction of such fundamental concepts as acid, base, Lewis acid, Lewis base, etc. It pervades almost all of chemistry. -- Morrel H. Cohen, and Adam Wasserman, J. Phys. Chem. A 2007, 111, 2229 § Molecular Orbital Theory • • Fukui’s Frontier Orbital (HOMO/LUMO) model Woodward-Hoffman rules Well developed: Nobel prize in Chemistry, 1981 Problem: conceptual simplicity disappears as computational accuracy increases because it’s based on the molecular orbital description § Density Functional Theory (DFT) • Conceptual DFT, also called Chemical DFT, DF Reactivity Theory • Proposed by Robert G. Parr of UNC-CH, 1980 s • Still in development its. unc. edu 60
![DFT Reactivity Theory § General Consideration • E E [N, (r)] E [ ] DFT Reactivity Theory § General Consideration • E E [N, (r)] E [ ]](http://slidetodoc.com/presentation_image_h/bd5f3e42f1972ae383b7f12c672c605e/image-61.jpg)
DFT Reactivity Theory § General Consideration • E E [N, (r)] E [ ] • Taylor Expansion: Perturbation resulted from an external attacking agent leading to changes in N and (r), N and (r), Assumptions: existence and well-behavior of all above partial/functional derivatives its. unc. edu 61
![Conceptual DFT § Basic assumptions • E E [N, (r)] E [ ] • Conceptual DFT § Basic assumptions • E E [N, (r)] E [ ] •](http://slidetodoc.com/presentation_image_h/bd5f3e42f1972ae383b7f12c672c605e/image-62.jpg)
Conceptual DFT § Basic assumptions • E E [N, (r)] E [ ] • Chemical processes, responses, and changes expressible via Taylor expansion • Existence, continuous, and well-behavedness of the partial derivatives its. unc. edu 62

DFT Reactivity Indices § Electronegativity (chemical potential) § Hardness / Softness § Maximum Hardness Principle (MHP) its. unc. edu 63
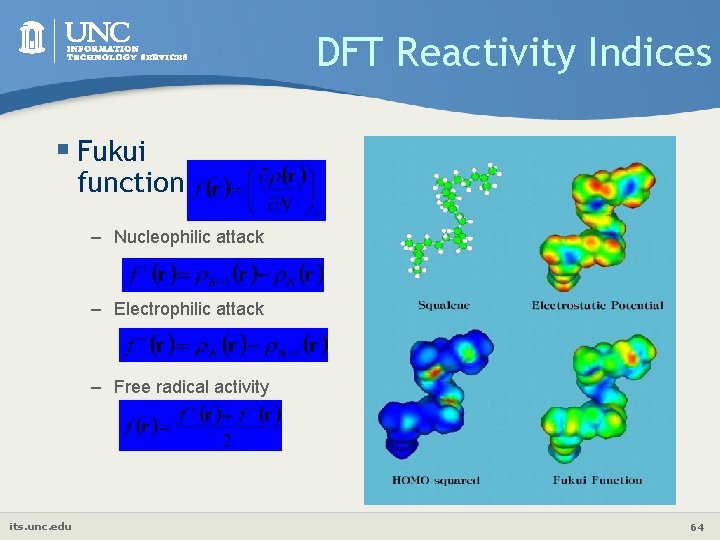
DFT Reactivity Indices § Fukui function – Nucleophilic attack – Electrophilic attack – Free radical activity its. unc. edu 64
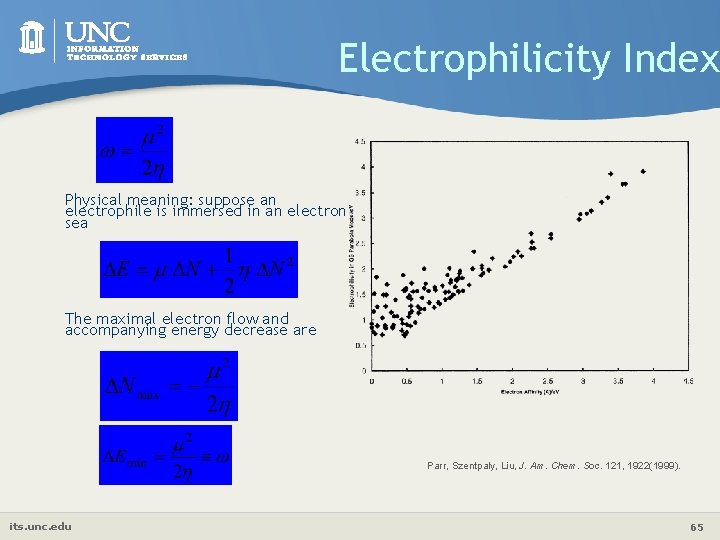
Electrophilicity Index Physical meaning: suppose an electrophile is immersed in an electron sea The maximal electron flow and accompanying energy decrease are Parr, Szentpaly, Liu, J. Am. Chem. Soc. 121, 1922(1999). its. unc. edu 65
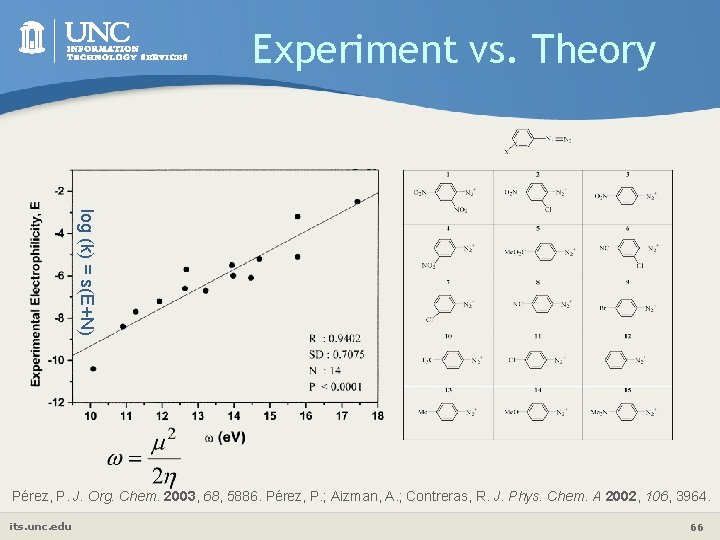
Experiment vs. Theory log (k) = s(E+N) Pérez, P. J. Org. Chem. 2003, 68, 5886. Pérez, P. ; Aizman, A. ; Contreras, R. J. Phys. Chem. A 2002, 106, 3964. its. unc. edu 66

Minimum Electrophilicity Principle § Analogous to the maximum hardness principle (MHP) § Separately proposed by Noorizadeh and Chattaraj § Concluded that “the natural direction of a chemical reaction is toward a state of minimum electrophilicity. ” Noorizadeh, S. Chin. J. Chem. 2007, 25, 1439. Noorizadeh, S. J. Phys. Org. Chem. 2007, 20, 514. Chattaraj, P. K. Ind. J. Phys. Proc. Ind. Natl. Sci. Acad. Part A 2007, 81, 871. non- 1 2 3 4 5 6 7 LA Aa -0. 091 -0. 085 -0. 093 - -0. 087 -0. 083 -0. 090 -0. 087 -0. 0842 - 0. 088 Bb -0. 089 -0. 084 -0. 088 -0. 089 0. 087 Aa -0. 172 -0. 247 -0. 230 -0. 220 - 0. 0892 -0. 226 -0. 2518 0. 218 Bb -0. 171 -0. 246 -0. 247 -0. 233 0. 221 0. 2161 -0. 226 -0. 2506 0. 2157 its. unc. edu 67 Yue Xia, Dulin Yin, Chunying Rong, Qiong Xu, Donghong Yin , and Shubin Liu, J. Phys. Chem. A, 2008, 112, 9970.
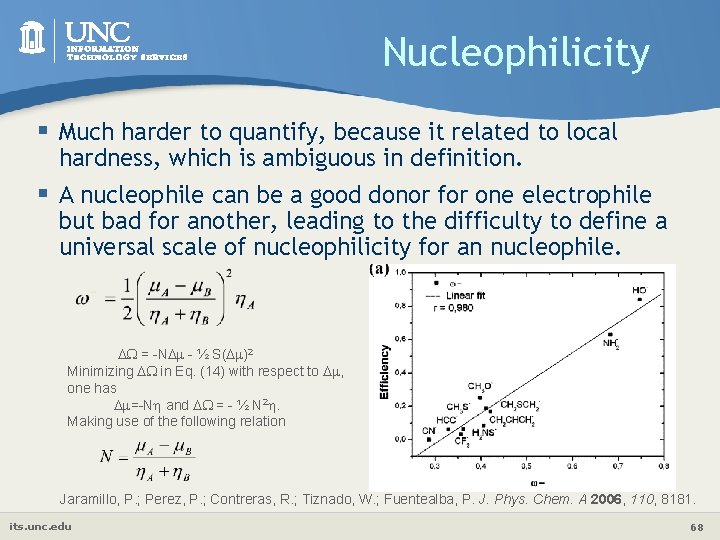
Nucleophilicity § Much harder to quantify, because it related to local hardness, which is ambiguous in definition. § A nucleophile can be a good donor for one electrophile but bad for another, leading to the difficulty to define a universal scale of nucleophilicity for an nucleophile. = -N - ½ S( )2 Minimizing in Eq. (14) with respect to , one has =-N and = - ½ N 2. Making use of the following relation Jaramillo, P. ; Perez, P. ; Contreras, R. ; Tiznado, W. ; Fuentealba, P. J. Phys. Chem. A 2006, 110, 8181. its. unc. edu 68

Philicity and Fugality § Philicity: defined as ·f(r) • • Chattaraj, Maiti, & Sarkar, J. Phys. Chem. A 107, 4973(2003) Still a very controversial concept, see JPCA 108, 4934(2004); Chattaraj, et al. JPCA, in press. § Spin-Philicity: defined same as but in spin resolution • Perez, Andres, Safont, Tapia, & Contreras. J. Phys. Chem. A 106, 5353(2002) § Nuclofugality & Electrofugality Ayers, P. W. ; Anderson, J. S. M. ; Rodriguez, J. I. ; Jawed, Z. Phys. Chem. Phys. 2005, 7, 1918. Ayers, P. W. ; Anderson, J S. M. ; Bartolotti, L. J. Int. J. Quantum Chem. 2005, 101, 520. its. unc. edu 69
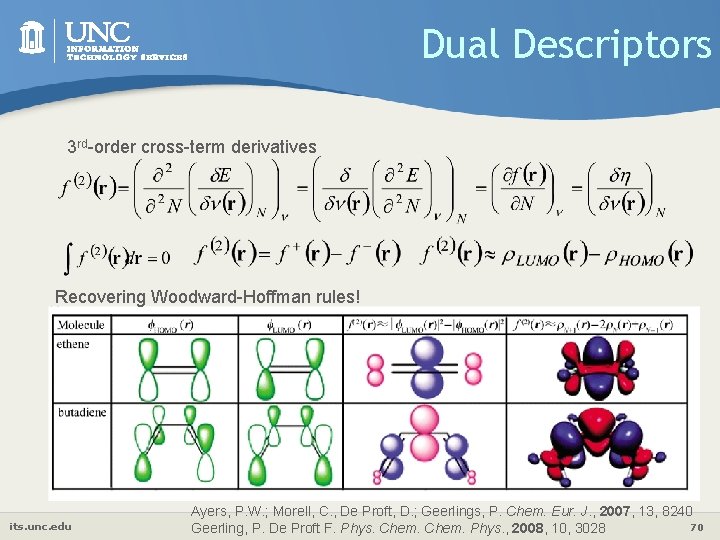
Dual Descriptors 3 rd-order cross-term derivatives Recovering Woodward-Hoffman rules! its. unc. edu Ayers, P. W. ; Morell, C. , De Proft, D. ; Geerlings, P. Chem. Eur. J. , 2007, 13, 8240 70 Geerling, P. De Proft F. Phys. Chem. Phys. , 2008, 10, 3028

Steric Effect § one of the most widely used concepts in chemistry § originates from the space occupied by atom in a molecule § previous work attributed to the electron exchange correlation § Weisskopf thought of as “kinetic energy pressure” its. unc. edu Weisskopf, V. F. , Science 187, 605 -612(1975). 71
![Steric effect: a DFT description Assume since E[ ] ≡ Es[ ] + Ee[ Steric effect: a DFT description Assume since E[ ] ≡ Es[ ] + Ee[](http://slidetodoc.com/presentation_image_h/bd5f3e42f1972ae383b7f12c672c605e/image-72.jpg)
Steric effect: a DFT description Assume since E[ ] ≡ Es[ ] + Ee[ ] + Eq[ ] E[ ] = Ts[ ] + Vne[ ] + J[ ] + Vnn[ ] + Exc[ ] Ee[ ] = Vne[ ] + J[ ] + Vnn[ ] Eq[ ] = Exc[ ] + EPauli[ ] = Exc[ ] + Ts[ ] - Tw[ ] Es[ ] ≡ E[ ] - Ee[ ] - Eq[ ] = Tw[ ] we have S. B. Liu, J. Chem. Phys. 2007, 126, 244103. S. B. Liu and N. Govind, J. Phys. Chem. A 2008, 112, 6690. S. B. Liu, N. Govind, and L. G. Pedersen, J. Chem. Phys. 2008, 129, 094104. M. Torrent-Sucarrat, S. B. Liu and F. De Proft, J. Phys. Chem. A 2009, 113, 3698. its. unc. edu 72
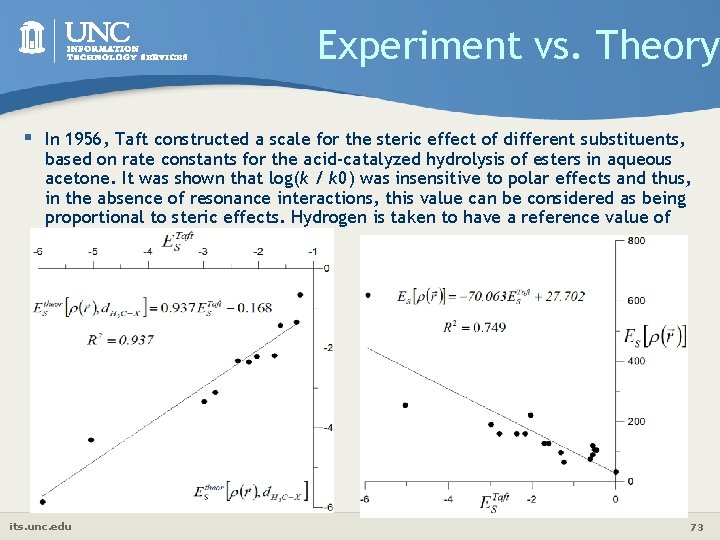
Experiment vs. Theory § In 1956, Taft constructed a scale for the steric effect of different substituents, based on rate constants for the acid-catalyzed hydrolysis of esters in aqueous acetone. It was shown that log(k / k 0) was insensitive to polar effects and thus, in the absence of resonance interactions, this value can be considered as being proportional to steric effects. Hydrogen is taken to have a reference value of Es. Taft= 0 its. unc. edu 73
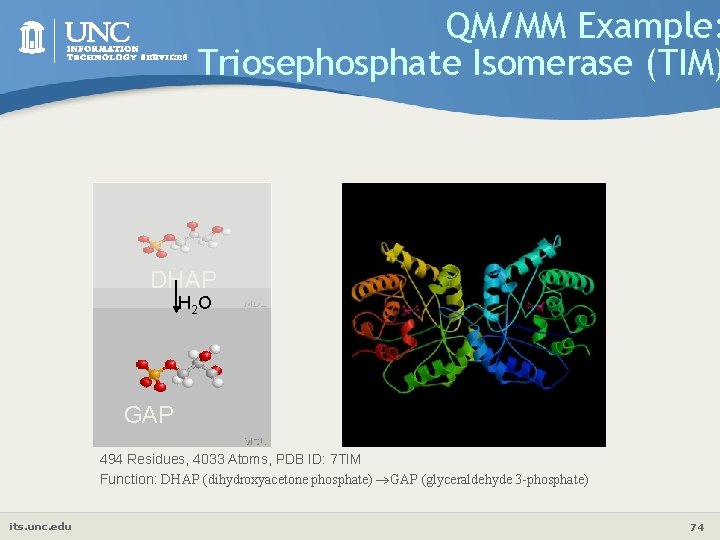
QM/MM Example: Triosephosphate Isomerase (TIM) DHAP H 2 O GAP 494 Residues, 4033 Atoms, PDB ID: 7 TIM Function: DHAP (dihydroxyacetone phosphate) GAP (glyceraldehyde 3 -phosphate) its. unc. edu 74
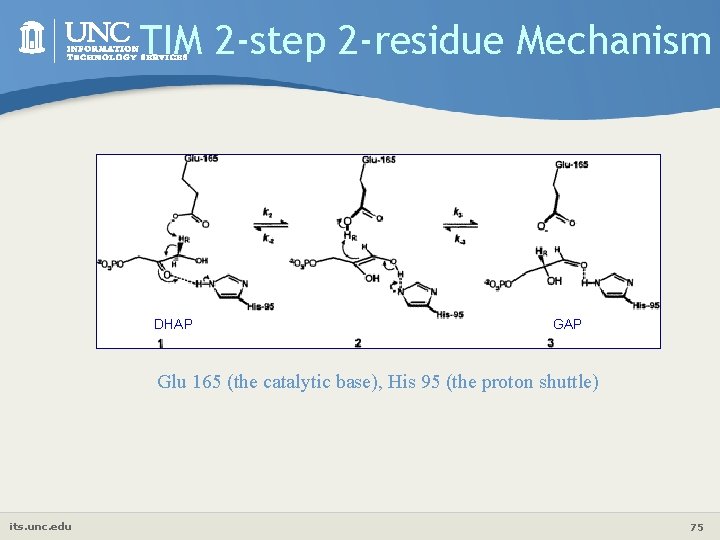
TIM 2 -step 2 -residue Mechanism DHAP Glu 165 (the catalytic base), His 95 (the proton shuttle) its. unc. edu 75
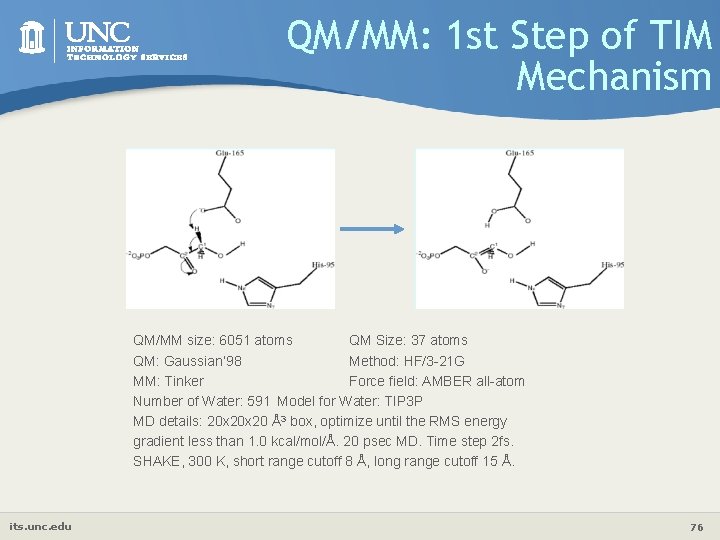
QM/MM: 1 st Step of TIM Mechanism QM/MM size: 6051 atoms QM Size: 37 atoms QM: Gaussian’ 98 Method: HF/3 -21 G MM: Tinker Force field: AMBER all-atom Number of Water: 591 Model for Water: TIP 3 P MD details: 20 x 20 Å3 box, optimize until the RMS energy gradient less than 1. 0 kcal/mol/Å. 20 psec MD. Time step 2 fs. SHAKE, 300 K, short range cutoff 8 Å, long range cutoff 15 Å. its. unc. edu 76
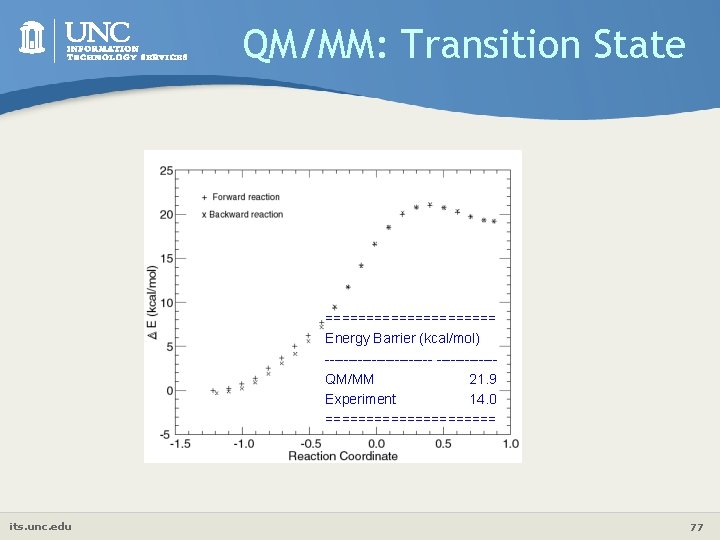
QM/MM: Transition State =========== Energy Barrier (kcal/mol) ------------------QM/MM 21. 9 Experiment 14. 0 =========== its. unc. edu 77

What’s New: Linear Scaling O(N) Method § Numerical Bottlenecks: • • • diagonalization ~N 3 orthonormalization ~N 3 matrix element evaluation ~N 2 -N 4 § Computational Complexity: N log N § Theoretical Basis: near-sightedness of density matrix or orbitals § Strategy: • • sparsity of localized orbital or density matrix direct minimization with conjugate gradient Diagonalization OLMO NOLMO § Models: divide-and-conquer and variational methods § Applicability: ~10, 000 atoms, dynamics its. unc. edu 78

What Else … ? § Solvent effect • Implicit model vs. explicit model § § § its. unc. edu Relativity effect Transition state Excited states Temperature and pressure Solid states (periodic boundary condition) Dynamics (time-dependent) 79

Limitations and Strengths of ab initio quantum chemistry its. unc. edu 80

Popular QM codes its. unc. edu Gaussian (Ab Initio, Semi-empirical, DFT) Gamess-US/UK (Ab Initio, DFT) Spartan (Ab Initio, Semi-empirical, DFT) NWChem (Ab Initio, DFT, MD, QM/MM) MOPAC/2000 (Semi-Empirical) DMol 3/CASTEP (DFT) Molpro (Ab initio) ADF (DFT) ORCA (DFT) 81

Reference Books § Computational Chemistry (Oxford Chemistry Primer) G. H. Grant and W. G. Richards (Oxford University Press) § Molecular Modeling – Principles and Applications, A. R. Leach (Addison Wesley Longman) § Introduction to Computational Chemistry, F. Jensen (Wiley) § Essentials of Computational Chemistry – Theories and Models, C. J. Cramer (Wiley) § Exploring Chemistry with Electronic Structure Methods, J. B. Foresman and A. Frisch (Gaussian Inc. ) its. unc. edu 82
Questions & Comments Please direct comments/questions about research computing to E-mail: research@unc. edu Please direct comments/questions pertaining to this presentation to E-Mail: shubin@email. unc. edu The PPT format of this presentation is available here: http: //its 2. unc. edu/divisions/rc/training/scientific/ /afs/isis/depts/its/public_html/divisions/rc/training/scientific/short_courses/ its. unc. edu 83

Hands-on: Part I Purpose: to get to know the available ab initio and semi-empirical methods in the Gaussian 03 / Gauss. View package • ab initio methods w Hartree-Fock w MP 2 w CCSD • Semiempirical methods w AM 1 The WORD. doc format of this hands-on exercises is available here: http: //its 2. unc. edu/divisions/rc/training/scientific/ /afs/isis/depts/its/public_html/divisions/rc/training/scientific/short_courses/lab. Directions_compchem_2009. doc its. unc. edu 84

Hands-on: Part II Purpose: To use LDA and GGA DFT methods to calculate IR/Raman spectra in vacuum and in solvent. To build QM/MM models and then use DFT methods to calculate IR/Raman spectra • DFT w LDA (SVWN) w GGA (B 3 LYP) • QM/MM its. unc. edu 85
- Slides: 85