Introduction to Clinical Trials Afshin Ostovar Bushehr University

Introduction to Clinical Trials Afshin Ostovar Bushehr University of Medical Sciences Bushehr, 2011 12/15/2021 1

Research Design Epidemiology l Descriptive Studies l l Case Reports Case Series Cross Sectional Survey Analytic Studies l Observational Studies l l l Intervention Studies l 12/15/2021 Case-Control or Case-Comparison Cohort Studies Clinical Trials 2

Definition • A clinical trial is defined as a prospective study comparing the effect and value of intervention(s) against a control in human beings. • They need not all be followed from an identical calendar date. In fact, this will occur only rarely. Each participant, however, must be followed from a well-defined point, which becomes time zero or baseline for the study. 12/15/2021 3

Definition “cont’d” • A clinical trial must employ one or more intervention techniques. These may be "prophylactic, diagnostic or therapeutic agents, devices, regimens, procedures, etc. “ • Intervention techniques should be applied to participants in a standard fashion in an effort to change some aspect of the participants. • Follow-up of people over time without active intervention may measure the natural history of a disease process, but it does not constitute a clinical trial. Without active intervention the study is observational because no experiment is being performed. 12/15/2021 4

Definition “cont’d” l A clinical trial must contain a control group against which the intervention group is compared. l At baseline, the control group must be sufficiently similar in relevant respects to the intervention group so that differences in outcome may reasonably be attributed to the action of the intervention. l Most often a new intervention is compared with best current standard therapy. If no such standard exists, the people in the intervention group may be compared with people who are on no active intervention. "No active intervention" means that the participant may receive either a placebo or no intervention at all. 12/15/2021 5

Counterfactual concept l The effect of any treatment for a given patient is the difference between what happened to the patient as a result of giving the treatment and what would have happened had treatment been denied. (counterfactual view) 12/15/2021 6

Phases of Clinical Trials l l 12/15/2021 Phase III Phase IV 7

Clinical trial designs 12/15/2021 8
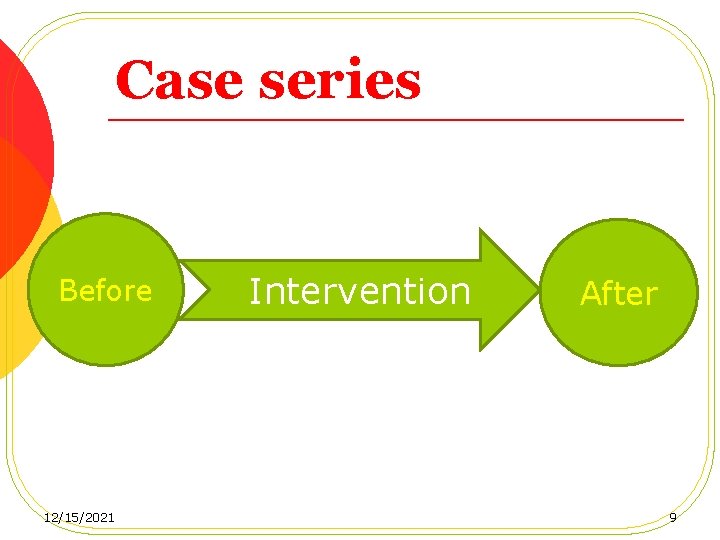
Case series Before 12/15/2021 Intervention After 9
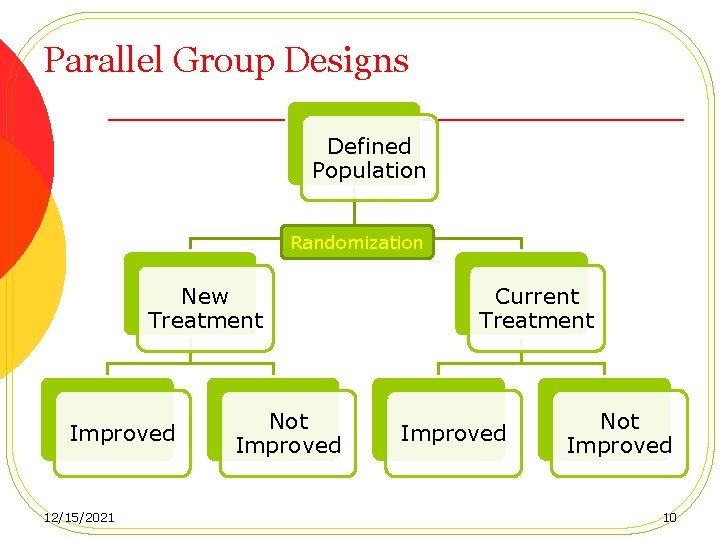
Parallel Group Designs Defined Population Randomization New Treatment Improved 12/15/2021 Not Improved Current Treatment Improved Not Improved 10

Parallel Group Designs (Adventages) • Simple and easy to implement • Universally accepted • Applicable to acute conditions • Analysis is less complicated and interpretation of the results is straightforward 12/15/2021 11

Parallel Group Designs (Types) • Group comparison (parallel-group) design • Two-group design For ethical consideration with control (placebo), we can allocate patients unequally to treatment groups (at random) to allow more patients to receive the treatment. • • Three-group design Matched pairs parallel designs • • Power increased Two drawbacks: » » 12/15/2021 The prognostic characteristics are not easily defined Patient recruitment is usually slow 12

Parallel group designs (run in periods) l Advantages Acts as a washout period to remove effects of previous therapy l Can be used to obtain baseline data and to evaluate if patient fulfills study entry criteria l Can be used as a training period for patients and investigators l Helps in identifying placebo responders l Provides useful information regarding patient compliance l l Disadvantage: l Increases the length of a study: l l Extra visit and costs Decreases in enthusiasm of patients and investigators

Crossover Designs l A crossover design is a modified randomized block design in which each block receives more than one treatment at different periods. l A p × q crossover design: there are p sequences of treatments administered at q different periods 12/15/2021 14
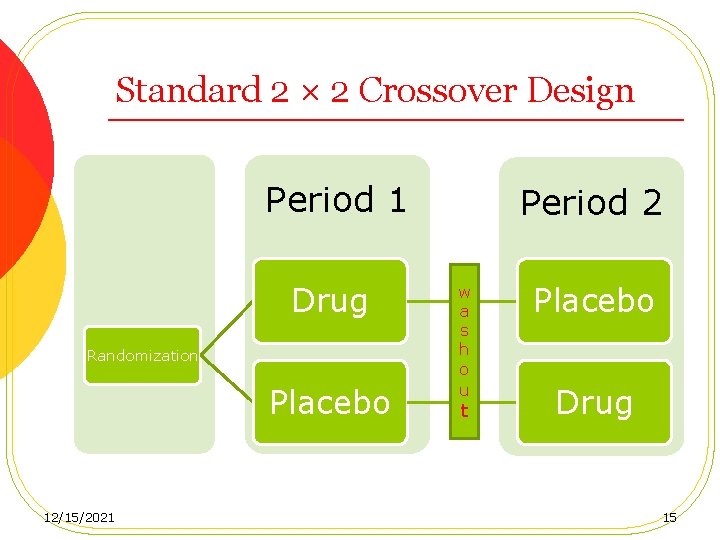
Standard 2 × 2 Crossover Design Period 1 Drug Randomization Placebo 12/15/2021 Period 2 w a s h o u t Placebo Drug 15

Crossover Designs Advantages: Ø Allows within-patients comparisons of treatments Ø Removes intrapatient variability Ø Provides the best unbiased estimates for the differences between treatments Ø Decreases number of patients needed 12/15/2021 16
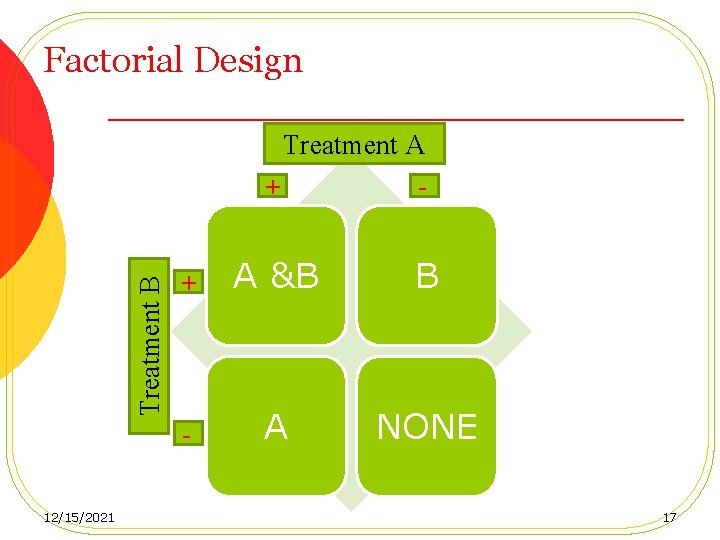
Factorial Design Treatment B Treatment A 12/15/2021 + - + A &B B - A NONE 17

Factorial Design Two applications: 1. 2. Quantifying the interaction between the two treatments Opportunistic situations Treatments groups: AO BO AB OO 12/15/2021 18

Designs with ethical considerations l l l Adaptive Randomization Zelen design Variations of placebo-controlled trials: Add-on design l Replacement design l Randomized Withdrawal design l l Sequential analysis 12/15/2021 19

Multicenter trials • The limitations of patient population in a single center and/or sources and capacity in a single center make a multicenter trial justified. • A multicenter study is a single study involving several study centers. The data collected from these centers are intended to be analyzed as a whole. • At each center an identical study protocol is used. A center or site is considered a natural blocking or stratified variable. • A rule of thumb is that the number of patients in each center should not be less than the number of centers 12/15/2021 20

The Randomization Process l The randomized clinical trial is the standard by which all trials are judged l In the simplest case, randomization is a process by which each participant has the same chance of being assigned to either intervention or control 12/15/2021 21

Purposes of Randomization l To generate comparative groups l To enable valid statistical tests 12/15/2021 22

Blindness • Open label (unblinding) • Single blinding (patients only) • Double blinding (patients and investigators) • Triple blinding (patients and investigators and Monitoring investigators) 12/15/2021 23

Classic classification of Randomization Simple l Systematic l Balanced block l Stratified l 12/15/2021 24

Complete randomization (Simple randomization) l Random allocation It is a form of restricted randomization It randomly selects the N/2 out of a total of N patients without replacement and assigns these N/2 patients to receive the test drug and the other half to receive the placebo. 12/15/2021 25

Permuted block randomization (Balanced block randomization) l One of the major disadvantages of simple randomization is that treatment imbalance can occur periodically l If the demographic factors or baseline characteristics change over time, it is quite possible to have a serious covariate imbalance between treatment groups. 12/15/2021 26
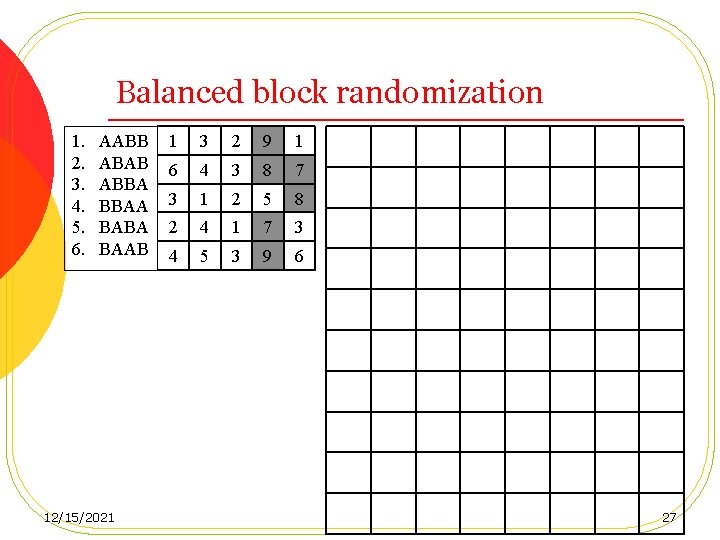
Balanced block randomization 1. 2. 3. 4. 5. 6. AABB ABAB ABBA BBAA BABA BAAB 12/15/2021 1 3 2 9 1 6 4 3 8 7 3 1 2 5 8 2 4 1 7 3 4 5 3 9 6 27

Treatment adaptive randomization l l The Treatment adaptive randomization adjusts for the assigning probability of the current patient with respect to the number of patients who have been randomized to each treatment group. Methods: l biased coin randomization l l Urn randomization l l 12/15/2021 A constant assigning probability is used during the entire course of the study The probability of the assignment of the current patient is a function of the current treatment imbalance It requires a much more complicated analysis 28

Steps in Analysis l l Baseline data analysis Main analysis l Protocol deviation l l l 12/15/2021 Intention to treat and per protocol approaches Covariates analysis Multiple outcomes analysis Subgroup analysis Multiplicity in analysis of clinical trials 29

Baseline Data Analysis • To: – – • The variables should be considered: – – • Check generalizability Check comparability of treatment groups The characteristics of the disease (type, severity, duration, …) Prognostic variables Other coincidence diseases Previous treatments Use of statistical tests! 12/15/2021 30

Main analysis • Three approaches: Ø Outcome analysis Ø Change score analysis Ø Analysis of Covariance (ANCOVA) 12/15/2021 31

Covariates analysis • Covariate = prognostic factor = confounder • In the case where covariates are not balanced between the treatment groups, to obtain a valid inference of treatment effect, it is necessary to adjust for covariates • An adjustment of covariates not only provides unbiased statistical inference but also increases precision of the statistical inference 12/15/2021 32

Study Protocol • The study protocol can be viewed as a written agreement between the investigator, the participants, and the scientific community. • The contents provide the background, specify the objectives and describe the design and organization of the trial. • Every detail explaining how the trial is carried out does not need to be included, provided that the comprehensive manual of procedures contains such information. 12/15/2021 33

Study Protocol • • • The protocol serves as a document to assist communication among those working in the trial. It should be also be made available to others on request. The protocol should be developed before the beginning of participant enrollment and should remain essentially unchanged except perhaps for minor updates. 12/15/2021 34

Study Protocol l Careful thought and justification should go into any changes. l Major revisions that alter the direction of the trial should be rare. l If they occur, the rational behind such changes need to be clearly descried. 12/15/2021 35

A. Background of the study 1. Reviewing the related articles. 2. Why is the results of this trial needed? 3. How does the study provide evidence for decision making? 12/15/2021 36

B. Objectives 1. 2. 3. 4. 12/15/2021 Primary question and response variable Secondary question and response variables Subgroup hypothesis Adverse effects 37

C. Design of the study 1. 12/15/2021 Study population a) Inclusion criteria b) Exclusion Criteria 38
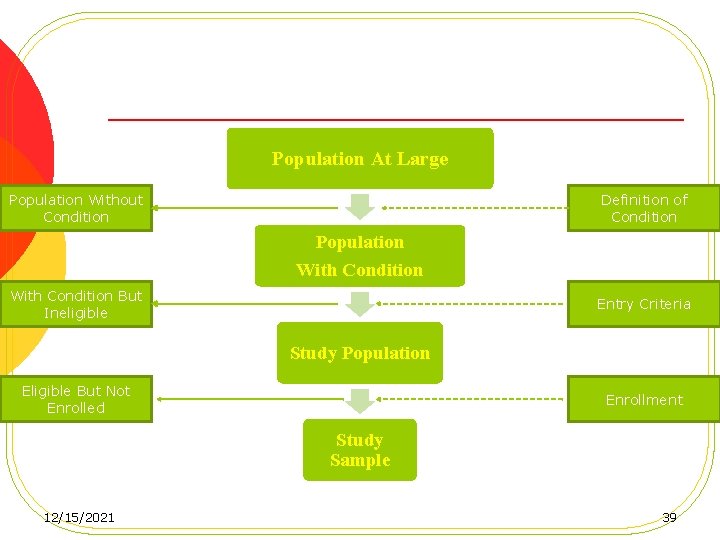
Population At Large Population Without Condition Definition of Condition Population With Condition But Ineligible Entry Criteria Study Population Eligible But Not Enrolled Enrollment Study Sample 12/15/2021 39

C. Design of the study 2. Sample Size assumptions and estimates 12/15/2021 40

C. Design of the study 3. Enrollment of participants a. b. c. d. 12/15/2021 Informed consent Assessment of eligibility Baseline examination Intervention allocation (e. g. , Randomization method) 41

C. Design of the study 4. Intervention a. b. 12/15/2021 Description and schedule Measures of compliance 42

C. Design of the study 5. Follow-up visit description and schedule 12/15/2021 43

C. Design of the study 6. Ascertainment of response variables a. b. c. 12/15/2021 Training Data collection Quality control 44
C. Design of the study 7. Data analysis a. b. 12/15/2021 Interim monitoring Final analysis 45

C. Design of the study 8. Termination policy 12/15/2021 46

D. Organization 1. Participating Investigators a. Statistical unit or data coordinating center b. Laboratories and other special units c. Clinical center(s) 12/15/2021 47

D. Organization 2. Study administration a. b. c. 12/15/2021 Steering committees and subcommittees Data monitoring committee Funding organization 48

Appendix l l Definitions of eligibility criteria Definition of response variables 12/15/2021 49

Any Question? Thank you 12/15/2021 50
- Slides: 50